Advanced Synthesis of 1-Methylcarbapenem Intermediates: High Stereoselectivity and Industrial Scalability
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, driven by the relentless demand for more efficient and cost-effective synthetic routes. Patent CN101500992A introduces a groundbreaking industrially suitable production method for generating a critical 1-methylcarbapenem intermediate, specifically a beta-propiolactam derivative represented by general formula (II). This technology addresses long-standing challenges in the synthesis of carbapenems, which are renowned for their excellent antibacterial activity and high safety profile. Unlike conventional methods that rely on expensive and structurally complex auxiliary groups, this novel approach utilizes inexpensive raw materials and eliminates complicated processing steps. By leveraging a unique sulfonanilide auxiliary system combined with zirconium-mediated catalysis, the process achieves superior stereoselectivity and operational simplicity. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates while drastically reducing the cost of goods sold.
The limitations of conventional methods for synthesizing 1-methylcarbapenem intermediates have long been a bottleneck in antibiotic manufacturing. Traditional approaches, such as those disclosed in Japanese Patent Laid-Open Publications, often necessitate the use of auxiliary groups with special ring structures to pursue high stereoselectivity during carbon-carbon bond formation. These auxiliaries are not only expensive to procure but also require intricate removal steps, typically involving hydrolysis followed by carboxyl activation and subsequent chain extension. This multi-step sequence increases the potential for yield loss and impurity generation, complicating the purification process and inflating production costs. Furthermore, the reliance on specific reagents for smooth bond formation adds another layer of financial burden. In contrast, the novel approach detailed in this patent circumvents these issues by employing a sulfonanilide auxiliary group that is both inexpensive and versatile. This auxiliary can be easily prepared from common industrial raw materials like aniline and sulfonic acid derivatives, and crucially, it serves effectively as a leaving group without the need for the complex activation steps seen in prior art.
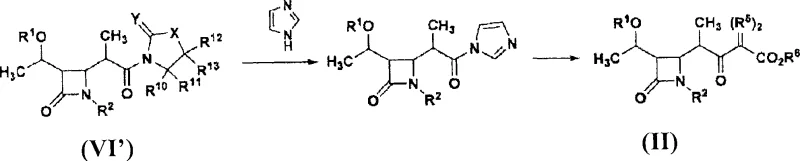
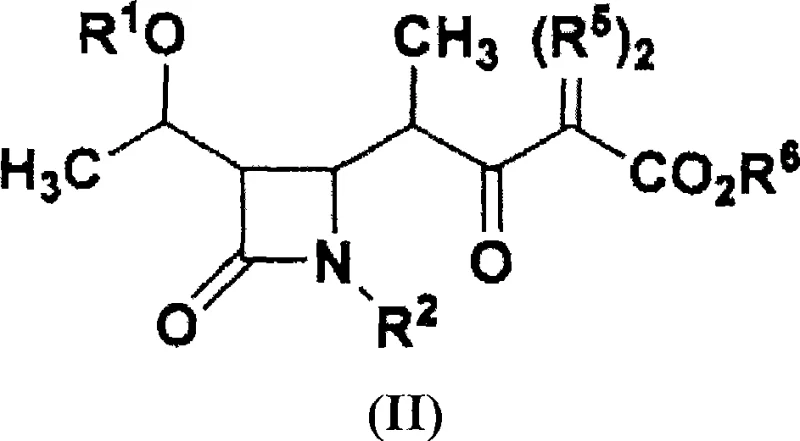
Mechanistic insights into the zirconium-catalyzed coupling reaction reveal why this method offers such robust performance for producing 1-beta-methylcarbapenem precursors. The core of this innovation lies in the reaction between a 4-acetoxy-beta-lactam derivative (Formula III) and an N-propionyl-sulfonanilide compound (Formula IV) in the presence of a zirconium reagent. While known methods using titanium or other Lewis acids might struggle to maintain sufficient stereoselectivity with simple sulfonanilide auxiliaries, the introduction of zirconium compounds, such as zirconium tetrachloride, consistently drives the formation of the desired beta-isomer. The zirconium center likely coordinates with the carbonyl oxygen and the auxiliary nitrogen, creating a rigid transition state that favors the specific spatial arrangement required for the 1-beta-methyl configuration. This high level of stereocontrol is paramount, as the biological activity of the final carbapenem antibiotic is heavily dependent on this specific stereochemistry. Additionally, the subsequent conversion of the intermediate (Formula I) to the target (Formula II) involves a mild reaction with imidazole followed by coupling with a malonate ester, ensuring that the delicate beta-lactam ring remains intact throughout the synthesis.
