Advanced Vacuum-Driven Synthesis of Vinyl Amide Compounds for Commercial Agrochemical Production
The chemical industry constantly seeks methods to optimize the synthesis of key intermediates, balancing yield, purity, and operational simplicity. Patent CN1073551C presents a significant breakthrough in the production of vinyl amide compounds, which serve as vital precursors in the manufacture of herbicides and other agrochemical agents. This technology addresses long-standing challenges in the acylation of Schiff bases by shifting the reaction paradigm from atmospheric pressure to a controlled reduced-pressure environment. By integrating the removal of hydrogen halide by-products directly into the reaction phase through distillation, this process achieves exceptional purity levels ranging from 90% to 99.9% without the need for cumbersome post-reaction purification steps. For R&D directors and procurement specialists alike, this represents a move towards more streamlined, cost-effective, and scalable manufacturing protocols that align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vinyl amide compounds via the reaction of Schiff bases with acid halides has been plagued by inefficiencies inherent to atmospheric pressure conditions. Traditional methods, such as those disclosed in earlier patents like US 4,600,433, typically operate at normal pressure in solvents like toluene. A major drawback of these conventional approaches is the accumulation of hydrogen halide by-products within the reaction mixture. This acidic buildup not only consumes the starting Schiff base materials through unwanted side reactions but also necessitates the use of hydrogen halide scavengers. Consequently, the crude product requires extensive downstream processing, including filtration to remove scavenger salts and rigorous purification via recrystallization or high-vacuum distillation to meet purity standards. These additional steps increase manpower requirements, extend production cycles, and introduce significant thermal risks, as the final vinyl amide products are prone to thermal decomposition if distilled under the extreme vacuums often required for purification.
The Novel Approach
The methodology described in patent CN1073551C fundamentally alters this landscape by conducting the reaction under reduced pressure, preferably between 1.3×10^2 Pa and 1×10^5 Pa. This innovative approach leverages the volatility of the hydrogen halide by-product, allowing it to be continuously distilled out of the reaction system as it forms. By physically removing the acid from the equilibrium, the process prevents the degradation of the sensitive Schiff base starting materials and suppresses the formation of addition by-products. Furthermore, the optional introduction of an inert gas, such as nitrogen, enhances the stripping effect, ensuring that the reaction proceeds rapidly and cleanly. The result is a crude product of such high purity that, after simple solvent removal, it can be utilized directly in subsequent synthesis steps. This elimination of scavengers and complex purification stages translates directly into substantial operational savings and a more robust supply chain for high-purity agrochemical intermediates.
Mechanistic Insights into Vacuum-Driven Acylation
The core of this technological advancement lies in the manipulation of reaction thermodynamics and kinetics through pressure control. In the standard acylation of a Schiff base, the generation of hydrogen halide (HX) creates an acidic environment that can protonate the imino nitrogen or attack the adjacent carbon atoms, leading to the formation of stable adducts that reduce the overall yield of the desired vinyl amide. By applying reduced pressure, the boiling point of the reaction medium is lowered, and the partial pressure of the volatile HX is kept minimal. This continuous removal shifts the chemical equilibrium towards the product side according to Le Chatelier's principle, driving the reaction to completion more effectively than static atmospheric conditions. Additionally, the lower operating temperatures achievable under vacuum (often refluxing at 40-100°C depending on the solvent) mitigate the risk of thermal degradation of the sensitive vinyl double bond, which is crucial for maintaining the integrity of the final herbicide precursor.
Structural versatility is another key aspect of this mechanism, accommodating a wide range of substituents on both the Schiff base and the acid halide. The Schiff base component, generally represented by a structure containing an imino group adjacent to a carbon with at least one hydrogen atom, can vary significantly in its aromatic or heteroaromatic substitutions.
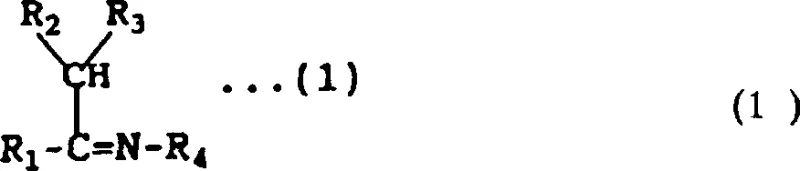
Similarly, the acid halide component allows for various acyl groups, enabling the tuning of the final molecule's properties. The resulting vinyl amide product retains the critical amide bond and the alpha-beta unsaturation necessary for biological activity in agrochemical applications.

This mechanistic robustness ensures that impurity profiles remain consistent and manageable, a critical factor for regulatory compliance in the pharmaceutical and agrochemical sectors. The ability to produce such complex molecules with purity exceeding 99% in a single pot reaction underscores the sophistication of this vacuum-driven protocol.
