Advanced Synthesis of Unprotected Single-Arm Sugar Compounds for Commercial Glycopolymer Production
The landscape of bio-material synthesis is undergoing a significant transformation driven by the need for more efficient and biocompatible building blocks. Patent CN114773412A introduces a groundbreaking methodology for the preparation of non-protecting group single-arm sugar-containing compounds, which serve as critical precursors for advanced glycopolymers. Unlike traditional approaches that rely on complex protection-deprotection sequences, this innovation leverages a streamlined combination of Suzuki cross-coupling and thiol-ene click chemistry. This technical breakthrough addresses long-standing challenges in the synthesis of carbohydrate-based monomers, offering a pathway to high-purity intermediates with reduced environmental impact. For R&D teams focused on drug delivery systems and biosensors, this patent provides a robust framework for constructing multivalent ligands that mimic natural glycoproteins with high fidelity.
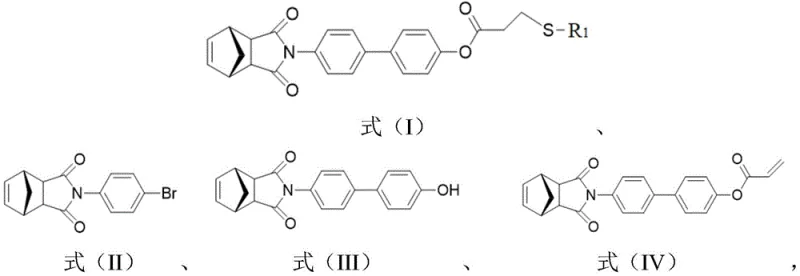
Traditional methods for synthesizing sugar-containing monomers often suffer from excessive step counts and low overall yields due to the necessity of protecting hydroxyl groups on the sugar moiety. Conventional routes typically involve multiple protection steps using silyl or benzyl groups, followed by polymerization, and finally, harsh deprotection conditions that can degrade the sensitive carbohydrate structure. This multi-step process not only increases the consumption of reagents and solvents but also generates substantial chemical waste, complicating purification and driving up manufacturing costs. Furthermore, the use of heavy metal catalysts in some traditional coupling methods requires rigorous removal steps to meet pharmaceutical purity standards, adding another layer of complexity to the production workflow.
In stark contrast, the novel approach detailed in this patent circumvents these bottlenecks by utilizing unprotected thiol-sugars directly in a thiol-ene Michael addition. By first constructing a rigid biphenyl-norbornene scaffold via Suzuki coupling, the method creates a stable platform for subsequent functionalization. The introduction of the terminal alkene via esterification with acryloyl chloride sets the stage for a highly efficient click reaction. This strategy eliminates the need for orthogonal protection strategies, thereby drastically simplifying the synthetic route. The result is a more direct path to the final monomer, preserving the integrity of the sugar units while ensuring high selectivity and yield under mild reaction conditions suitable for sensitive biological applications.
Mechanistic Insights into Suzuki Coupling and Thiol-Ene Click Chemistry
The core of this synthesis lies in the precise orchestration of palladium-catalyzed cross-coupling and radical-mediated addition. The process begins with the formation of a norbornene-imide derivative, which acts as a rigid spacer. The subsequent Suzuki coupling between the bromo-imide and 4-hydroxyphenylboronic acid is catalyzed by Pd(PPh3)4 in the presence of anhydrous sodium carbonate. This step is critical for establishing the biphenyl linkage, which provides the necessary spatial arrangement for protein recognition. The reaction proceeds through a standard catalytic cycle involving oxidative addition, transmetallation, and reductive elimination, facilitated by the electron-rich phosphine ligands that stabilize the palladium center. Following this, esterification with acryloyl chloride introduces the reactive alkene handle, converting the phenolic hydroxyl into an acrylate ester ready for conjugation.
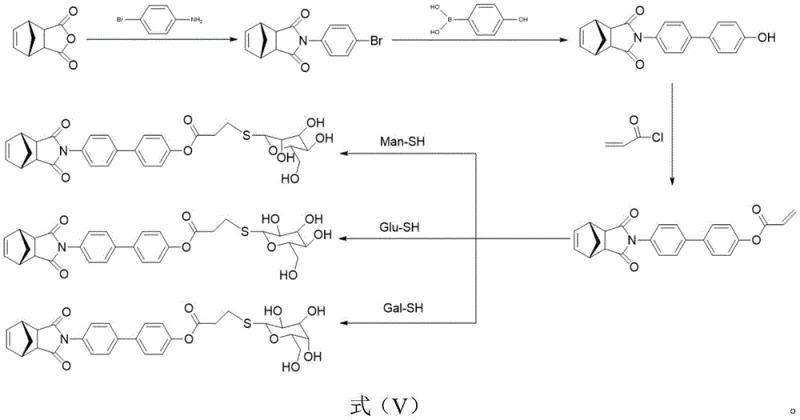
The final and most innovative step involves the thiol-ene Michael addition, a quintessential click chemistry reaction. In this stage, the acrylate-functionalized intermediate reacts with unprotected thiol-sugars such as thioglucose or thiomannose. The reaction is initiated by a base catalyst, specifically n-hexylamine, which facilitates the nucleophilic attack of the thiol group on the electron-deficient double bond of the acrylate. This mechanism ensures high regioselectivity and prevents side reactions that could compromise the sugar structure. The absence of protecting groups on the sugar during this step is particularly advantageous, as it avoids the steric hindrance and solubility issues often associated with protected carbohydrates. This direct conjugation method guarantees that the resulting monomer retains the native hydrogen-bonding capability of the sugar, which is essential for its biological function in protein recognition assays.
