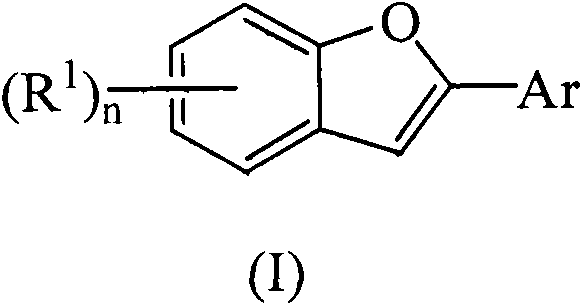
Advanced One-Step Synthesis of 2-Arylbenzofuran Compounds for Commercial Pharmaceutical Applications
Introduction to Next-Generation Benzofuran Synthesis
The field of fused heterocyclic compounds has long recognized the benzofuran skeleton as a cornerstone structure for drug discovery, attributed to its profound biopharmacological activities ranging from antitumor and antioxidant properties to immunosuppressive and anti-inflammatory effects. Notable examples such as XH-14, obovaten, and amiodarone underscore the critical demand for efficient access to these scaffolds. Addressing the historical challenges of multi-step syntheses and low yields, the innovative methodology disclosed in Chinese Patent CN103224479A introduces a transformative one-step approach. This technique leverages a sophisticated palladium catalytic system comprising palladium trifluoroacetate, bipyridine, and trifluoroacetic acid to couple o-hydroxyphenylacetonitriles with aryl trifluoroborates. By streamlining the construction of the 2-arylbenzofuran core, this patent provides a robust foundation for the reliable pharmaceutical intermediates supplier seeking to enhance their portfolio with high-value heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzofuran ring system has been plagued by significant synthetic inefficiencies that hinder large-scale manufacturing. Prior art methodologies, such as those utilizing olefin metathesis for ring closure, often necessitate cumbersome multi-step sequences that result in diminished overall throughput and complicated purification protocols. Alternative routes employing copper iodide catalysis with beta-ketoesters or titanium tetrachloride-mediated hydration of alkynes frequently suffer from harsh reaction conditions, limited substrate tolerance, and the generation of difficult-to-remove metal impurities. Furthermore, methods relying on Wittig reagents or complex hydrazone derivatives introduce additional safety hazards and cost burdens due to the instability of precursors. These traditional pathways collectively represent a bottleneck in cost reduction in benzofuran manufacturing, as the cumulative loss of material across multiple stages and the expense of specialized reagents render them less attractive for modern, lean chemical production environments.
The Novel Approach
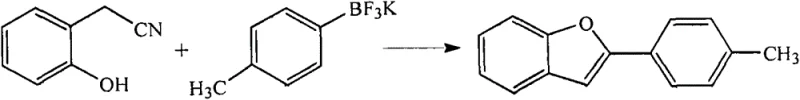
In stark contrast to these legacy techniques, the novel methodology described in CN103224479A achieves the direct cyclization of readily available starting materials in a single operational step. By utilizing stable aryl trifluoroborate salts instead of sensitive boronic acids, the process ensures superior handling safety and shelf-life stability. The core innovation lies in the specific catalytic triad of palladium trifluoroacetate, bipyridine ligands, and trifluoroacetic acid, which synergistically activate the nitrile and aryl groups for rapid ring closure. This streamlined protocol eliminates the need for isolating unstable intermediates, thereby drastically simplifying the workflow and minimizing waste generation. As illustrated in the general reaction scheme below, the transformation proceeds with remarkable efficiency, offering a direct path from simple precursors to complex 2-arylbenzofuran derivatives with high atom economy.
Mechanistic Insights into Pd-Catalyzed Cyclization
The success of this synthetic route is fundamentally rooted in the unique electronic properties of the palladium trifluoroacetate catalyst combined with the bipyridine ligand system. Mechanistically, the reaction likely initiates with the oxidative addition of the palladium species to the aryl trifluoroborate, facilitated by the acidic environment provided by trifluoroacetic acid. The electron-deficient nature of the trifluoroacetate ligands enhances the Lewis acidity of the palladium center, promoting the coordination and subsequent insertion of the nitrile group from the o-hydroxyphenylacetonitrile substrate. The bipyridine ligand plays a critical stabilizing role, preventing the aggregation of palladium black and maintaining the catalyst in its active oxidation state throughout the catalytic cycle. This precise tuning of the coordination sphere allows for the selective formation of the furan ring via reductive elimination, while suppressing side reactions such as homocoupling of the aryl boron species or hydrolysis of the nitrile.
From an impurity control perspective, this mechanism offers distinct advantages over copper-catalyzed alternatives. The high selectivity of the palladium system minimizes the formation of regioisomers and polymeric byproducts that often contaminate crude reaction mixtures in less controlled environments. The use of trifluoroacetic acid not only accelerates the cyclization kinetics but also helps in solubilizing intermediate species, ensuring a homogeneous reaction phase that leads to consistent product quality. Comparative studies within the patent data reveal that substituting the specific palladium source or omitting the acid additive results in catastrophic drops in yield, confirming that the observed high purity is a direct consequence of this specific mechanistic pathway. For R&D teams, understanding this delicate balance is key to troubleshooting and optimizing the process for diverse substrate libraries.
