Advanced Catalyst-Free Synthesis of 1,2-Dihydro-3,6-Pyridazinedione for Commercial Scale-Up
The global demand for high-performance agrochemical and pharmaceutical intermediates is driving a critical shift towards greener, safer, and more cost-effective synthetic methodologies. Patent CN108546248B introduces a groundbreaking preparation method for 1,2-dihydro-3,6-pyridazinedione, a vital heterocyclic building block used extensively as a selective herbicide and plant growth inhibitor. Unlike conventional routes that rely on hazardous hydrazine derivatives, this innovation utilizes a benign oxidative cyclization strategy involving maleic anhydride, urea, and hydrogen peroxide in an aqueous medium. This technical breakthrough not only addresses severe environmental compliance issues associated with traditional manufacturing but also delivers a product with exceptional purity profiles exceeding 98% content by HPLC analysis. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this catalyst-free protocol represents a paradigm shift in sustainable chemical manufacturing, offering a robust pathway for the commercial scale-up of complex heterocyclic intermediates without the burden of toxic residue management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1,2-dihydro-3,6-pyridazinedione has been plagued by significant safety and environmental drawbacks inherent to hydrazine-based chemistry. Prior art, such as methods described in CN101735154A and CN102108063A, typically involves the reaction of hydrazine hydrate with maleic anhydride in the presence of sulfuric acid and organic acid catalysts. These processes generate substantial volumes of wastewater contaminated with inorganic salts and organic acid byproducts, creating a heavy burden on effluent treatment facilities and increasing operational expenditures. Furthermore, the use of hydrazine hydrate poses acute toxicity risks to personnel and necessitates rigorous purification steps to reduce residual hydrazine levels, which are strictly regulated due to their carcinogenic potential. Even advanced variations utilizing rare earth catalysts or ion-exchange resins fail to completely eliminate the fundamental hazards of the hydrazine feedstock, often resulting in higher production costs due to the expense of specialized catalytic systems and the complexity of downstream processing required to meet international purity standards.
The Novel Approach
The methodology disclosed in CN108546248B fundamentally reengineers the synthetic pathway by replacing the hazardous hydrazine source with urea and employing hydrogen peroxide as a clean oxidant. This approach leverages the structural similarity between urea and hydrazine while bypassing the toxicity issues entirely through an in-situ oxidative transformation. The reaction proceeds efficiently in water, eliminating the need for volatile organic compounds (VOCs) and expensive acid catalysts. As illustrated in the reaction scheme below, the process involves the condensation of maleic anhydride with urea followed by oxidative cyclization, releasing carbon dioxide as the only gaseous byproduct. This streamlined mechanism not only simplifies the reactor setup by removing the need for corrosion-resistant linings required for strong acids but also drastically reduces the E-factor of the process. By adopting this novel approach, manufacturers can achieve cost reduction in pharma intermediates manufacturing through simplified workup procedures and the avoidance of costly waste disposal fees associated with heavy metal or acidic effluents.
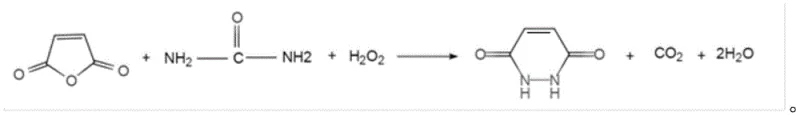
Mechanistic Insights into Aqueous Oxidative Cyclization
The core of this technological advancement lies in the unique mechanistic pathway where urea serves as a safe nitrogen source that is activated by hydrogen peroxide under thermal conditions. In the initial phase, maleic anhydride undergoes hydrolysis and subsequent amidation with urea in the aqueous phase at controlled temperatures between 10-40°C. The subsequent addition of hydrogen peroxide initiates an oxidative dehydrogenation sequence that facilitates the closure of the pyridazine ring. This oxidative cyclization is highly selective, minimizing the formation of polymeric byproducts or over-oxidized species that often plague radical-based oxidations. The absence of transition metal catalysts prevents the introduction of trace metal impurities, which is a critical quality attribute for active pharmaceutical ingredients (APIs) and high-end agrochemicals. The reaction kinetics are optimized by maintaining a molar ratio of maleic anhydride to urea to hydrogen peroxide at approximately 1:1.08:1.03, ensuring complete conversion of the anhydride while preventing the accumulation of unreacted oxidant that could degrade the product stability during the reflux period at 95-110°C.
Impurity control is inherently built into this aqueous system, addressing one of the most persistent challenges in the production of high-purity 1,2-dihydro-3,6-pyridazinedione. Traditional hydrazine routes often struggle with the removal of unreacted hydrazine sulfate or hydrazine hydrate, requiring multiple recrystallizations or chromatographic separations. In contrast, the urea-based route generates byproducts such as carbon dioxide and water, which are easily removed during the reaction or workup. The final crystallization from the aqueous mother liquor yields a product with a purity profile exceeding 98%, as confirmed by HPLC analysis against standard references. This high level of purity is achieved without the use of organic solvents for extraction, thereby reducing the risk of solvent inclusion in the crystal lattice. For supply chain managers focused on reducing lead time for high-purity agrochemical intermediates, this inherent purity advantage translates to fewer quality control failures and faster release times for batch production, ensuring a consistent supply of material that meets stringent regulatory specifications for herbicide formulations.