How to Synthesize 1-Methylcarbapenem Intermediate Efficiently
The synthesis of this critical antibiotic intermediate follows a logical and scalable pathway designed for industrial implementation. The process begins with the preparation of the key coupled product (Formula I) using the zirconium-mediated strategy described above, ensuring high diastereomeric excess from the outset. Following isolation, the intermediate undergoes a transformation where the sulfonanilide moiety is displaced. This is achieved by first activating the carbonyl with imidazole to form a reactive imidazolide species, which is then attacked by a malonate monoester in the presence of a magnesium compound and a base. This step effectively installs the side chain required for the final carbapenem structure. If the specific target requires a diazo functionality at the alpha-position, a subsequent diazotization step using an azide compound can be performed directly on the crude or purified material. This modular approach allows manufacturers to adapt the process for different downstream derivatives while maintaining a consistent and high-yielding core synthesis.
- Prepare the key precursor (Formula I) by reacting a 4-acetoxy-beta-lactam with an N-propionyl-sulfonanilide derivative in the presence of a zirconium reagent and base to ensure high stereoselectivity.
- Convert the Formula I intermediate into the target beta-keto ester (Formula II) by reacting with imidazole to form an imidazolide, followed by coupling with a malonate monoester using a magnesium compound.
- Optionally perform diazotization on the resulting compound using an azide compound and base to introduce the diazo functionality required for subsequent carbapenem ring closure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits beyond mere technical feasibility. The shift away from exotic, high-cost chiral auxiliaries to commodity chemicals like substituted anilines and sulfonyl chlorides fundamentally alters the cost structure of the intermediate. This substitution eliminates the dependency on niche suppliers for specialized reagents, thereby enhancing supply chain resilience and reducing the risk of raw material shortages. Furthermore, the simplification of the workflow by removing the activation and chain-extension steps translates directly into reduced processing time and lower utility consumption. The ability to isolate intermediates via simple precipitation and filtration, rather than complex chromatographic separations, further drives down operational expenditures. These factors combine to create a manufacturing process that is not only chemically elegant but also economically robust, enabling significant cost reduction in pharmaceutical intermediate manufacturing without compromising on quality or purity standards.
- Cost Reduction in Manufacturing: The elimination of expensive auxiliary groups and the reduction of synthetic steps lead to substantial savings in raw material and processing costs. By utilizing readily available sulfonanilide derivatives instead of complex cyclic auxiliaries, the direct material cost is significantly lowered. Additionally, the avoidance of carboxyl activation and re-chain extension steps reduces the consumption of reagents and solvents, streamlining the overall production budget and improving the margin profile for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Sourcing becomes more predictable and secure as the process relies on bulk chemicals such as anilines and sulfonic acid derivatives which are widely produced globally. This diversification of the supply base mitigates the risk of single-source bottlenecks often associated with specialized chiral reagents. The robustness of the zirconium-catalyzed reaction also ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results and ensuring a steady flow of materials to downstream formulation sites.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like cooling, stirring, and filtration that are easily replicated in large reactors. The simplified work-up procedures minimize the generation of hazardous waste streams associated with complex purification techniques. This aligns well with modern environmental, health, and safety (EHS) standards, facilitating regulatory approval and reducing the environmental footprint of the manufacturing facility, which is increasingly important for sustainable supply chain management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this patented synthesis method. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Whether you are concerned about stereochemical outcomes, raw material availability, or process safety, these insights provide a solid foundation for further technical discussions and feasibility assessments with our engineering team.
Q: What is the primary advantage of the sulfonanilide auxiliary group in this synthesis?
A: Unlike conventional auxiliary groups which often possess complex cyclic structures and require expensive removal steps, the sulfonanilide auxiliary (-N(R3)SO2R4) used in this patent is inexpensive, easily prepared from common anilines, and effectively functions as a leaving group, significantly simplifying the workflow.
Q: How does the zirconium reagent improve the synthesis of 1-beta-methylcarbapenem intermediates?
A: The use of zirconium reagents, such as zirconium tetrachloride, during the coupling of the beta-lactam and the sulfonanilide derivative ensures consistently high stereoselectivity. This is critical for obtaining the desired 1-beta-methyl configuration, which is often difficult to achieve with high purity using traditional titanium-based methods.
Q: Can this process be scaled for commercial production of antibiotic intermediates?
A: Yes, the process is designed for industrial suitability. It avoids complicated multi-step activation and chain extension procedures found in older methods. The use of stable reagents and standard work-up procedures like precipitation and filtration makes it highly amenable to large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Methylcarbapenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for life-saving antibiotics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 1-methylcarbapenem intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies like the zirconium-mediated synthesis described in CN101500992A, we can offer our partners a competitive edge through superior quality and consistent availability.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume requirements. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to this optimized route. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your next project milestone. Let us help you secure your supply chain with a partner dedicated to excellence in fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
