Advanced One-Pot Synthesis of 1,3-bis(methylthio)propan-2-ol for High-Purity Nifuratel Manufacturing
Introduction to Patented Impurity Synthesis
The pharmaceutical industry faces constant pressure to ensure the highest levels of purity and safety for active pharmaceutical ingredients (APIs), particularly for broad-spectrum antibiotics like Nifuratel. A critical aspect of quality control involves the precise characterization and quantification of related substances, or impurities, which can arise during synthesis or storage. Patent CN112745249A introduces a groundbreaking preparation method for Nifuratel Related Substance B, chemically known as 1,3-bis(methylthio)propan-2-ol. This patent addresses a significant gap in the market where previous methods relied on difficult isolation from reaction mixtures, resulting in low yields and poor purity. By establishing a dedicated synthetic route, this technology enables the reliable production of reference standards and high-purity intermediates essential for rigorous QC protocols.
The significance of this development extends beyond mere compliance; it fundamentally alters the supply dynamics for this critical intermediate. Traditionally, obtaining sufficient quantities of specific impurities for method validation was a bottleneck, often requiring complex chromatographic separations that were neither cost-effective nor scalable. The disclosed method utilizes a straightforward one-pot strategy starting from epichlorohydrin, a commodity chemical, thereby democratizing access to high-quality material. For R&D directors and procurement specialists, this represents a shift towards more predictable supply chains and reduced dependency on scarce, low-yield byproducts. The ability to synthesize this substance on demand with high fidelity supports the broader goal of maintaining stringent drug safety standards globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN112745249A, the acquisition of Nifuratel Related Substance B was fraught with inefficiencies and technical hurdles. The conventional approach primarily involved attempting to isolate the substance directly from the crude reaction liquid of the main Nifuratel synthesis. This method suffered from inherently low yields because the impurity was formed only in trace amounts as a side reaction, making its concentration in the mixture negligible. Consequently, isolating meaningful quantities required processing vast volumes of reaction mass, leading to excessive solvent consumption and waste generation. Furthermore, the separation process was technically demanding, often necessitating preparative liquid chromatography, which is slow, expensive, and difficult to scale beyond gram quantities.
From a supply chain perspective, relying on isolation from a main reaction stream creates significant vulnerability. The availability of the impurity becomes tied to the production schedule of the parent drug, creating bottlenecks when large batches of the reference standard are needed for regulatory filings or stability testing. Additionally, the purity of the isolated material was often inconsistent, varying with each batch of the main API produced. This inconsistency complicates the validation of analytical methods, as stable and well-characterized references are paramount for accurate quantification. The high cost and low reliability of this traditional sourcing method hindered comprehensive impurity profiling, potentially leaving gaps in the safety assessment of the final drug product.
The Novel Approach
The novel approach presented in the patent revolutionizes this landscape by decoupling the production of the impurity from the main API synthesis. Instead of scavenging for trace byproducts, the method employs a targeted, constructive synthesis starting from epichlorohydrin and sodium methyl mercaptide. This strategic shift allows for the deliberate formation of the 1,3-bis(methylthio)propan-2-ol skeleton in high yields, reported in the examples to range from 86.5% to 89.5%. The process is designed as a one-pot reaction, which drastically simplifies the operational workflow by eliminating the need for intermediate isolation steps. This consolidation of steps not only saves time but also reduces the physical loss of material that typically occurs during multiple work-up procedures.
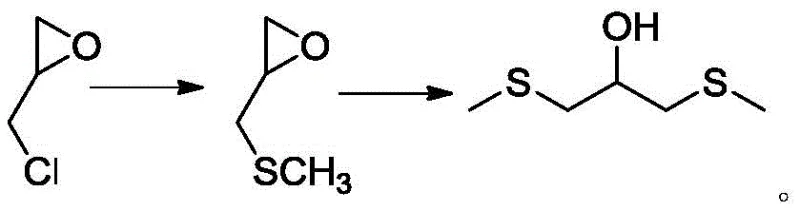
Furthermore, the new route utilizes robust and scalable chemistry. The reaction conditions are mild, with the first step occurring at 0-10°C and the second step at moderate heating of 50-90°C. These temperatures are easily achievable in standard industrial reactors without requiring specialized cryogenic or high-pressure equipment. The use of specific catalysts, namely 18-crown-6 and Zinc Chloride (ZnCl2), ensures high regioselectivity and conversion rates, minimizing the formation of other unwanted byproducts. This results in a crude product of such high purity that simple distillation under reduced pressure is sufficient for final purification. For manufacturers, this translates to a streamlined process that is both economically viable and technically robust, ensuring a steady supply of high-purity material for the pharmaceutical sector.
Mechanistic Insights into the Catalytic Cascade
The success of this synthesis lies in the careful orchestration of nucleophilic substitution reactions facilitated by dual catalytic systems. The process begins with the ring-opening of the epoxide moiety in epichlorohydrin. Sodium methyl mercaptide acts as a potent nucleophile, attacking the less hindered carbon of the epoxide ring. The addition of 18-crown-6 ether plays a critical role here as a phase transfer catalyst. By complexing with the sodium cation, the crown ether increases the nucleophilicity of the methyl mercaptide anion in the organic phase, facilitating a rapid and efficient SN2 attack even at low temperatures (0-10°C). This step selectively installs the first methylthio group while retaining the chloromethyl functionality, setting the stage for the subsequent transformation.
