Scalable Synthesis of Deuterium-Labeled Betamethasone for Pharmacokinetic Research
Scalable Synthesis of Deuterium-Labeled Betamethasone for Pharmacokinetic Research
The pharmaceutical industry's relentless pursuit of precision medicine has elevated the status of stable isotope-labeled compounds from mere analytical tools to critical components in drug development pipelines. Patent CN108440628B discloses a robust and highly efficient preparation method for deuterium-labeled betamethasone, specifically targeting the substitution of five hydrogen atoms at the 4, 6, 11, and 12 positions with deuterium. This technological breakthrough addresses the longstanding challenges associated with synthesizing complex steroid isotopologues, offering a pathway to materials with over 98% chemical purity and greater than 97% isotopic abundance. For R&D directors and procurement specialists alike, this method represents a significant leap forward in securing reliable supplies of high-quality reference standards necessary for FDA-compliant metabolic safety evaluations.
Deuterium labeling is not merely an academic exercise; it is a strategic necessity for understanding the metabolic fate of potent glucocorticoids like betamethasone. The carbon-deuterium bond possesses superior stability compared to the carbon-hydrogen bond, which can directly influence the metabolic half-life of the drug, potentially reducing dosing frequency and improving patient compliance. The disclosed synthesis leverages a semi-synthetic approach starting from commercially available betamethasone, thereby bypassing the prohibitive costs and low yields often associated with de novo total synthesis of labeled steroids. This document analyzes the technical merits of this five-step sequence and its implications for cost reduction in API manufacturing and supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of site-specifically deuterated steroids has been plagued by inefficient synthetic routes that rely on expensive, specialized starting materials or harsh reaction conditions that compromise stereochemical integrity. Conventional methods often involve multiple protection and deprotection cycles that result in substantial material loss, driving up the cost per gram to levels that are unsustainable for large-scale pharmacokinetic studies. Furthermore, achieving high isotopic enrichment at specific positions without scrambling the label to unwanted sites requires exquisite control over reaction parameters, which is difficult to maintain in a manufacturing environment. The reliance on transition metal catalysts in some older methodologies also introduces the risk of heavy metal contamination, necessitating additional purification steps that further erode yield and increase production timelines.
The Novel Approach
The methodology outlined in patent CN108440628B circumvents these bottlenecks by employing a logical sequence of protection, oxidation, exchange, reduction, and deprotection. By starting with the native hormone betamethasone, the process capitalizes on the existing chiral centers, eliminating the need for asymmetric synthesis. The strategic use of a cyclic acetal to protect the sensitive 17,21-dihydroxy side chain allows for selective manipulation of the A and C rings without degradation. This approach not only simplifies the operational complexity but also ensures that the final product retains the critical biological configuration required for receptor binding. The result is a streamlined process that delivers high-purity deuterium-labeled betamethasone with exceptional isotopic fidelity, suitable for both research and potential therapeutic applications.
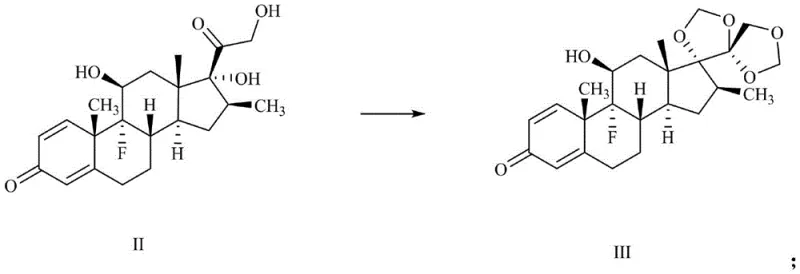
Mechanistic Insights into Selective Hydrogen-Deuterium Exchange
The core innovation of this synthesis lies in the precise installation of deuterium atoms via a base-catalyzed exchange mechanism. Following the initial protection of the 17,21-dihydroxy groups with paraformaldehyde in an acidic medium to yield compound III, the 11-hydroxyl group is oxidized to a ketone using Dess-Martin periodinane. This oxidation is crucial as it activates the adjacent C12 position for enolization. When treated with a base such as sodium methoxide in a deuterated solvent system (methanol-D1 or deuterium oxide), the alpha-protons at positions 4, 6, and 12 become labile and are exchanged for deuterium. The presence of the C3 ketone in the A-ring also facilitates exchange at positions 4 and 6. This tandem exchange capability allows for the simultaneous labeling of multiple sites in a single operational step, drastically improving atom economy.
Subsequent reduction of the 11-ketone with sodium borodeuteride reintroduces the hydroxyl functionality while potentially incorporating deuterium at the C11 position or ensuring the retention of the label at C12 during the stereochemical restoration. The final deprotection step using acid removes the cyclic acetal, revealing the native 17,21-dihydroxy motif without disturbing the newly installed deuterium labels. This mechanistic elegance ensures that the isotopic label is robust and stable under physiological conditions, making the resulting compound an ideal tracer for metabolic studies. The high yields observed in each step, ranging from 84% to 99% in the provided examples, underscore the robustness of this chemical logic.
