Advanced Catalytic Synthesis of 2,4-Dichloro-5-Fluoroacetophenone for Commercial Scale-Up
Introduction to Next-Generation Fluoroacetophenone Manufacturing
The pharmaceutical industry continuously demands more efficient and sustainable pathways for critical API intermediates, particularly for broad-spectrum antibiotics like ciprofloxacin. A groundbreaking technical solution detailed in patent CN113248354B introduces a superior synthetic method for 2,4-dichloro-5-fluoroacetophenone, a pivotal building block in quinolone antibiotic production. This innovation shifts away from traditional, waste-intensive acylation protocols toward a streamlined three-step sequence involving catalytic alkylation, selective photo-chlorination, and mild hydrolysis. By fundamentally re-engineering the carbon-carbon bond formation strategy, this approach addresses long-standing pain points regarding atom economy, operational safety, and downstream purification complexity. For R&D and procurement leaders seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages of this route is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2,4-dichloro-5-fluoroacetophenone has relied heavily on Friedel-Crafts acylation using acetyl chloride or acetic anhydride as the acylating agent. These conventional routes suffer from significant economic and environmental drawbacks that hinder cost reduction in pharmaceutical intermediates manufacturing. Specifically, the acylation process typically requires stoichiometric or excess amounts of aluminum chloride catalyst, often in ratios exceeding 1:1 relative to the substrate, which generates massive quantities of aluminum-containing sludge upon hydrolysis. Furthermore, the use of acetyl chloride poses severe safety risks due to its violent exothermic reaction with water during the quenching phase, necessitating energy-intensive ice-cooling systems to manage heat release. The generation of substantial hydrogen chloride gas during hydrolysis also imposes heavy burdens on scrubbing systems and increases neutralization costs, rendering these legacy processes increasingly unviable in a modern regulatory environment focused on green chemistry principles.
The Novel Approach
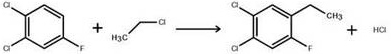
In stark contrast, the patented methodology employs a strategic Friedel-Crafts alkylation followed by side-chain functionalization, offering a transformative alternative for high-purity pharmaceutical intermediates production. Instead of direct acylation, the process initiates with the alkylation of 2,4-dichlorofluorobenzene using chloroethane, a significantly cheaper and safer gaseous reagent. Crucially, this step utilizes anhydrous aluminum chloride in merely catalytic quantities, specifically optimized at a molar ratio of 1:0.015, which drastically reduces catalyst consumption and subsequent waste generation. The resulting ethylbenzene intermediate is then subjected to photo-chlorination, leveraging the activating effect of the benzene ring to achieve high selectivity at the alpha-position. This clever sequence bypasses the need for hazardous acylating agents entirely, simplifying the post-treatment workflow to basic washing and distillation while maintaining exceptional conversion rates.
Mechanistic Insights into Photo-Chlorination and Hydrolysis
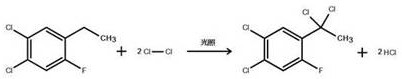
The core technical brilliance of this synthesis lies in the precise control of radical chemistry during the chlorination phase, which ensures superior regioselectivity and impurity profiles. Upon formation of the 2,4-dichloro-5-fluoroethylbenzene intermediate, the process introduces chlorine gas under ultraviolet irradiation at a controlled temperature of 40°C. The presence of the aromatic ring activates the benzylic alpha-hydrogens, making them highly susceptible to abstraction by photo-activated chlorine radicals. This electronic effect directs the chlorination exclusively to the alpha-carbon of the ethyl group, preventing unwanted ring chlorination or beta-position substitution that could complicate purification. The reaction proceeds to form an alpha,alpha-dichloro compound with high fidelity, utilizing approximately 2.2 equivalents of chlorine gas to drive the reaction to completion without excessive over-chlorination. This selectivity is paramount for R&D directors focused on impurity control, as it minimizes the formation of isomeric byproducts that are difficult to separate via standard distillation techniques.