How to Synthesize Non-Protecting Single-Arm Sugar-Containing Compound Efficiently
The synthesis protocol outlined in the patent offers a reproducible and scalable method for producing these valuable intermediates. The process is divided into four distinct stages, each optimized for maximum conversion and ease of purification. Starting materials such as exo-norbornene-2,3-dicarboxylic anhydride and 4-bromoaniline are commercially available and cost-effective. The reaction conditions are moderate, with temperatures ranging from ambient to 135°C, making them accessible for standard laboratory and pilot plant equipment. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- React exo-norbornene-2,3-dicarboxylic anhydride with 4-bromoaniline at 120-135°C to form the bromo-imide intermediate.
- Perform Suzuki coupling with 4-hydroxyphenylboronic acid using Pd(PPh3)4 catalyst at 95-115°C to generate the biphenyl phenol derivative.
- Conduct esterification with acryloyl chloride and triethylamine at 15-35°C to introduce the terminal alkene functionality.
- Execute thiol-ene Michael addition with unprotected thiol-sugars (mannose, glucose, or galactose) using n-hexylamine catalyst at room temperature.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers compelling economic and operational benefits. The elimination of protection and deprotection steps translates directly into a reduction in raw material consumption and waste disposal costs. By shortening the synthetic timeline, manufacturers can achieve faster turnaround times for custom orders, enhancing responsiveness to market demands. Furthermore, the use of robust chemistry like Suzuki coupling and thiol-ene addition ensures consistent batch-to-batch quality, reducing the risk of production failures and supply disruptions. This reliability is crucial for maintaining continuous operations in the competitive fine chemicals sector.
- Cost Reduction in Manufacturing: The streamlined synthesis significantly lowers the cost of goods sold by removing expensive protecting group reagents and the associated labor for additional reaction steps. The high atom economy of the click chemistry step ensures that a larger proportion of input materials end up in the final product, minimizing waste. Additionally, the mild reaction conditions reduce energy consumption compared to processes requiring extreme temperatures or pressures, contributing to overall operational efficiency and sustainability goals.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as norbornene anhydride, bromoaniline, and common boronic acids mitigates the risk of raw material shortages. These starting materials are widely produced and have stable supply chains, unlike specialized protected sugar derivatives which may have limited suppliers. This diversification of sourcing options strengthens the supply chain resilience, ensuring that production schedules can be maintained even during periods of market volatility or logistical constraints.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of well-understood unit operations like filtration, extraction, and column chromatography. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, lowering compliance costs and improving the corporate sustainability profile. The ability to produce high-purity intermediates without heavy metal contamination simplifies downstream processing, making the technology suitable for large-scale commercial production of pharmaceutical and biomedical materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. They are intended to provide clarity on the feasibility and advantages of adopting this method for industrial applications.
Q: What is the primary advantage of this non-protecting group synthesis method?
A: The primary advantage is the elimination of tedious protection and deprotection steps typically required for sugar hydroxyl groups. This significantly reduces the total number of reaction steps, minimizes waste generation, and improves the overall atom economy compared to conventional glycopolymer monomer synthesis.
Q: Can this method be scaled for industrial production of glycopolymers?
A: Yes, the synthesis utilizes robust reactions such as Suzuki coupling and thiol-ene click chemistry, which are known for their scalability and mild conditions. The use of commercially available starting materials like norbornene anhydride and common boronic acids further supports feasible commercial scale-up.
Q: What types of sugars can be incorporated using this platform?
A: The platform is versatile and supports the incorporation of various monosaccharides including mannose, glucose, and galactose via their corresponding thiol-derivatives. This allows for the creation of diverse glycopolymer libraries to study specific protein-carbohydrate interactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Non-Protecting Single-Arm Sugar-Containing Compound Supplier
The technological potential of this non-protecting group synthesis route is immense, offering a clear path to next-generation glycomaterials. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our team is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of intermediate meets the highest industry standards. We understand the critical nature of supply continuity in the pharmaceutical sector and are committed to being a partner you can trust for long-term projects.
We invite you to explore how this innovative chemistry can optimize your production costs and accelerate your R&D timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us help you engineer the future of bio-materials with precision and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
