Advanced Synthesis of Deuterated P-Phenylenediamine-d4 for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is witnessing a paradigm shift towards deuterated drugs, driven by the need for enhanced metabolic stability and reduced toxicity profiles. Patent CN115073301B introduces a groundbreaking preparation method for partially deuterated p-phenylenediamine-d4, a critical building block for next-generation therapeutics. This innovation addresses the longstanding challenges in synthesizing amine-deuterated aromatic compounds, offering a route that is not only chemically efficient but also industrially viable. By leveraging a specific base-catalyzed exchange mechanism, the technology enables the precise replacement of hydrogen atoms on the amino groups with deuterium, achieving deuteration rates between 95% and 98%. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable deuterated pharmaceutical intermediate supplier capable of delivering high-purity materials essential for modern drug development pipelines.
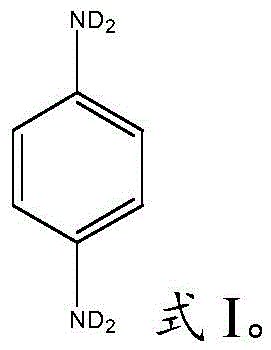
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-phenylenediamine and its derivatives has been plagued by severe operational hazards and environmental inefficiencies. Traditional methods, such as the hydrogenation of nitro compounds using Raney Nickel, often necessitate extreme reaction pressures and inert organic solutions, creating substantial safety risks for plant operators and requiring expensive high-pressure reactor infrastructure. Alternative routes involving iron powder reduction under acidic conditions are notoriously pollution-intensive, generating vast amounts of iron sludge that complicate waste treatment and drive up disposal costs. Furthermore, older synthetic strategies like the Hofmann degradation from terephthalic acid suffer from multi-step complexity and low overall yields, often failing to exceed 65%, which severely impacts the economic feasibility of large-scale production. These conventional pathways also lack the specificity required for deuteration, typically resulting in non-deuterated products or random deuteration on the benzene ring rather than the targeted amino positions, rendering them unsuitable for precision medicine applications.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a mild, base-catalyzed deuteration strategy that fundamentally simplifies the manufacturing landscape. By dissolving p-phenylenediamine in anhydrous deuterated solvents and employing catalysts like sodium methoxide or triethylamine, the process achieves high conversion rates without the need for dangerous high-pressure hydrogenation equipment. The method operates at moderate temperatures ranging from 70°C to 100°C, significantly lowering energy consumption and reducing the thermal stress on reaction vessels. Crucially, the iterative nature of the process, involving repeated dissolution and reaction cycles, ensures that the deuteration is concentrated specifically on the amino groups, providing the structural integrity required for effective deuterated drug molecules. This streamlined workflow not only enhances operation safety by eliminating explosive gas handling but also facilitates easier purification through simple distillation and washing steps, marking a substantial improvement in cost reduction in deuterated drug manufacturing.
Mechanistic Insights into Base-Catalyzed Deuteration
The core of this technological advancement lies in the efficient proton-deuterium exchange mechanism facilitated by strong bases in deuterated media. When p-phenylenediamine is introduced to a deuterated solvent such as anhydrous deuterated tetrahydrofuran or diethyl ether, the amino protons become susceptible to exchange in the presence of a catalytic amount of alkoxide or amine base. The catalyst activates the deuterated reagent, typically deuterated methanol or heavy water, generating a highly reactive deuteroxide species that rapidly attacks the N-H bonds. This nucleophilic attack promotes the reversible exchange of hydrogen for deuterium, driving the equilibrium towards the fully deuterated amine species through Le Chatelier's principle, especially when excess deuterated reagent is employed. The repetition of the dissolution and reaction steps three times serves to push this equilibrium further, effectively scrubbing residual protium from the amino sites and ensuring the final product meets the stringent isotopic purity standards demanded by regulatory bodies.
From an impurity control perspective, this mechanism offers distinct advantages over reduction-based methods which often generate over-reduced byproducts or metal-contaminated residues. Since the reaction does not involve transition metal catalysts like nickel or iron, the risk of heavy metal contamination in the final API intermediate is virtually eliminated, simplifying the downstream purification burden. The use of anhydrous conditions and a protective nitrogen atmosphere prevents the ingress of atmospheric moisture, which could otherwise introduce non-deuterated hydrogen and lower the isotopic enrichment. Furthermore, the choice of solvent plays a critical role in maintaining the solubility of the diamine while allowing for easy removal via reduced pressure distillation post-reaction. This clean reaction profile ensures that the resulting partially deuterated p-phenylenediamine-d4 possesses a narrow impurity spectrum, facilitating faster regulatory approval and more robust clinical trial outcomes for the final deuterated medicaments.
