Scalable and Safe Synthesis of N-Methylated Nitrogen-Containing Aromatic Heterocyclic Compounds for Commercial Production
The landscape of fine chemical synthesis is undergoing a critical transformation driven by the urgent need for safer, more sustainable, and highly selective manufacturing processes. Patent CN112694453B introduces a groundbreaking preparation method for N-methylated nitrogen-containing aromatic heterocyclic compounds that directly addresses the severe safety and selectivity limitations of legacy technologies. Traditionally, the introduction of a methyl group onto a nitrogen atom within an aromatic heterocycle has relied heavily on hazardous alkylating agents such as methyl iodide or dimethyl sulfate, which pose significant health risks and environmental burdens. This new methodology replaces those toxic reagents with specific aliphatic alcohol methyl ethers, utilized under the catalysis of an acidic substance. By shifting the reaction paradigm from basic to acidic conditions and utilizing benign methylating agents, this technology offers a robust pathway for producing high-purity pharmaceutical intermediates and agrochemical building blocks. For global supply chains, this represents a pivotal opportunity to enhance operational safety while maintaining rigorous quality standards for complex heterocyclic structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N-methylated heterocycles has been plagued by the reliance on extremely toxic methylating agents like methyl iodide and dimethyl sulfate. These reagents are not only carcinogenic and highly volatile, creating severe occupational health hazards, but they also necessitate complex containment systems and expensive waste treatment protocols to meet environmental regulations. Furthermore, conventional N-methylation reactions often lack the necessary chemoselectivity, particularly when the substrate molecule contains both aromatic nitrogen atoms and aliphatic amine groups. In many traditional processes, the methylating agent reacts indiscriminately with all available nucleophilic nitrogen sites, leading to a mixture of mono-methylated, di-methylated, and over-methylated byproducts. This lack of selectivity drastically complicates downstream purification, requiring energy-intensive chromatography or multiple recrystallization steps to isolate the desired product, thereby inflating production costs and reducing overall yield. Additionally, many standard protocols require basic conditions to deprotonate the nitrogen, which renders the process incompatible with base-sensitive functional groups commonly found in advanced drug candidates.
The Novel Approach
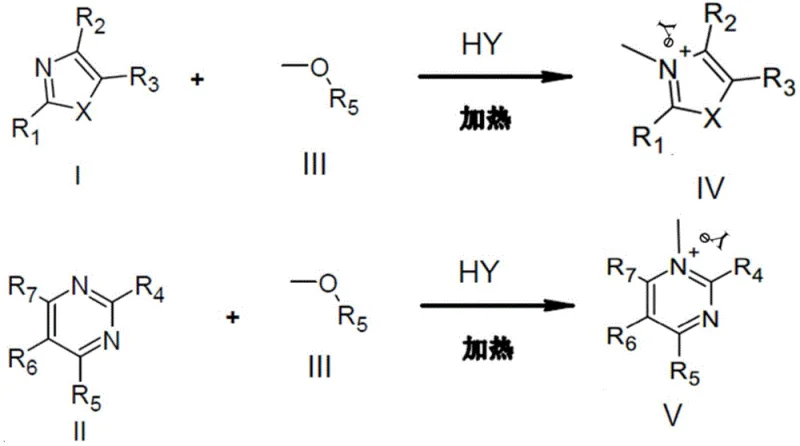
The methodology disclosed in patent CN112694453B fundamentally reengineers the N-methylation process by employing aliphatic alcohol methyl ethers as the methyl source in the presence of an acidic catalyst. This approach eliminates the need for toxic halogenated methylating agents, significantly lowering the EHS (Environment, Health, and Safety) footprint of the manufacturing process. The use of acidic catalysts, such as sodium bisulfate or potassium bisulfate, allows the reaction to proceed efficiently without the addition of basic substances, making it uniquely suitable for substrates that are unstable under alkaline conditions. Crucially, this method exhibits exceptional chemoselectivity, preferentially methylating the nitrogen atom within the aromatic heterocyclic ring while leaving aliphatic amine side chains untouched. This intrinsic selectivity minimizes the formation of impurities, simplifying the isolation process and enabling the direct crystallization of high-purity products. By combining safety, selectivity, and operational simplicity, this novel approach provides a commercially viable route for the large-scale production of complex nitrogen-containing heterocycles.
