Advanced Catalytic Reduction Technology for High-Purity ACE Inhibitor Intermediates and Commercial Scalability
Introduction to Patent CN1218454A: A Breakthrough in ACE Inhibitor Synthesis
The pharmaceutical industry continuously seeks robust manufacturing routes for critical cardiovascular medications, particularly Angiotensin-Converting Enzyme (ACE) inhibitors like Enalapril and Lisinopril. Patent CN1218454A discloses a highly efficient and productive process for preparing 1-alkoxycarbonyl-3-phenylpropyl derivatives, which serve as pivotal intermediates in these drug syntheses. The core innovation lies in a catalytic reduction strategy that operates under specific strong acidic conditions, fundamentally altering the reaction landscape to minimize impurity formation. Unlike traditional methods that struggle with side reactions, this technology leverages a precise balance of strong acid concentration and catalytic hydrogenation to achieve superior product quality. The general structure of the starting material, 1-alkoxycarbonyl-3-oxo-3-phenylpropyl derivatives, is represented by Formula I, where specific amino acid residues define the final therapeutic application.
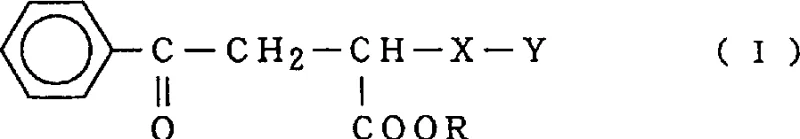
This patent addresses the critical need for high-purity intermediates by solving long-standing issues related to by-product generation during the reduction of the ketone group. By strictly controlling the acidity and reaction parameters, manufacturers can significantly reduce the burden on downstream purification units. The method is particularly effective for producing the 1S stereoisomer, which is essential for biological activity in many ACE inhibitors. For R&D directors and process chemists, this represents a viable pathway to enhance overall process mass intensity (PMI) and reduce the environmental footprint associated with solvent-intensive purification steps. The following analysis details the mechanistic advantages and commercial implications of adopting this strong-acid mediated catalytic reduction technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the catalytic reduction of 1-alkoxycarbonyl-3-oxo-3-phenylpropyl derivatives has been plagued by significant selectivity challenges. Conventional processes typically employ transition metal catalysts such as palladium or nickel in alcoholic solvents with low concentrations of acid or under neutral conditions. A major drawback of these legacy methods is the tendency for the phenyl ring to undergo unwanted hydrogenation, leading to the formation of 1-alkoxycarbonyl-3-cyclohexyl propyl derivatives. This cyclohexyl by-product, represented by Formula III, possesses a structure remarkably similar to the target molecule, making its removal via standard crystallization or chromatography extremely difficult and costly.
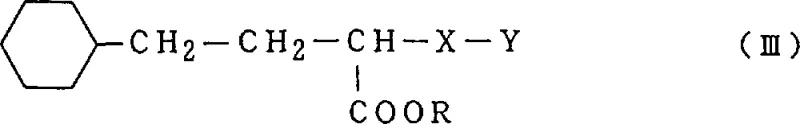
Furthermore, conventional methods often suffer from the hydrolysis of the ester group, generating 1-carboxyl-3-phenylpropyl derivatives (Formula IV). This carboxyl impurity not only reduces the overall yield of the desired ester but also complicates the isolation process. In many prior art examples, such as those cited in Japanese Patent publications, the reaction times are excessively long (up to 35 hours), and the separation procedures involve multiple extraction steps using large volumes of hazardous organic solvents like dichloromethane. These inefficiencies result in lower throughput, higher operational costs, and increased waste generation, posing significant challenges for supply chain managers aiming for sustainable and cost-effective production.
The Novel Approach
The technology disclosed in CN1218454A introduces a paradigm shift by utilizing a high concentration of strong acid, specifically ranging from 0.4N to 5N, during the catalytic reduction step. This aggressive acidic environment serves a dual purpose: it stabilizes the reaction intermediate and effectively suppresses the hydrogenation of the aromatic ring. By maintaining a molar ratio of at least 3 equivalents of strong acid relative to the substrate, the process drastically minimizes the formation of the troublesome cyclohexyl by-product (Formula III). Additionally, the presence of water in the alcoholic solvent system, combined with the specific acid concentration, helps prevent the hydrolysis of the ester group, thereby reducing the generation of the carboxyl impurity (Formula IV).