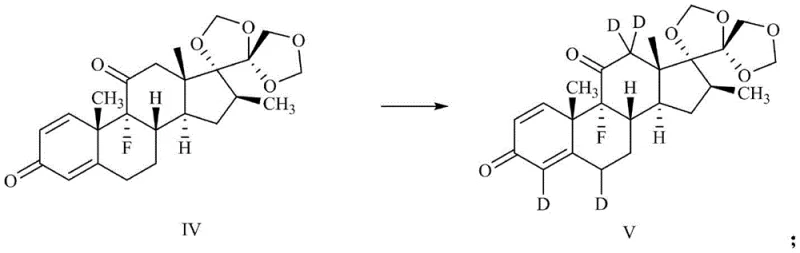
How to Synthesize Deuterium-Labeled Betamethasone Efficiently
The synthesis of this high-value intermediate requires strict adherence to the optimized reaction conditions detailed in the patent to ensure maximum isotopic incorporation and chemical purity. The process involves five distinct chemical transformations that must be carefully monitored to prevent isotopic dilution or side reactions. Operators should pay particular attention to the stoichiometry of the deuterating agents and the temperature control during the exchange and reduction phases. The following guide outlines the critical operational phases derived from the patent data, serving as a framework for process development teams aiming to replicate this high-efficiency route.
- Protect the 17,21-dihydroxy groups of betamethasone using paraformaldehyde and acid to form a cyclic acetal intermediate.
- Oxidize the 11-hydroxyl group to a ketone using Dess-Martin periodinane, followed by base-catalyzed H/D exchange in deuterated solvents.
- Reduce the 11-ketone back to a hydroxyl group using sodium borodeuteride and remove the acetal protection to yield the final D5 product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits beyond mere technical feasibility. The reliance on betamethasone as a bulk starting material leverages an established and abundant supply chain, mitigating the risks associated with sourcing exotic precursors. The elimination of transition metal catalysts in favor of organic oxidants and simple inorganic bases simplifies the waste stream and reduces the regulatory burden associated with heavy metal clearance. This translates directly into a more predictable manufacturing timeline and lower overall production costs, enabling more competitive pricing for downstream users.
- Cost Reduction in Manufacturing: The process utilizes commodity chemicals such as paraformaldehyde, sodium methoxide, and hydrofluoric acid, which are significantly cheaper than specialized organometallic reagents. By avoiding the need for custom-synthesized chiral building blocks, the raw material costs are drastically minimized. Furthermore, the high yields reported in the patent examples suggest that material throughput is maximized, reducing the cost of goods sold per gram of final product. This economic efficiency makes large-scale production of deuterated standards financially viable for the first time.
- Enhanced Supply Chain Reliability: Sourcing deuterium-labeled compounds has historically been a bottleneck due to limited global suppliers. This method democratizes production by using standard laboratory equipment and widely available reagents. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in raw material quality, ensuring consistent output. For supply chain planners, this reliability means reduced lead times and the ability to secure long-term contracts without the fear of supply disruption common with niche isotope producers.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing solvents like dichloromethane and ethanol that are manageable in large-scale reactors with appropriate recovery systems. The absence of toxic heavy metals simplifies the environmental, health, and safety (EHS) profile of the manufacturing process. Waste treatment is more straightforward, and the overall green chemistry metrics are improved compared to traditional multi-step total syntheses. This alignment with modern environmental standards facilitates easier regulatory approval for manufacturing sites.
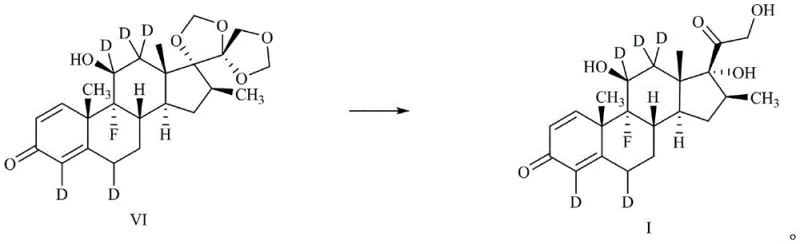
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of deuterium-labeled betamethasone. These answers are derived directly from the experimental data and technical specifications provided in the source patent, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for integrating this material into your research or production workflows effectively.
Q: What is the isotopic purity of the deuterium-labeled betamethasone produced by this method?
A: The patented process achieves an isotope abundance of greater than 97% and a chemical purity exceeding 98%, making it suitable for rigorous metabolic tracing and pharmacokinetic studies.
Q: How does this synthesis route improve supply chain stability compared to traditional methods?
A: By utilizing readily available betamethasone as a starting material and avoiding complex total synthesis from scratch, the route significantly reduces lead times and reliance on scarce chiral building blocks.
Q: Does the deuterium labeling affect the anti-inflammatory efficacy of the compound?
A: Biological evaluations indicate that the deuterated compound retains strong anti-inflammatory and anti-allergic activities, with the added benefit of a potentially prolonged metabolic half-life due to the kinetic isotope effect.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterium-Labeled Betamethasone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity isotopically labeled compounds play in accelerating drug discovery and development. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of deuterium-labeled betamethasone meets the >98% purity and >97% isotopic abundance benchmarks required for regulatory submissions.
We invite you to collaborate with us to optimize your supply chain for steroid intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can support your next breakthrough in pharmacokinetic research.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