How to Synthesize Vinyl Amide Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the vacuum distillation technique. The process begins with the dissolution of the specific Schiff base compound in a suitable inert organic solvent, such as toluene, xylene, or benzene, within a reactor capable of maintaining reduced pressure. The acid halide is then introduced, either dropwise or in a single charge, while the system is heated to reflux under the specified vacuum conditions. It is imperative to monitor the pressure and temperature to ensure they remain within the optimal window of 1.3×10^3 to 6.7×10^4 Pa to facilitate the efficient removal of hydrogen halide without excessive solvent loss. For enhanced performance, a stream of inert gas like nitrogen can be bubbled through the mixture or introduced into the headspace to assist in stripping the acid by-product. Once the reaction is complete, indicated by the cessation of HX evolution, the solvent is removed under reduced pressure to yield the final high-purity vinyl amide. The detailed standardized synthesis steps for specific derivatives are outlined below.
- Dissolve the Schiff base compound in an inert organic solvent such as toluene or xylene within a reactor equipped for reduced pressure operation.
- Add the acid halide gradually while maintaining the reaction system under reduced pressure (preferably 1.3×10^3 to 6.7×10^4 Pa) and heat to reflux.
- Continue heating until the reaction is complete, allowing the hydrogen halide by-product to distill off continuously, then remove the solvent to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this vacuum-driven synthesis method offers compelling economic and logistical benefits that extend beyond simple yield improvements. The most significant advantage is the drastic simplification of the downstream processing workflow. By eliminating the need for hydrogen halide scavengers, manufacturers avoid the costs associated with purchasing these reagents and the subsequent waste disposal fees for the salt by-products they generate. Furthermore, the removal of recrystallization and complex distillation steps reduces the energy consumption per kilogram of product and frees up valuable reactor time, effectively increasing the throughput capacity of existing manufacturing facilities. This streamlined process also minimizes the handling of hazardous materials and reduces the potential for human error during multiple transfer and purification stages, leading to a safer and more reliable production environment.
- Cost Reduction in Manufacturing: The elimination of expensive purification reagents and the reduction in energy-intensive distillation steps lead to significant cost savings. By avoiding the need for industrial-scale recrystallization, which is labor and time-intensive, the overall production cost per unit is drastically lowered. Additionally, the high yield achieved through this method means less raw material is wasted, optimizing the utilization of costly Schiff base precursors and acid halides. The ability to use the crude product directly after solvent removal further reduces the capital expenditure required for specialized purification equipment.
- Enhanced Supply Chain Reliability: A simpler process with fewer unit operations inherently reduces the risk of batch failures and delays. The robustness of the vacuum reaction against variations in starting material quality ensures consistent output, which is critical for maintaining steady supply lines to downstream herbicide manufacturers. The reduced processing time per batch allows for greater flexibility in scheduling and faster response to market demand fluctuations. Moreover, the use of common solvents and standard vacuum equipment means that the process can be easily replicated across different manufacturing sites, diversifying supply sources and mitigating geopolitical or logistical risks.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward because it relies on well-understood unit operations like vacuum distillation and reflux. The continuous removal of hydrogen halide minimizes corrosion issues in large-scale reactors compared to holding high concentrations of acid. From an environmental perspective, the reduction in solvent usage and the elimination of solid waste from scavengers contribute to a smaller environmental footprint. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the supply chain more resilient to future environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its implementation. The following questions address common inquiries regarding the operational parameters and quality outcomes of the vacuum-driven process. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Whether you are concerned about the specific pressure ranges required or the compatibility of different substituents, the information below provides a clear overview of the method's capabilities and limitations in an industrial setting.
Q: Why is reduced pressure critical for this vinyl amide synthesis?
A: Reduced pressure facilitates the continuous removal of the hydrogen halide by-product during the reaction. This prevents the accumulation of acidic species that would otherwise react with the Schiff base starting material to form unwanted adducts, thereby significantly improving both yield and purity without needing post-reaction scavengers.
Q: Does this process require complex purification steps like recrystallization?
A: No, one of the primary advantages of this method is that the residue obtained after removing the reaction solvent can be used directly as the target product. The in-situ removal of impurities during the reaction eliminates the need for labor-intensive industrial recrystallization or high-vacuum distillation of the final product.
Q: Can inert gases be used to further enhance the reaction efficiency?
A: Yes, introducing an inert gas such as nitrogen into the reaction system while under reduced pressure can further accelerate the removal of hydrogen halide. This dual approach of vacuum and gas sparging has been shown to improve reaction rates and achieve even higher product purity levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinyl Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation agrochemicals. Our team of expert chemists has extensively evaluated the vacuum-driven synthesis route described in patent CN1073551C and possesses the technical capability to implement this advanced methodology at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with sophisticated vacuum reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of vinyl amide intermediate we deliver meets the highest standards of consistency and performance required by global herbicide manufacturers.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with us, you gain access to our deep process knowledge and the ability to request specific COA data and route feasibility assessments for your target molecules. Let us help you secure a reliable source of high-purity vinyl amide compounds that will drive the success of your agricultural chemical products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
