Scalable Production of Renewable Dicarboxylic Acid Derivatives via Olefin Metathesis
The chemical industry is currently witnessing a paradigm shift towards sustainable manufacturing, driven by the urgent need to replace petroleum-dependent processes with renewable alternatives. Patent CN101558027A introduces a groundbreaking methodology for synthesizing alpha, omega-dicarboxylic acid alkene derivatives through advanced olefin metathesis chemistry. This technology leverages abundant natural feedstocks, such as vegetable oils and animal fats, to produce industrially critical organic compounds with unprecedented precision. By utilizing a strategic cross-metathesis step followed by isolation and subsequent self-metathesis, the process overcomes the historical challenge of obtaining mixed product distributions common in direct oil modification. For R&D directors and procurement specialists, this represents a viable pathway to high-purity pharmaceutical intermediates and polymer additives that align with green chemistry principles while maintaining economic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for producing long-chain dicarboxylic acids often rely on the oxidation of cyclic hydrocarbons derived from fossil fuels or the fermentation of sugars, both of which present significant logistical and chemical hurdles. Oxidation processes frequently suffer from poor selectivity, leading to complex mixtures of mono-acids, di-acids, and various chain-length byproducts that require energy-intensive separation protocols to purify. Furthermore, fermentation routes are limited by substrate specificity and often struggle to achieve the high concentrations necessary for cost-effective industrial recovery. These conventional pathways result in inconsistent supply chains and elevated production costs due to the extensive downstream processing required to meet the stringent purity specifications demanded by the pharmaceutical and specialty chemical sectors. The reliance on non-renewable petrochemical precursors also exposes manufacturers to volatile market fluctuations and regulatory pressures regarding carbon footprints.
The Novel Approach
In stark contrast, the novel approach detailed in the patent employs a sophisticated two-stage metathesis strategy that transforms simple unsaturated fatty esters into precise bifunctional building blocks. The process initiates with a cross-metathesis reaction between a renewable starting composition, such as soybean oil derivatives, and a short-chain olefin like 3-hexene. This step effectively cleaves the natural fatty acid chain at a predetermined double bond position, generating a functionalized olefin intermediate with a specific chain length. Crucially, this intermediate is isolated from the reaction mixture, often via distillation, ensuring that only the desired molecular species proceeds to the next stage. The purified intermediate then undergoes self-metathesis to dimerize, forming the target alpha, omega-dicarboxylic acid derivative with high structural fidelity. 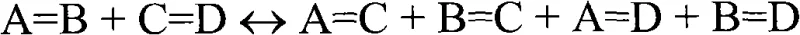
Mechanistic Insights into Ruthenium-Catalyzed Olefin Metathesis
The core of this transformative technology lies in the utilization of transition metal carbene catalysts, specifically those based on ruthenium, which facilitate the breaking and reforming of carbon-carbon double bonds with remarkable efficiency. The mechanism involves the formation of a metallacyclobutane intermediate upon the interaction of the metal carbene with the olefinic substrate, allowing for the exchange of alkylidene groups. In the context of this patent, catalysts such as C827 and C627 are employed to drive the cross-metathesis of unsaturated fatty esters with short-chain internal olefins. The choice of catalyst is critical, as it must tolerate the ester functional groups present in the renewable feedstocks without deactivation. The reaction conditions are carefully optimized, often involving inert atmospheres and controlled temperatures ranging from ambient to moderately elevated levels, to maximize turnover numbers while minimizing side reactions such as isomerization. This catalytic precision ensures that the double bond is relocated exactly where needed for the subsequent dimerization step.
Following the initial cross-metathesis, the separation of the functionalized olefin intermediate is a pivotal unit operation that distinguishes this process from bulk metathesis applications. Techniques such as fractional distillation or reactive distillation are utilized to separate the desired intermediate from unreacted starting materials and low-boiling olefinic byproducts. This purification step is essential for driving the equilibrium of the subsequent self-metathesis reaction towards the formation of the high-molecular-weight diester product. By removing volatile olefins generated during the self-metathesis, often under reduced pressure, the reaction equilibrium is shifted significantly to favor product formation. 
