Industrial Scale-Up of 2,3,4-Trifluorobenzoic Acid via Safe Grignard Carboxylation
The pharmaceutical and agrochemical industries continuously seek robust synthetic pathways for fluorinated aromatic building blocks, driven by the unique metabolic stability and lipophilicity these motifs impart to bioactive molecules. Patent CN1155555C presents a pivotal advancement in the manufacturing of 2,3,4-trifluorobenzoic acid, a critical intermediate for liquid crystal materials and advanced drug substances. Unlike legacy methods that rely on hazardous elemental fluorine or pyrophoric organolithium reagents, this intellectual property outlines two distinct, industrially viable routes originating from the readily available 2,3,4-trifluoroaniline. The first pathway leverages a Sandmeyer-type nitrilation followed by acidic hydrolysis, while the second, more mechanistically intriguing route, employs a diazotization-bromination sequence to generate an aryl bromide, which is subsequently converted into the target carboxylic acid via Grignard carboxylation. This dual-pathway approach provides manufacturers with strategic flexibility, allowing process chemists to select the optimal route based on local reagent availability and waste treatment capabilities, thereby securing the supply chain against single-point failures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyfluorinated benzoic acids has been plagued by significant safety and operational hurdles that hinder large-scale commercialization. One prominent conventional method involves the direct fluorination of 4-fluorobenzoic acid using elemental fluorine gas, a process that demands highly specialized, corrosion-resistant reactor vessels and rigorous safety protocols due to the extreme toxicity and reactivity of fluorine. Furthermore, this electrophilic fluorination often suffers from poor regioselectivity, generating a complex mixture of isomeric byproducts that drastically reduce the overall yield and necessitate expensive purification steps. Another traditional approach utilizes 1,2,3-trifluorobenzene as a starting material, requiring lithiation with n-butyllithium at cryogenic temperatures. This method imposes severe constraints on manufacturing infrastructure, as it mandates strictly anhydrous and anaerobic conditions to prevent catastrophic quenching of the reactive lithio-species. The inherent instability of organolithium intermediates and the high energy costs associated with maintaining sub-zero reaction environments make these conventional routes economically unattractive for multi-ton production campaigns.
The Novel Approach
In stark contrast to these high-risk methodologies, the novel approach detailed in the patent utilizes 2,3,4-trifluoroaniline as a stable, commodity-grade starting material, fundamentally shifting the risk profile of the synthesis. By employing a diazotization strategy, the process converts the amino group into a versatile leaving group that can be displaced by either a cyano or a bromo functionality under relatively mild thermal conditions. This transformation avoids the need for cryogenic cooling or exotic fluorinating agents, relying instead on standard mineral acids and inorganic salts that are ubiquitous in fine chemical plants. The subsequent conversion of the aryl bromide to the carboxylic acid via a Grignard intermediate represents a classic yet highly effective carbon-carbon bond-forming reaction that scales predictably. This route not only simplifies the operational complexity by eliminating the need for specialized fluorination equipment but also enhances the overall atom economy by utilizing carbon dioxide as the one-carbon source, aligning the synthesis with modern principles of green chemistry and sustainable manufacturing practices.
Mechanistic Insights into Grignard Carboxylation and Diazotization
The core of the second synthetic route lies in the precise control of the diazotization and subsequent Grignard formation, a sequence that requires meticulous attention to reaction kinetics and thermodynamics. The process initiates with the treatment of 2,3,4-trifluoroaniline with sodium nitrite in an acidic medium, typically sulfuric or hydrochloric acid, at temperatures ranging from -5°C to 10°C to generate the unstable diazonium salt. This electrophilic species is then immediately subjected to a Sandmeyer reaction with cuprous bromide, where the copper catalyst facilitates the radical substitution of the diazo group with a bromine atom, yielding 1-bromo-2,3,4-trifluorobenzene. The success of this step is critical, as residual diazonium salts can lead to phenolic impurities upon hydrolysis, compromising the purity of the downstream Grignard reagent. Following isolation, the aryl bromide is reacted with activated magnesium metal in a mixed solvent system of tetrahydrofuran (THF) and toluene. The THF serves to solvate the magnesium cation and stabilize the Grignard species, while toluene improves the solubility of the organic substrate and moderates the exothermicity of the initiation phase.
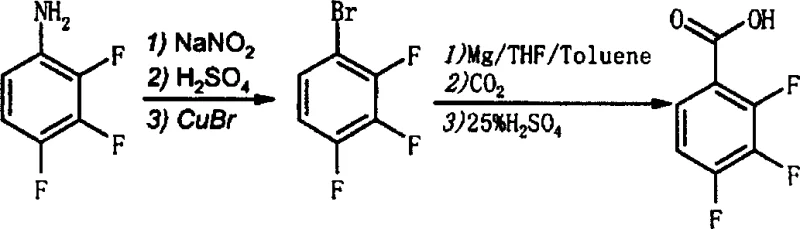
Once the Grignard reagent is formed, the introduction of dry, oxygen-free carbon dioxide acts as the electrophile, attacking the nucleophilic carbon-magnesium bond to form a carboxylate salt. This step must be conducted under strictly anhydrous conditions to prevent protonation of the Grignard reagent, which would revert the intermediate back to the starting aryl bromide and consume valuable magnesium. The final acidification step, typically performed with dilute sulfuric acid, protonates the carboxylate to release the free 2,3,4-trifluorobenzoic acid. The patent highlights the importance of the solvent ratio, recommending a toluene to THF volume ratio between 1:0.1 and 1:10 to optimize both the formation rate and the stability of the organometallic intermediate. This mechanistic understanding allows process engineers to fine-tune the addition rates and temperature profiles, ensuring that the highly exothermic Grignard formation does not lead to thermal runaway, thereby guaranteeing a safe and reproducible manufacturing process suitable for kilolab and plant-scale operations.