Mechanistic Insights into Acid-Catalyzed N-Methylation
The core innovation of this technology lies in the acid-mediated activation of the methyl ether bond, which transforms a typically inert ether into a potent electrophilic methylating agent. Under the influence of a strong acid (HY), the oxygen atom of the aliphatic alcohol methyl ether becomes protonated, significantly increasing the electrophilicity of the adjacent methyl group. This activated complex is then susceptible to nucleophilic attack by the lone pair of electrons on the nitrogen atom of the aromatic heterocycle. Unlike traditional SN2 reactions that might favor less hindered aliphatic amines, the specific electronic environment created by the acid catalyst and the aromatic system favors attack at the heterocyclic nitrogen. This mechanistic pathway ensures that the methyl group is transferred specifically to the aromatic ring nitrogen, forming a quaternary ammonium salt intermediate or the final neutral product depending on the workup. The reaction proceeds through a transition state that is stabilized by the acidic medium, allowing for efficient conversion even at moderate temperatures ranging from 20°C to 160°C.
Furthermore, the mechanism inherently suppresses side reactions that typically plague basic methylation conditions. In the absence of a base, there is no risk of base-catalyzed hydrolysis or elimination reactions that could degrade sensitive functional groups on the heterocyclic scaffold. The acid catalyst also serves a dual purpose by providing the counter-ion for the resulting quaternary ammonium salt, which often facilitates precipitation and isolation of the product directly from the reaction mixture. This streamlined mechanistic pathway reduces the number of unit operations required, as the product can often be isolated simply by distilling off the excess ether solvent and inducing precipitation with a non-solvent like diethyl ether. The ability to control the reaction kinetics through acid strength and temperature allows for fine-tuning of the process to maximize yield and purity, ensuring consistent quality across different batches of production.
How to Synthesize N-Methylated Nitrogen-Containing Aromatic Heterocyclic Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible framework for implementing this technology in a pilot or commercial setting. The process begins with the precise mixing of the nitrogen-containing aromatic heterocyclic substrate, the selected acidic catalyst, and the aliphatic alcohol methyl ether solvent-reagent. The reaction is typically conducted under an inert atmosphere, such as nitrogen, to prevent oxidation, although the method is robust enough to tolerate air in some variations. Heating the mixture to the optimal temperature range initiates the methylation, with reaction times varying from 2 to 120 hours depending on the specific reactivity of the substrate. Following the reaction completion, the workup procedure is remarkably simple, involving the removal of excess ether via distillation and subsequent precipitation. For a comprehensive understanding of the specific parameters and stoichiometry required for your target molecule, please refer to the detailed standardized synthesis steps provided below.
- Mix the nitrogen-containing aromatic heterocyclic compound (Formula I or II) with an acidic substance (HY) and an aliphatic alcohol methyl ether (Formula III) in a reactor.
- Heat the reaction mixture to a temperature between 20°C and 160°C, preferably under a nitrogen atmosphere, and maintain for 2 to 120 hours to allow N-methylation to occur.
- Distill off the remaining methyl ether solvent, dissolve the residue in methanol, precipitate with ether, and filter to collect the pure N-methylated product salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this acid-catalyzed N-methylation technology translates into tangible strategic advantages regarding cost, reliability, and regulatory compliance. The primary driver for cost optimization is the elimination of hazardous reagents, which removes the substantial overhead associated with the storage, handling, and disposal of toxic materials like methyl iodide. By replacing these controlled substances with common, low-cost methyl ethers, manufacturers can significantly reduce raw material procurement costs and minimize the need for specialized containment infrastructure. Moreover, the high selectivity of the reaction means that fewer resources are spent on purification; the reduction in byproduct formation leads to higher effective yields and less solvent consumption during recrystallization or chromatography. This efficiency gain directly impacts the cost of goods sold (COGS), making the final intermediates more competitive in the global market without compromising on quality specifications.
