Advanced Manufacturing of 1,2,6,7-Tetrahydro-8H-indeno[5,4-b]furan-8-one for Global Pharma Supply Chains
Introduction to Patent CN113402490B Technology
The pharmaceutical industry continuously seeks robust, scalable, and cost-efficient pathways for critical API intermediates, particularly for high-demand therapeutics like Ramelteon. Patent CN113402490B introduces a groundbreaking preparation method for 1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-one, a pivotal building block in the synthesis of melatonin receptor agonists. This technology addresses long-standing inefficiencies in prior art by utilizing p-hydroxy-phenyl propionic acid as a readily available, low-cost starting material, thereby fundamentally shifting the economic landscape of this intermediate's production. The disclosed route achieves a remarkable total yield of 73-78% through a concise five-step sequence that integrates halogenation, etherification, acylation, cyclization, and catalytic hydrogenation. By avoiding hazardous reagents like sodium hydride and cryogenic ozone treatments found in legacy methods, this process offers a safer, greener, and commercially superior alternative for global supply chains seeking reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-one has been plagued by complex multi-step sequences that rely on prohibitively expensive raw materials and dangerous reaction conditions. For instance, earlier patents such as WO9732871A1 utilized 2,3-dihydrobenzofuran as a starting material, which commands a significantly higher market price compared to simple phenolic acids, thereby inflating the overall cost of goods sold. Furthermore, these traditional routes often necessitate the use of sodium hydride, a pyrophoric reagent that introduces severe safety risks and requires specialized handling equipment, making industrial scale-up challenging and costly. Other approaches, like those described in WO2011044990A1, depend on ozonolysis at cryogenic temperatures of -78°C, demanding energy-intensive refrigeration infrastructure and creating bottlenecks in production throughput. Additionally, many conventional pathways suffer from poor atom economy and low yields in key cyclization steps, with some reports indicating yields as low as 9% for monobromide intermediates, rendering them economically unviable for commercial manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN113402490B leverages a strategic retrosynthetic analysis that begins with p-hydroxy-phenyl propionic acid, a commodity chemical that ensures consistent supply and minimal cost volatility. This innovative route streamlines the synthesis into just five operational steps, effectively collapsing what were previously eight or more distinct stages into a cohesive, high-efficiency workflow. A standout feature of this approach is the implementation of 'one-pot' strategies, particularly during the halogenation-etherification and acylation-cyclization sequences, which drastically reduce solvent consumption and waste generation. By replacing hazardous reagents with safer alternatives like hydrobromic acid and thionyl chloride, the process mitigates operational risks while maintaining high reaction selectivity. The result is a robust manufacturing protocol that delivers high-purity products with a total yield exceeding 73%, representing a substantial improvement over the fragmented and low-yielding processes of the past.
![General reaction scheme for the synthesis of 1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-one from p-hydroxy-phenyl propionic acid](/insights/img/ramelteon-intermediate-synthesis-supplier-20260308190722-06.webp)
Mechanistic Insights into Dual Friedel-Crafts Cyclization
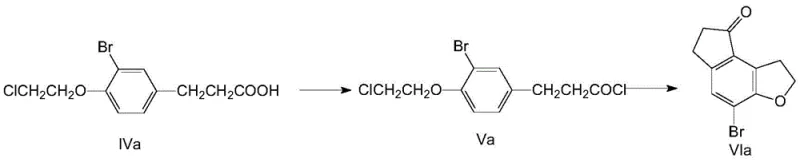
The core chemical innovation of this patent lies in the sophisticated execution of a dual Friedel-Crafts reaction sequence, which constructs the rigid indeno-furan scaffold with exceptional precision. The process initiates with the conversion of the chloroethoxy intermediate into an acyl chloride using thionyl chloride, activating the side chain for electrophilic attack. Upon the addition of aluminum trichloride (AlCl3) at controlled low temperatures (0-10°C), an intramolecular acylation occurs, forming the initial ketone ring structure. Subsequently, the reaction temperature is elevated to 60-70°C with the addition of further Lewis acid catalyst, triggering a second intramolecular alkylation where the chloroethyl side chain cyclizes to form the furan ring. This tandem cyclization strategy is critical because it builds two rings in a single operational vessel, minimizing intermediate isolation steps that typically lead to material loss and increased processing time. The careful modulation of temperature and catalyst stoichiometry ensures that the reaction proceeds through the desired transition states without generating excessive polymeric byproducts or isomeric impurities.
Impurity control is further enhanced in the final stage through a selective catalytic hydrogenation step that serves a dual purpose: removing the halogen substituent (bromine or iodine) and ensuring the final product meets stringent purity specifications. The use of palladium on carbon (Pd/C) in the presence of sodium acetate creates a mild yet effective environment for hydrodehalogenation, preventing the reduction of the sensitive ketone functionality which could otherwise lead to alcohol impurities. This selectivity is paramount for R&D directors focused on impurity profiles, as it simplifies downstream purification and ensures the intermediate is suitable for direct coupling in Ramelteon synthesis. The mechanistic elegance of using a halogen as a temporary directing group that is cleanly removed at the end exemplifies a 'traceless' synthetic strategy, maximizing atom economy and reducing the burden on waste treatment facilities.