How to Synthesize 2,3,4-Trifluorobenzoic Acid Efficiently
Executing this synthesis at an industrial level requires a disciplined approach to unit operations, beginning with the careful preparation of the diazonium salt solution under controlled cooling to manage nitrogen evolution. The subsequent bromination step benefits from a biphasic workup to remove copper residues, which are potent catalysts for decomposition and must be reduced to trace levels before the Grignard step. The final purification often involves an acid-base extraction sequence followed by recrystallization from an alcohol-water system, which effectively removes non-acidic organic impurities and inorganic salts. Adhering to these standardized protocols ensures that the final product meets the stringent purity specifications required for pharmaceutical applications, where even trace metal contaminants can catalyze degradation in the final drug product.
- Diazotize 2,3,4-trifluoroaniline using sodium nitrite and acid at low temperature (-5 to 10°C) to form the diazonium salt.
- React the diazonium salt with cuprous bromide to generate 1-bromo-2,3,4-trifluorobenzene via Sandmeyer reaction.
- Form the Grignard reagent using magnesium in THF/Toluene, react with dry CO2, and hydrolyze with sulfuric acid to isolate the target acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patent-protected methodology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio; by replacing hazardous elemental fluorine and pyrophoric butyllithium with stable anilines and magnesium turnings, the facility eliminates the need for specialized storage tanks and emergency containment systems. This reduction in hazard classification directly translates to lower insurance premiums and reduced regulatory compliance burdens, allowing for more flexible site selection and faster permitting processes. Furthermore, the reliance on commodity chemicals such as carbon dioxide and sulfuric acid insulates the production cost from the volatility associated with specialty fluorinating agents, providing a more predictable cost structure for long-term supply agreements. The robustness of the Grignard carboxylation also implies a higher tolerance for minor variations in feedstock quality, reducing the rejection rate of incoming materials and minimizing production downtime caused by out-of-spec batches.
- Cost Reduction in Manufacturing: The elimination of cryogenic infrastructure required for organolithium chemistry results in significant capital expenditure savings, as standard glass-lined or stainless steel reactors can be utilized without the need for extreme low-temperature cooling jackets. Additionally, the use of carbon dioxide as a C1 building block is inherently cheaper than cyanide sources or other carboxylation reagents, driving down the variable cost per kilogram of the active pharmaceutical ingredient. The process also avoids the generation of heavy metal waste streams associated with certain palladium-catalyzed carbonylations, simplifying wastewater treatment and reducing disposal costs.
- Enhanced Supply Chain Reliability: Sourcing 2,3,4-trifluoroaniline is significantly more reliable than procuring custom-synthesized fluorinated precursors, as it is a bulk chemical produced by multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks that could disrupt production schedules. Moreover, the intermediate 1-bromo-2,3,4-trifluorobenzene is a stable liquid that can be stockpiled if necessary, providing a buffer against upstream supply fluctuations and enabling just-in-time manufacturing strategies for the final acid.
- Scalability and Environmental Compliance: The reaction conditions described, particularly the moderate temperatures for diazotization and Grignard formation, are easily transferable from pilot plant to full commercial scale without significant re-engineering. The process generates primarily inorganic salts and organic solvents that can be recovered and recycled, aligning with increasingly strict environmental regulations regarding volatile organic compound emissions. The absence of persistent organic pollutants or highly toxic byproducts simplifies the environmental impact assessment, facilitating smoother audits and certifications from international regulatory bodies.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation of this synthesis route in a GMP environment. These responses are derived directly from the experimental data and process parameters outlined in the patent literature, ensuring that the guidance provided is grounded in verified chemical principles rather than theoretical speculation.
Q: Why is the Grignard route preferred over direct fluorination for this intermediate?
A: Direct fluorination often requires hazardous elemental fluorine gas and specialized equipment, whereas the Grignard route utilizes standard industrial reagents like magnesium and carbon dioxide, significantly lowering safety barriers and capital expenditure.
Q: What are the critical purity specifications for the starting aniline?
A: High-purity 2,3,4-trifluoroaniline is essential to minimize side reactions during diazotization. Impurities can lead to complex byproduct profiles that are difficult to separate during the final recrystallization steps.
Q: How does the solvent system impact the Grignard reaction efficiency?
A: The patent recommends a mixed solvent system of Toluene and THF. This balance optimizes the solubility of the organic halide while maintaining the reactivity of the magnesium surface, preventing passivation and ensuring consistent conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,4-Trifluorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory bench to commercial production requires more than just a patent license; it demands deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of the Grignard carboxylation is maintained regardless of batch size. We operate state-of-the-art rigorous QC labs equipped with advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every lot of 2,3,4-trifluorobenzoic acid meets the exacting standards required for next-generation pharmaceutical and electronic material applications. Our facility is designed to handle fluorinated intermediates safely, with dedicated containment systems and waste treatment protocols that exceed industry norms.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthetic route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain visibility into the potential economic benefits of switching to this safer, more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring that your development pipeline remains uninterrupted by supply constraints.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