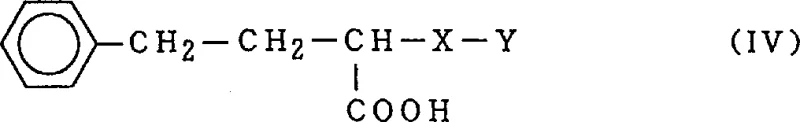
This novel approach enables the reaction to proceed rapidly, often completing within 10 hours or less, compared to the multi-day timelines of older methods. The result is a reaction mixture with a significantly cleaner profile, allowing for simpler downstream processing. Instead of relying on complex solvent extractions, the product can often be isolated directly through crystallization from the aqueous reaction mixture after neutralization. This simplification of the workflow not only enhances safety by reducing solvent handling but also improves the overall economic viability of the manufacturing process, making it an attractive option for large-scale commercial production of ACE inhibitor intermediates.
Mechanistic Insights into Strong Acid-Mediated Catalytic Reduction
The success of this synthesis route hinges on the intricate interplay between the strong acid medium and the palladium catalyst surface. Mechanistically, the reduction of the ketone group in Formula I to the methylene group in Formula II is believed to proceed via a stepwise hydriding process. The first step involves the hydrogenation of the carbonyl group to form a hydroxyl intermediate (alcohol). In conventional weakly acidic or neutral media, this hydroxyl intermediate is prone to cyclization, forming a lactone species. This lactone can subsequently undergo hydrogenation to yield the undesired 1-carboxyl-3-phenylpropyl derivative (Formula IV), effectively destroying the ester functionality required for the final drug molecule.
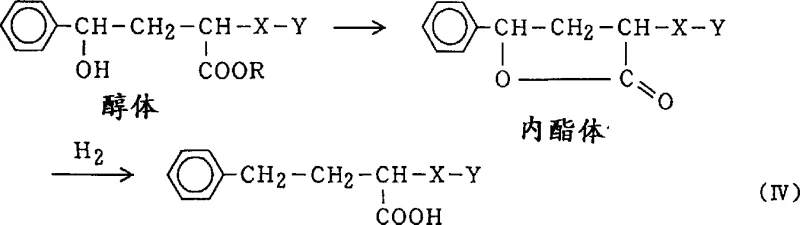
However, under the high acidity conditions prescribed in this patent (e.g., using sulfuric acid), the equilibrium is shifted to prevent lactone formation. The high proton concentration likely protonates the hydroxyl group or the carbonyl oxygen, inhibiting the nucleophilic attack necessary for cyclization. Furthermore, the strong acid environment appears to modify the electronic properties of the catalyst or the substrate in a way that disfavors the adsorption and subsequent reduction of the stable aromatic ring. This selectivity is crucial because once the phenyl ring is reduced to a cyclohexyl ring, the resulting impurity is chemically very similar to the product and difficult to separate. By stopping the reaction when hydrogen absorption reaches approximately 90% of the theoretical value, the process ensures that the reduction stops at the desired stage without over-reducing the aromatic system.
From an impurity control perspective, the mechanism also facilitates a unique purification strategy. The target 1-alkoxycarbonyl-3-phenylpropyl derivative has a specific isoelectric point, typically around pH 4.6. By neutralizing the strongly acidic reaction mixture to this specific pH range, the solubility of the target compound in the aqueous phase is minimized, causing it to precipitate. Conversely, the carboxyl impurity (Formula IV), being more acidic and polar, remains soluble in the aqueous salt solution generated during neutralization. This differential solubility allows for the effective removal of the carboxyl impurity simply by filtration, bypassing the need for organic extraction. This mechanistic understanding provides R&D teams with the confidence to scale the process, knowing that the chemistry inherently drives towards high purity through controlled crystallization rather than reliance on arbitrary separation techniques.
How to Synthesize 1-Alkoxycarbonyl-3-Phenylpropyl Derivatives Efficiently
The synthesis of these critical intermediates requires precise control over reaction parameters to maximize yield and stereochemical purity. The patented method outlines a streamlined procedure that begins with the dissolution of the keto-ester precursor in an alcoholic solvent containing a defined concentration of strong acid. The choice of acid, typically sulfuric acid, and its equivalence ratio are critical variables that must be optimized to suppress side reactions while maintaining catalyst activity. The reaction is then subjected to catalytic hydrogenation using a palladium-based catalyst, such as Pd-C, under mild pressure conditions. Monitoring the hydrogen uptake is essential to prevent over-reduction, with the reaction ideally terminated once the theoretical hydrogen consumption approaches 90%. Following the reduction, the workup involves a carefully controlled neutralization step to induce crystallization, leveraging the solubility differences between the product and impurities to achieve high purity without extensive chromatography.