How to Synthesize Dicarboxylic Acid Esters Efficiently
The synthesis of these valuable intermediates requires a disciplined approach to reaction engineering, beginning with the preparation of the renewable feedstock. Unsaturated triglycerides are typically transesterified to free fatty acid methyl esters to ensure homogeneity and reactivity in the metathesis reactor. The process demands rigorous exclusion of oxygen and moisture to preserve catalyst activity, often utilizing argon purging and Schlenk line techniques during the laboratory optimization phase. Once the cross-metathesis is complete, the isolation of the intermediate must be performed with care to prevent thermal degradation, followed by the final dimerization step under vacuum to remove ethylene or other small olefins. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Perform cross-metathesis of unsaturated fatty esters with short-chain olefins using a ruthenium catalyst to form functionalized intermediates.
- Separate the functionalized olefin intermediate from the reaction mixture using distillation or other purification techniques.
- Subject the isolated intermediate to self-metathesis under vacuum to produce the desired alpha, omega-dicarboxylic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metathesis-based technology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the decoupling of production from volatile petrochemical markets by utilizing globally available renewable resources like soybean oil. This shift not only stabilizes raw material costs but also enhances the sustainability profile of the final product, which is increasingly a requirement for downstream customers in the pharmaceutical and consumer goods industries. The ability to produce specific chain-length diacids on demand reduces the need for holding large inventories of diverse specialty chemicals, thereby optimizing working capital. Furthermore, the high selectivity of the metathesis process minimizes waste generation and simplifies purification, leading to a more streamlined manufacturing workflow that reduces overall operational expenditures.
- Cost Reduction in Manufacturing: The elimination of complex multi-step oxidation sequences and the associated heavy metal waste treatment significantly lowers the cost of goods sold. By using highly active ruthenium catalysts that operate at lower loadings, the process reduces the expense associated with precious metal consumption and recovery. The simplified downstream processing, driven by the high purity of the intermediates, further contributes to cost efficiency by reducing solvent usage and energy consumption during distillation. These factors combine to create a leaner production model that is highly competitive against traditional petrochemical routes.
- Enhanced Supply Chain Reliability: Sourcing feedstocks from the agricultural sector provides a diversified supply base that is less susceptible to the geopolitical instabilities often affecting oil-producing regions. The modular nature of the metathesis process allows for flexible production scheduling, enabling manufacturers to respond quickly to changes in market demand for specific chain-length derivatives. Additionally, the robustness of the catalyst systems ensures consistent batch-to-batch quality, reducing the risk of supply disruptions caused by failed production runs or off-specification material. This reliability is crucial for maintaining long-term contracts with major pharmaceutical and polymer clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in reactor systems ranging from small laboratory flasks to larger pilot-scale vessels without loss of efficiency. The use of renewable feedstocks and the generation of benign byproducts like short-chain olefins, which can be recycled or used as fuel, align perfectly with modern environmental regulations and corporate sustainability goals. The reduction in hazardous waste streams simplifies compliance with environmental protection agencies, lowering the administrative and financial burden associated with waste disposal. This eco-friendly profile enhances the marketability of the final products in regions with strict carbon emission standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metathesis technology. They are derived from the specific embodiments and data presented in the patent documentation to provide clarity on process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing infrastructure. The answers reflect the current state of the art as described in the intellectual property, offering a realistic view of performance expectations.
Q: What feedstocks are suitable for this metathesis process?
A: The process utilizes renewable unsaturated fatty acids, esters, or salts derived from natural oils such as soybean oil, canola oil, and fish oil.
Q: How is product purity ensured in this synthesis?
A: High purity is achieved by isolating the functionalized olefin intermediate via distillation before the final self-metathesis step, removing mixed byproducts.
Q: Which catalysts are recommended for industrial scale-up?
A: Ruthenium-based metal carbene catalysts, such as Grubbs-type catalysts (e.g., C827, C627), are specified for their activity and tolerance to functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicarboxylic Acid Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of olefin metathesis in creating next-generation chemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity dicarboxylic acid esters that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in handling sensitive catalytic processes allows us to optimize yield and minimize impurities, providing our partners with a consistent and reliable supply of critical building blocks for their drug synthesis or polymer manufacturing needs.
We invite you to collaborate with us to explore how this renewable technology can enhance your product portfolio and reduce your environmental impact. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our metathesis-derived intermediates can serve as a superior alternative to traditional petrochemical sources. Let us help you secure a sustainable and cost-effective supply chain for your most critical chemical inputs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