- Cost Reduction in Manufacturing: The shift away from expensive and heavily regulated toxic methylating agents results in substantial savings on raw material acquisition and waste management. Since the process does not require basic additives or complex quenching steps to neutralize hazardous byproducts, the operational expenditure related to chemical consumption and effluent treatment is drastically lowered. The simplified workup procedure, which often allows for direct precipitation of the product, reduces the demand for energy-intensive distillation and separation equipment, further driving down manufacturing costs. Additionally, the ability to recover and recycle the excess methyl ether solvent adds another layer of economic efficiency to the process, ensuring maximum resource utilization.
- Enhanced Supply Chain Reliability: Relying on toxic reagents like dimethyl sulfate often introduces supply chain vulnerabilities due to strict transportation regulations and limited supplier availability. By utilizing widely available aliphatic alcohol methyl ethers and common inorganic acids, this method diversifies the supply base and mitigates the risk of procurement bottlenecks. The robustness of the reaction conditions, which can tolerate a range of temperatures and atmospheres, ensures consistent production output even when facing minor variations in utility availability. This stability is crucial for maintaining continuous supply lines to downstream pharmaceutical customers who depend on just-in-time delivery of critical intermediates for their own synthesis campaigns.
- Scalability and Environmental Compliance: Scaling up chemical processes involving toxic gases or volatile liquids presents significant engineering challenges and regulatory hurdles. This novel method operates with liquid reagents under relatively mild conditions, facilitating a smoother transition from laboratory scale to multi-ton commercial production. The reduced toxicity profile aligns perfectly with increasingly stringent global environmental regulations, minimizing the permitting timeline for new production lines. Furthermore, the generation of less hazardous waste simplifies compliance reporting and reduces the long-term liability associated with chemical manufacturing, making it a sustainable choice for forward-thinking chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-methylation technology. These answers are derived directly from the experimental data and technical specifications detailed in the patent documentation, providing clarity on substrate scope, reaction conditions, and product quality. Understanding these nuances is essential for R&D teams evaluating the feasibility of this route for their specific pipeline compounds and for procurement teams assessing the reliability of the supply source.
Q: Why is this N-methylation method safer than traditional methods?
A: Traditional methods often use highly toxic reagents like methyl iodide or dimethyl sulfate. This patent utilizes aliphatic alcohol methyl ethers, which are significantly safer and less toxic, reducing EHS risks in manufacturing.
Q: Does this method work for compounds sensitive to base?
A: Yes, unlike conventional methods that often require basic conditions, this process operates under acidic catalysis. This makes it ideal for substrates that are unstable or sensitive to alkaline environments.
Q: What is the selectivity profile for molecules with multiple amine groups?
A: The method demonstrates high chemoselectivity for aromatic heterocyclic nitrogen over aliphatic amines. For example, it can selectively methylate the ring nitrogen in benzothiazole derivatives without affecting side-chain amine groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methylated Nitrogen-Containing Aromatic Heterocyclic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic routes is critical for the future of the pharmaceutical and fine chemical industries. Our team of expert process chemists has extensively evaluated the technology described in patent CN112694453B and possesses the capability to adapt this acid-catalyzed N-methylation strategy for a wide array of complex heterocyclic substrates. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits observed at the bench level are fully realized in large-scale manufacturing. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of N-methylated intermediates we produce, meeting the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain for these critical building blocks. By leveraging our expertise in this novel methylation chemistry, we can help you achieve significant process improvements and cost efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your development timelines and commercial goals effectively.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