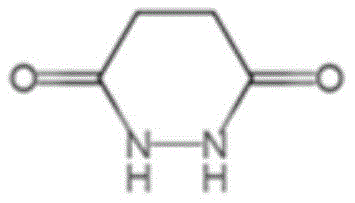
How to Synthesize 1,2-Dihydro-3,6-Pyridazinedione Efficiently
Implementing this synthesis on an industrial scale requires precise control over reaction parameters to maximize yield and safety. The process begins with the preparation of an aqueous slurry of maleic anhydride, followed by the controlled addition of a urea solution to manage the exotherm. The subsequent oxidative step with hydrogen peroxide must be managed carefully to prevent rapid gas evolution of carbon dioxide, which can cause foaming in large-scale reactors. Detailed operational protocols regarding stirring rates, addition times, and crystallization cooling curves are essential for reproducibility. The following guide outlines the standardized procedure derived from the patent examples to ensure optimal results.
- Charge maleic anhydride and water into a reactor, then slowly add urea aqueous solution while cooling to maintain temperature between 10-40°C.
- Dropwise add hydrogen peroxide solution while controlling the material temperature, then heat the mixture to reflux (95-110°C) for 3-8 hours.
- Cool the reaction mixture to crystallize the product, separate via centrifugation, wash with water, and dry to obtain >98% pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this urea-based synthesis route offers profound advantages that extend beyond mere technical feasibility. The elimination of hydrazine hydrate removes a highly regulated and dangerous raw material from the supply chain, mitigating risks associated with transportation, storage, and handling of toxic substances. This shift significantly enhances supply chain reliability by relying on commodity chemicals like urea and hydrogen peroxide, which are globally available and subject to less volatile pricing compared to specialized hydrazine derivatives or rare earth catalysts. Furthermore, the aqueous nature of the reaction simplifies the engineering requirements for production facilities, allowing for the use of standard glass-lined or stainless steel reactors without the need for exotic alloys resistant to strong mineral acids. These factors collectively contribute to a more resilient and cost-efficient manufacturing ecosystem.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the raw material bill and the reduction in waste treatment costs. By eliminating the need for organic solvents and acid catalysts, the process avoids the significant expenses associated with solvent recovery systems and neutralization of acidic waste streams. The use of water as the sole reaction medium means that washing and crystallization steps are inexpensive and environmentally benign. Additionally, the high yield range of 87-91% ensures efficient utilization of the maleic anhydride feedstock, minimizing material loss. The absence of expensive catalysts further lowers the variable cost per kilogram, providing a competitive margin advantage for producers aiming for cost reduction in pharma intermediates manufacturing.
- Enhanced Supply Chain Reliability: Securing a stable supply of critical intermediates is paramount for downstream formulators of herbicides and pharmaceuticals. This synthesis route relies on urea and hydrogen peroxide, which are produced on a massive global scale for fertilizer and pulp industries, respectively, ensuring that raw material availability is never a bottleneck. Unlike hydrazine, which is subject to strict security controls and limited production capacity in many regions, urea is a ubiquitous commodity. This accessibility reduces the risk of supply disruptions and allows for flexible sourcing strategies. Moreover, the simplified purification process shortens the overall production cycle time, enabling manufacturers to respond more rapidly to market demand fluctuations and reducing the inventory holding periods for finished goods.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but the aqueous nature of this reaction makes it inherently scalable from pilot plants to multi-tonne commercial reactors. The lack of organic solvents eliminates fire and explosion hazards, lowering insurance premiums and safety infrastructure costs. From an environmental standpoint, the process aligns perfectly with modern green chemistry principles by generating minimal hazardous waste. The primary byproduct, carbon dioxide, is vented safely, and the aqueous mother liquor contains mostly unreacted urea and salts that are far easier to treat biologically than organic solvent waste. This compliance with stringent environmental regulations future-proofs the manufacturing asset against tightening global emission standards and facilitates easier permitting for capacity expansion.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is crucial for stakeholders evaluating its integration into existing supply chains. The following questions address common concerns regarding safety, purity, and process robustness based on the data provided in the patent literature. These insights are intended to clarify the operational benefits and quality assurances associated with this innovative manufacturing route.
Q: Why is the urea-based route superior to traditional hydrazine hydrate methods?
A: Traditional methods utilize toxic hydrazine hydrate which often leaves hazardous residues in the final product. The novel urea-based route eliminates this risk entirely, ensuring the product meets international safety standards with no residual hydrazine.
Q: Does this process require organic solvents or catalysts?
A: No, the process is designed to be entirely green chemistry compliant. It uses water as the sole solvent and requires no organic acid or rare earth catalysts, significantly simplifying waste treatment and reducing raw material costs.
Q: What are the typical yield and purity specifications for this method?
A: According to patent data, this method consistently achieves a product content of greater than 98% (HPLC) with yields ranging from 87% to 91%, making it highly efficient for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Dihydro-3,6-Pyridazinedione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst-free aqueous synthesis described in CN108546248B for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle aqueous oxidative reactions with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 1,2-dihydro-3,6-pyridazinedione meets the >98% purity threshold required for sensitive agrochemical and pharmaceutical applications. We are committed to delivering not just a chemical product, but a comprehensive supply solution that prioritizes safety, sustainability, and consistency.
We invite procurement leaders and R&D teams to collaborate with us to optimize their supply chains using this advanced technology. By leveraging our expertise, you can achieve significant operational efficiencies and secure a long-term source of high-quality intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patent can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