How to Synthesize 2-Arylbenzofuran Efficiently
Implementing this high-yield synthesis requires strict adherence to the optimized reaction parameters defined in the patent to ensure reproducibility and safety. The general procedure involves charging a reaction vessel equipped with agitation and temperature control with an appropriate organic solvent, such as toluene or tetrahydrofuran, followed by the sequential addition of the o-hydroxyphenylacetonitrile and the aryl trifluoroborate salt. The catalyst system is then introduced, typically comprising 0.5% to 3% palladium trifluoroacetate relative to the total mass of reactants, along with a stoichiometric amount of bipyridine and trifluoroacetic acid. The mixture is subjected to an inert atmosphere, usually nitrogen, to prevent oxidative degradation of the catalyst, and heated to temperatures ranging between 60°C and 140°C depending on the reactivity of the specific substrates. A representative example of this protocol, demonstrating the synthesis of 2-(p-tolyl)benzofuran, is depicted below to guide practical execution.
- Combine o-hydroxyphenylacetonitrile and aryl trifluoroborate salt in an organic solvent such as toluene or THF under inert atmosphere.
- Add palladium trifluoroacetate catalyst, bipyridine ligand, and trifluoroacetic acid additive in specific molar ratios.
- Heat the reaction mixture to 60-140°C for 1-10 hours, then cool, filter, and dry to obtain high-purity solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits beyond mere chemical yield. The shift from multi-step sequences to a one-pot cyclization significantly reduces the operational footprint required for production, lowering utility consumption and labor costs associated with intermediate isolation and purification. By utilizing aryl trifluoroborates, which are chemically stable and resistant to protodeboronation compared to traditional boronic acids, the supply chain gains resilience against raw material degradation during storage and transport. This stability ensures consistent quality input for the manufacturing process, reducing the risk of batch failures due to reagent variability. Furthermore, the simplified workup procedure, often involving simple filtration and drying, minimizes the volume of organic waste solvents generated, aligning with increasingly stringent environmental compliance standards and reducing disposal costs.
- Cost Reduction in Manufacturing: The elimination of expensive and sensitive reagents like Wittig salts or titanium tetrachloride removes significant cost drivers from the bill of materials. Additionally, the low catalyst loading requirement of 0.5% to 3% means that precious metal costs are kept to a minimum, while the high conversion rates reduce the need for costly recycling of unreacted starting materials. The qualitative improvement in process efficiency means that capital equipment can be turned over more rapidly, increasing overall plant capacity without the need for new infrastructure investment.
- Enhanced Supply Chain Reliability: Sourcing stable aryl trifluoroborate salts is generally more straightforward than managing the cold-chain logistics often required for sensitive organometallic reagents. This robustness allows for larger batch sizes and longer campaign runs, ensuring a steady flow of high-purity 2-arylbenzofuran intermediates to downstream API manufacturers. The reduced sensitivity to moisture and oxygen during the reaction setup further lowers the barrier for entry for contract manufacturing organizations, expanding the pool of qualified suppliers capable of executing this chemistry reliably.
- Scalability and Environmental Compliance: The reaction conditions, operating between 60°C and 140°C in common solvents like toluene or alcohols, are well-suited for standard stainless steel reactors found in most fine chemical facilities. The absence of highly toxic heavy metals like copper in the final product stream simplifies the purification process to meet stringent pharmaceutical specifications, thereby reducing the environmental burden of heavy metal waste treatment. This green chemistry profile supports sustainable manufacturing goals and facilitates smoother regulatory approvals for new drug applications containing these scaffolds.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed cyclization technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on catalyst selection, reaction conditions, and substrate scope. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production pipelines.
Q: Why is palladium trifluoroacetate preferred over other palladium sources?
A: Comparative data indicates that replacing palladium trifluoroacetate with palladium chloride or acetate significantly reduces yield or prevents reaction entirely, highlighting the unique efficacy of the trifluoroacetate ligand system.
Q: What is the role of trifluoroacetic acid in this catalytic system?
A: Trifluoroacetic acid acts as a crucial synergistic additive; experiments without it show yields dropping drastically to as low as 20%, proving its necessity for high-efficiency cyclization.
Q: Can this method be scaled for industrial production?
A: Yes, the method utilizes stable aryl trifluoroborate salts and standard organic solvents, avoiding sensitive reagents, which facilitates safe commercial scale-up of complex heterocyclic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Arylbenzofuran Supplier
As the global demand for bioactive heterocycles continues to surge, partnering with a technically proficient manufacturer is critical for securing your supply chain. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced catalytic technologies like the one described in CN103224479A to deliver superior value. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercialization. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-arylbenzofuran intermediate meets the exacting standards required for pharmaceutical synthesis.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this efficient one-step process. We encourage potential partners to contact us directly to obtain specific COA data for our catalog compounds and to receive detailed route feasibility assessments for your custom targets, ensuring a seamless transition from laboratory concept to commercial reality.