Following the initial substitution, the reaction mixture is treated with Zinc Chloride (ZnCl2) and heated. ZnCl2 acts as a Lewis acid catalyst, coordinating with the chlorine atom of the intermediate to enhance its leaving group ability. This activation lowers the energy barrier for the second nucleophilic substitution, where another equivalent of sodium methyl mercaptide displaces the chloride ion. The controlled heating to 50-90°C provides the necessary thermal energy to drive this second substitution to completion without degrading the sensitive thioether linkages. The result is the symmetrical 1,3-bis(methylthio)propan-2-ol structure. This mechanistic pathway avoids the formation of polymeric byproducts or over-alkylation, which are common pitfalls in thiol-epoxide chemistry, thereby ensuring the high purity observed in the patent examples.
Impurity control is intrinsic to this mechanism due to the stoichiometry and catalyst loading. By using a slight excess of sodium methyl mercaptide (2.2 to 3.0 molar equivalents) relative to epichlorohydrin, the reaction drives towards full conversion of the starting material, minimizing residual epichlorohydrin. The specific loading of ZnCl2 (0.01 to 0.05 molar equivalents) is optimized to catalyze the substitution without promoting side reactions such as elimination or rearrangement. The final distillation step effectively removes any unreacted thiols or solvent residues, leveraging the distinct boiling point of the target di-thioether. This combination of kinetic control via catalysis and thermodynamic separation via distillation creates a robust impurity profile, delivering a product suitable for the most demanding analytical applications.
How to Synthesize 1,3-bis(methylthio)propan-2-ol Efficiently
Implementing this synthesis requires precise adherence to the patented parameters to replicate the high yields and purity. The process is designed for operational simplicity, making it accessible for both laboratory-scale reference standard production and larger commercial batches. The key to success lies in the controlled addition of reagents and the maintenance of specific temperature profiles during the two distinct reaction phases. Operators must ensure that the exothermic nature of the initial epoxide opening is managed via ice bath cooling to prevent runaway reactions or non-selective ring opening. Following the initial addition, the introduction of the Lewis acid catalyst must be timed correctly to initiate the second substitution phase efficiently.
- Slowly add sodium methyl mercaptide to epoxy chloropropane at 0-10°C with 18-crown-6 catalyst.
- Add ZnCl2 catalyst and heat the mixture to 50-90°C for 3-5 hours.
- Cool to room temperature and distill under reduced pressure to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers profound strategic benefits that extend well beyond the laboratory. The transition from an isolation-based model to a dedicated synthesis model fundamentally de-risks the supply of this critical intermediate. By utilizing epichlorohydrin and sodium methyl mercaptide, both of which are commodity chemicals with established global supply chains, manufacturers eliminate the dependency on the erratic availability of Nifuratel crude reaction masses. This independence ensures business continuity and allows for better inventory planning, as production can be scheduled based on demand rather than the whims of API campaign schedules. The reliability of raw material sourcing is a cornerstone of a resilient supply chain, and this method secures that foundation.
- Cost Reduction in Manufacturing: The economic implications of this one-pot synthesis are substantial. By consolidating the reaction into a single vessel and eliminating the need for preparative chromatography, the process drastically reduces operational expenditures. The high yield (consistently approaching 90%) means that less raw material is wasted per unit of product, directly lowering the cost of goods sold (COGS). Furthermore, the avoidance of expensive transition metal catalysts or exotic reagents keeps input costs low. The simplified work-up, requiring only distillation, reduces solvent usage and energy consumption associated with complex purification trains. These factors combine to deliver a significantly more cost-effective manufacturing process compared to traditional isolation methods.
- Enhanced Supply Chain Reliability: The scalability of this process is a major asset for supply chain stability. The reaction conditions (0-10°C and 50-90°C) are compatible with standard glass-lined or stainless steel reactors found in most fine chemical facilities. There are no requirements for extreme pressures or cryogenic temperatures that would limit the number of qualified contract manufacturing organizations (CMOs). This universality means that production can be easily scaled from kilograms to tons without re-engineering the core chemistry. For buyers, this translates to shorter lead times and the assurance that suppliers can ramp up production quickly to meet surges in demand for Nifuratel quality control materials.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the process is inherently cleaner. The high atom economy of the substitution reactions minimizes waste generation. The use of catalytic amounts of ZnCl2 and 18-crown-6 reduces the load of heavy metals and organic additives in the waste stream compared to stoichiometric reagents. The simplified purification via distillation avoids the massive solvent volumes associated with column chromatography, reducing the facility's volatile organic compound (VOC) emissions. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental compliance burden on manufacturing sites, making it a sustainable choice for long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,3-bis(methylthio)propan-2-ol. These answers are derived directly from the technical specifications and experimental data provided in patent CN112745249A. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their quality control workflows or further synthesis pipelines. The clarity provided here aims to bridge the gap between patent theory and practical industrial application.
Q: What is the primary advantage of this new synthesis method for Nifuratel Related Substance B?
A: The primary advantage is the shift from low-yield isolation from reaction liquids to a direct, high-yield one-pot synthesis, significantly improving purity and availability.
Q: What catalysts are utilized in this patented process?
A: The process utilizes 18-crown-6 ether as a phase transfer catalyst in the first step and Zinc Chloride (ZnCl2) to facilitate the second substitution reaction.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method uses readily available starting materials like epichlorohydrin and operates under manageable temperature conditions, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-bis(methylthio)propan-2-ol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final pharmaceutical product depends on the quality of every component in your supply chain, including critical impurities and intermediates. Our team has extensively analyzed the synthetic route described in CN112745249A and possesses the technical expertise to execute this one-pot synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need grams for analytical method validation or tons for commercial manufacturing, our capacity meets your needs. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,3-bis(methylthio)propan-2-ol meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain for Nifuratel-related materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. By partnering with us, you gain access to a transparent and responsive supply partner dedicated to supporting your regulatory and commercial goals. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance the efficiency and reliability of your pharmaceutical operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