How to Synthesize 1,2,6,7-Tetrahydro-8H-indeno[5,4-b]furan-8-one Efficiently
Implementing this synthesis route requires precise control over reaction parameters, particularly during the exothermic acylation and cyclization phases, to ensure safety and reproducibility. The process begins with the halogenation of p-hydroxy-phenyl propionic acid using hydrogen peroxide and hydrobromic acid, followed immediately by etherification with 1-bromo-2-chloroethane under basic conditions to yield the key chloroethoxy precursor. This precursor is then activated to an acyl chloride and subjected to the dual Friedel-Crafts cyclization in nitrobenzene, a high-boiling solvent that facilitates the elevated temperatures required for ring closure. The final step involves catalytic hydrogenation in toluene to remove the halogen, yielding the target ketone with high purity. For detailed standard operating procedures and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Halogenation and Etherification: React p-hydroxy-phenyl propionic acid with HBr/H2O2 followed by 1-bromo-2-chloroethane to form the chloroethoxy intermediate.
- Dual Friedel-Crafts Cyclization: Convert the acid to acyl chloride using SOCl2, then perform sequential acylation and alkylation using AlCl3 in nitrobenzene to close the indeno-furan rings.
- Catalytic Hydrogenation: Perform debromination/deiodination using Pd/C and sodium acetate in toluene to yield the final high-purity ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages, primarily driven by the substitution of exotic raw materials with commodity chemicals. The shift from expensive specialty starting materials to p-hydroxy-phenyl propionic acid significantly reduces the baseline cost of raw materials, providing a buffer against market volatility and ensuring long-term price stability for the intermediate. Furthermore, the elimination of hazardous reagents like sodium hydride and ozone removes the need for specialized storage and handling protocols, thereby lowering insurance premiums and facility compliance costs associated with dangerous goods. The simplified workflow, characterized by fewer isolation steps and 'one-pot' operations, directly correlates to reduced labor hours and lower utility consumption per kilogram of product, driving down the overall manufacturing overhead.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the drastic simplification of the synthetic route, which reduces the number of unit operations from eight or more down to just five. By consolidating reaction steps and eliminating intermediate purification stages, the process minimizes solvent usage and waste disposal costs, which are often hidden but significant expenses in fine chemical manufacturing. The high overall yield of 73-78% means that less raw material is required to produce the same amount of finished goods, effectively amplifying the purchasing power of the procurement budget. Additionally, the use of recoverable catalysts and common solvents like toluene allows for efficient recycling loops, further enhancing the cost-efficiency profile of the production line.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the primary starting material, p-hydroxy-phenyl propionic acid, is a widely produced commodity chemical with a stable global supply base, unlike niche precursors that may face shortages. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high vacuum systems, ensures that production can be maintained consistently across different manufacturing sites without complex technology transfers. This operational resilience reduces the risk of supply disruptions caused by equipment failure or specialized utility limitations, guaranteeing a steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers. Consequently, partners can plan their inventory levels with greater confidence, knowing that lead times are predictable and not subject to the whims of fragile, high-maintenance synthetic routes.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is designed for seamless transition from pilot plant to commercial metric-ton scale, adhering to modern green chemistry principles. The avoidance of ozone and heavy metal contaminants simplifies the effluent treatment process, reducing the environmental footprint and ensuring compliance with increasingly stringent global regulatory standards. The process generates less hazardous waste and utilizes solvents that are easier to recover and recycle, aligning with corporate sustainability goals and reducing the carbon intensity of the supply chain. This scalability ensures that as demand for Ramelteon grows, the production capacity for its key intermediate can be expanded rapidly without the need for prohibitive capital investment in specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis pathway. These insights are derived directly from the experimental data and comparative analysis presented in patent CN113402490B, providing a clear understanding of its operational superiority. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing supply networks.
Q: What are the primary cost drivers eliminated in this new synthesis route?
A: The process eliminates the need for expensive starting materials like 2,3-dihydrobenzofuran and hazardous reagents such as sodium hydride or ozone, significantly lowering raw material and safety compliance costs.
Q: How does the 'one-pot' cyclization strategy impact production efficiency?
A: By combining acyl chloride formation and the subsequent dual Friedel-Crafts reactions without intermediate isolation, the method reduces solvent usage, processing time, and potential yield losses associated with multiple work-ups.
Q: Is this process scalable for commercial metric-ton production?
A: Yes, the use of common industrial solvents like toluene and nitrobenzene, along with robust catalysts like aluminum trichloride and palladium on carbon, ensures the route is highly amenable to large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,6,7-Tetrahydro-8H-indeno[5,4-b]furan-8-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals depends on the availability of high-quality, cost-effective intermediates produced via scalable routes. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,6,7-tetrahydro-8H-indeno[5,4-b]furan-8-one adheres to the highest industry standards. We are committed to leveraging advanced technologies like the one described in CN113402490B to deliver value-driven solutions that enhance your competitive edge in the global marketplace.
We invite you to collaborate with our technical procurement team to explore how this optimized synthesis route can specifically benefit your project timelines and budget. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic impact of switching to this superior manufacturing method. Please contact us today to discuss your requirements, and we will promptly provide specific COA data and comprehensive route feasibility assessments tailored to your unique development goals.