- Dissolve 1-alkoxycarbonyl-3-oxo-3-phenylpropyl derivatives in an alcoholic solvent containing 0.4 to 5N strong acid, ensuring a molar ratio of at least 3 equivalents of acid.
- Perform catalytic hydrogenation using a palladium catalyst at 10-35°C and low pressure until hydrogen absorption reaches approximately 90% of theoretical value.
- Neutralize the reaction mixture to pH 4.6 ± 1.5 and induce crystallization in an aqueous environment to separate the product from carboxyl and cyclohexyl impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits in terms of cost stability and supply reliability. The primary advantage lies in the substantial improvement in process yield and purity, which directly translates to better raw material utilization. By minimizing the formation of hard-to-remove impurities like the cyclohexyl derivative, the process reduces the loss of valuable intermediates during purification. This efficiency gain means that less starting material is required to produce the same amount of final API intermediate, effectively lowering the cost of goods sold (COGS). Furthermore, the ability to operate at low hydrogen pressures eliminates the need for expensive, specialized high-pressure reactors, reducing capital expenditure requirements for manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of complex organic extraction steps and the reduction in solvent usage significantly lower operational costs. Traditional methods often require large volumes of solvents like dichloromethane or ethyl acetate for extraction, followed by energy-intensive distillation for recovery. In contrast, the aqueous crystallization method described in the patent relies primarily on water and simple alcohols, which are cheaper and easier to handle. The reduction in solvent waste also lowers disposal costs and environmental compliance burdens. Additionally, the higher reaction yield means fewer batches are needed to meet production targets, optimizing labor and utility consumption across the manufacturing site.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes to a more reliable supply chain. Conventional methods are sensitive to minor variations in reaction conditions, which can lead to batch failures or off-spec material that requires reprocessing. The strong acid-mediated method provides a wider operating window and better control over impurity profiles, ensuring consistent quality batch after batch. This consistency reduces the risk of supply disruptions caused by quality issues. Moreover, the use of readily available catalysts like Pd-C and common acids like sulfuric acid ensures that the supply of critical reagents is stable and not subject to the volatility associated with exotic or specialized chemicals.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new challenges regarding heat transfer and mixing, but this technology is designed with scalability in mind. The exothermic nature of the hydrogenation is manageable under the described conditions, and the use of aqueous systems simplifies temperature control. From an environmental standpoint, the reduction in organic solvent usage aligns with green chemistry principles and increasingly stringent regulatory requirements. Minimizing the release of volatile organic compounds (VOCs) and hazardous waste makes the manufacturing process more sustainable. This environmental advantage is increasingly important for pharmaceutical companies aiming to meet corporate sustainability goals and maintain a positive public image.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic reduction technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing workflows. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: How does the strong acid condition prevent the formation of cyclohexyl by-products?
A: The presence of high concentration strong acid (e.g., sulfuric acid) stabilizes the reaction intermediate and suppresses the over-reduction of the phenyl ring, which is a common side reaction in conventional neutral or weakly acidic hydrogenation processes.
Q: What is the primary advantage of the aqueous crystallization separation method?
A: Adjusting the pH to the isoelectric point (around pH 4.6) allows the target 1-alkoxycarbonyl derivative to crystallize while keeping the more soluble 1-carboxyl-3-phenylpropyl impurity in the aqueous phase, eliminating the need for complex organic extractions.
Q: Can this process be scaled for industrial production of Enalapril intermediates?
A: Yes, the process utilizes standard palladium catalysts and operates at low hydrogen pressures (normal to 2 kg/cm²), making it highly suitable for large-scale commercial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Alkoxycarbonyl-3-Phenylpropyl Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of cardiovascular medications. Our team of expert chemists has extensively evaluated the technology described in Patent CN1218454A and possesses the capability to implement this advanced catalytic reduction process on a commercial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of 1-alkoxycarbonyl-3-phenylpropyl derivatives. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, including the precise control of stereoisomers and trace impurities such as cyclohexyl and carboxyl derivatives.
We invite pharmaceutical partners to collaborate with us to optimize their supply chains and reduce manufacturing costs. By leveraging our expertise in strong acid-mediated catalysis and aqueous crystallization, we can help you achieve significant efficiencies in your API production. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