How to Synthesize Partially Deuterated P-Phenylenediamine-d4 Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing this high-value intermediate with consistent quality and yield. The process begins with the careful selection of anhydrous deuterated solvents to minimize isotopic dilution, followed by the precise addition of deuteration reagents and catalysts under inert conditions. Operators must maintain strict temperature control between 70°C and 100°C during the reaction phase to optimize the exchange kinetics without degrading the sensitive aromatic diamine structure. The detailed standardized synthesis steps below outline the specific molar ratios and processing times required to achieve the reported 95-98% deuteration rates and high isolated yields.
- Dissolve p-phenylenediamine in an anhydrous deuterated solvent such as deuterated diethyl ether or tetrahydrofuran to form a clear solution.
- Sequentially add a deuteration reagent like deuterated methanol and a catalyst such as sodium methoxide under a protective nitrogen atmosphere.
- Heat the mixture to 70-100°C for 12-24 hours, then repeat the dissolution and reaction cycle three times to achieve high deuteration rates.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible strategic benefits regarding cost, safety, and continuity. The elimination of high-pressure hydrogenation steps removes the need for specialized, capital-intensive reactor systems, thereby lowering the barrier to entry for manufacturing and reducing the overall fixed asset investment required for production facilities. Additionally, the avoidance of toxic metal catalysts like Raney Nickel or iron powder significantly cuts down on hazardous waste generation and the associated costs of environmental compliance and disposal. The simplicity of the workup procedure, involving basic distillation and washing, allows for faster batch turnover times, enhancing the overall throughput of the manufacturing plant and ensuring a more responsive supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing readily available, commodity-grade starting materials such as p-phenylenediamine and common deuterated solvents, avoiding the need for exotic or prohibitively expensive reagents. By operating at atmospheric pressure and moderate temperatures, the energy footprint of the reaction is drastically simplified compared to high-pressure alternatives, leading to lower utility costs per kilogram of product. Furthermore, the high yield reported in the examples, reaching up to 91%, minimizes raw material waste and maximizes the output from each batch, directly improving the gross margin for the manufactured intermediate. The absence of heavy metal catalysts also negates the need for costly metal scavenging steps and rigorous testing for residual metals, streamlining the quality control budget.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous reagents and standard glass-lined or stainless steel equipment ensures that production can be maintained continuously without the frequent downtime associated with maintaining high-pressure hydrogenation units. The robustness of the chemistry against minor fluctuations in conditions means that batch-to-batch variability is minimized, providing customers with a consistent supply of high-purity material essential for GMP manufacturing. Since the raw materials are commercially available and the process does not depend on single-source proprietary catalysts, the risk of supply disruption due to vendor issues is significantly mitigated. This stability allows for better long-term planning and inventory management, securing the supply chain against market volatility.
- Scalability and Environmental Compliance: The method is inherently scalable, as the heat transfer and mixing requirements are straightforward and do not present the engineering challenges typical of exothermic high-pressure reactions. The use of deuterated solvents, which can be recovered and recycled through distillation, aligns with green chemistry principles by reducing solvent waste and minimizing the environmental impact of the manufacturing process. The lack of heavy metal sludge or acidic waste streams simplifies wastewater treatment protocols, ensuring full compliance with increasingly stringent global environmental regulations. This eco-friendly profile not only reduces liability but also enhances the corporate sustainability metrics for companies integrating this intermediate into their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this deuterated intermediate, based on the specific data points and advantages highlighted in the patent literature. Understanding these details is crucial for technical teams evaluating the feasibility of incorporating this material into their drug development programs. The answers provided reflect the operational realities and performance metrics observed during the optimization of this synthesis route.
Q: What are the advantages of this deuteration method over traditional hydrogenation?
A: Unlike traditional Raney Nickel hydrogenation which requires severe reaction media and high pressure, this base-catalyzed method operates under mild conditions (70-100°C) with significantly improved safety and operational simplicity.
Q: What is the achievable deuteration rate for this intermediate?
A: The patented process consistently achieves a deuteration rate of 95-98%, ensuring precise control over the deuterated position for downstream drug synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method avoids hazardous high-pressure equipment and uses commercially available reagents, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Partially Deuterated P-Phenylenediamine-d4 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality deuterated intermediates play in the development of safer and more effective pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of partially deuterated p-phenylenediamine-d4 meets the exacting standards required for clinical and commercial use. Our commitment to technical excellence allows us to navigate the complexities of deuterated chemistry, delivering materials that empower your R&D efforts.
We invite you to collaborate with us to optimize your supply chain and accelerate your project timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the deuterated drug market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