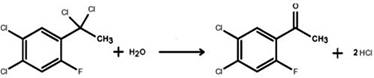
Following the chlorination step, the transformation concludes with a remarkably simple hydrolysis reaction that converts the gem-dichloro moiety directly into the target ketone functionality. Unlike the violent hydrolysis of acyl-aluminum complexes in traditional routes, this step involves the addition of pure water to the alpha-dichloro compound at a moderate temperature of 75°C. The mechanism involves the nucleophilic attack of water on the electron-deficient alpha-carbon, leading to the displacement of chloride ions and the formation of the carbonyl group. This mild hydrolytic condition eliminates the need for cryogenic cooling or complex pH adjustments, significantly reducing energy consumption and equipment corrosion risks. The final product is isolated via vacuum distillation, where tight control of the column top temperature between 125°C and 130°C ensures the collection of fractions with purity exceeding 99.90%, demonstrating the robustness of this chemical design for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2,4-Dichloro-5-Fluoroacetophenone Efficiently
Implementing this advanced synthetic route requires careful attention to reaction parameters to maximize yield and safety, particularly regarding gas flow rates and temperature gradients. The process begins with the introduction of chloroethane gas into the reactor containing the substrate and catalytic Lewis acid, where the feed rate is critically controlled to maintain a steady reaction exotherm over a period of approximately 1 hour. Following the alkylation, the photo-chlorination step demands precise UV light intensity and chlorine dosing to avoid the accumulation of unreacted starting material or polychlorinated impurities. Finally, the hydrolysis and distillation stages must be managed to prevent thermal degradation of the sensitive fluoro-ketone product. For detailed operational protocols, the standardized synthesis steps are outlined below.
- Perform Friedel-Crafts alkylation of 2,4-dichlorofluorobenzene with chloroethane using catalytic anhydrous aluminum chloride at 30°C.
- Conduct photo-chlorination of the resulting ethylbenzene derivative with chlorine gas under UV light at 40°C to form the alpha-dichloro intermediate.
- Hydrolyze the alpha-dichloro compound with water at 75°C followed by vacuum distillation to isolate the final ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound benefits that extend beyond mere technical feasibility, directly impacting the bottom line and supply reliability for global buyers. By shifting from stoichiometric acylation to catalytic alkylation, the process fundamentally alters the cost structure of manufacturing this key intermediate. The elimination of expensive acetyl chloride and the drastic reduction in aluminum chloride usage translate into substantial cost savings in raw material procurement. Furthermore, the simplified workup procedure, which avoids the generation of aluminum sludge and large volumes of acidic wastewater, significantly lowers waste disposal fees and environmental compliance costs. These factors combine to create a more resilient and economically attractive supply source for long-term contracts.
- Cost Reduction in Manufacturing: The transition to chloroethane as an alkylating agent represents a major shift in input costs, as this commodity chemical is generally more affordable and stable than corrosive acetyl chloride. Additionally, the reduction of the aluminum chloride catalyst from stoichiometric quantities to a mere 0.015 molar equivalent dramatically decreases the consumption of this reagent, which is often a significant cost driver in traditional Friedel-Crafts processes. The avoidance of ice-cooling infrastructure for exothermic quenching further reduces capital expenditure and operational energy costs, allowing for a leaner manufacturing footprint that enhances overall profit margins for suppliers.
- Enhanced Supply Chain Reliability: The simplicity of the reaction sequence, involving straightforward gas-liquid reactions and distillation, minimizes the risk of batch failures due to complex operational errors. The use of robust, non-hazardous reagents like chloroethane and chlorine gas, handled within standard industrial protocols, ensures consistent production schedules without the delays often associated with handling highly reactive acylating agents. This operational stability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturers receive their materials on schedule to meet their own production targets without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing unit operations that are easily replicated from pilot plant to multi-ton production scales without significant re-engineering. The environmental profile is markedly improved, as the primary byproducts are hydrogen chloride and water, which are easier to manage and treat compared to the complex organic-aluminum waste streams of conventional methods. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the risk of production shutdowns due to environmental violations, securing the long-term continuity of supply for partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthetic pathway. These insights are derived directly from the experimental data and process descriptions within the patent literature, providing a factual basis for evaluating the technology's potential impact on your supply chain. Understanding these details helps stakeholders make informed decisions about adopting this method for their specific manufacturing needs.
Q: How does this new synthetic route reduce production costs compared to traditional acylation?
A: The novel route replaces expensive acetyl chloride with cheaper chloroethane and reduces aluminum chloride usage from stoichiometric amounts to a catalytic 0.015 equivalents, significantly lowering raw material and waste treatment costs.
Q: What purity levels can be achieved with this photo-chlorination method?
A: The process demonstrates excellent selectivity, achieving gas-phase detection purity of 99.9% for the final 2,4-dichloro-5-fluoroacetophenone product after standard vacuum distillation.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method avoids hazardous ice-decomposition steps required for acetyl chloride hydrolysis and utilizes simple unit operations like gas introduction and distillation, making it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Dichloro-5-Fluoroacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the global pharmaceutical supply chain. Our team of expert chemists has thoroughly analyzed this patented route and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, ensuring that every batch of 2,4-dichloro-5-fluoroacetophenone performs consistently in your downstream antibiotic synthesis. Our facility is equipped to handle the specific gas-handling and distillation requirements of this process safely and efficiently.
We invite you to collaborate with us to optimize your sourcing strategy for this vital intermediate. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this new route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a more competitive and sustainable supply of high-quality pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
