Scalable Catalyst-Free Synthesis of 1,5-Azaspiro[2,4]heptane for Pharmaceutical Applications
The development of efficient synthetic routes for complex heterocyclic scaffolds remains a critical priority for the global pharmaceutical industry, particularly for structures exhibiting potent biological activity. Patent CN108299445B introduces a groundbreaking methodology for the synthesis of 1,5-azaspiro[2,4]heptane derivatives, a privileged structural motif found in various anticancer and antiviral agents. Unlike conventional approaches that rely on harsh chemical environments, this invention utilizes a thermal cyclization strategy starting from vinyl azide compounds. The process operates entirely without the need for transition metal catalysts or acidic and basic additives, representing a significant leap forward in green chemistry and process safety. By leveraging an intramolecular Alder-ene reaction mechanism, the technology ensures high stereochemical fidelity, yielding products with a single cis-configuration. This technical advancement addresses long-standing challenges in the manufacturing of spirocyclic intermediates, offering a streamlined pathway that is highly attractive for industrial scale-up and regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1-azaspiro[2,4]heptane core has been plagued by inefficient and environmentally burdensome synthetic strategies. One prevalent method involves the protonation and dehydration of (1-aminocyclopentyl)methanol under strongly acidic conditions, typically utilizing concentrated sulfuric acid followed by neutralization with sodium hydroxide. As illustrated in the reaction scheme below, this approach not only generates substantial amounts of inorganic salt waste but also poses severe safety risks due to the handling of corrosive reagents. Furthermore, the harsh acidic environment can lead to side reactions and decomposition of sensitive functional groups, complicating the purification process and lowering overall yields. Another reported route employs sulfenimide indolone substrates reacting with sulfur ylides generated in situ using strong bases like sodium hydride. This method suffers from similar drawbacks, requiring stringent anhydrous conditions and expensive, difficult-to-prepare starting materials. The reliance on strong bases and the generation of sulfur-containing byproducts create significant hurdles for waste treatment and increase the cost of goods significantly.
![Conventional acid-base catalyzed synthesis of 1-azaspiro[2,4]heptane showing harsh reaction conditions](/insights/img/1-5-azaspiro-heptane-synthesis-pharma-supplier-20260308125248-01.webp)
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes vinyl azide compounds as versatile precursors that undergo thermal activation to form the desired spirocyclic framework. The general reaction scheme demonstrates that by simply heating the vinyl azide substrate in an aprotic organic solvent such as toluene, the molecule undergoes a spontaneous rearrangement and cyclization. This catalyst-free protocol eliminates the need for any external acid or base promoters, thereby drastically simplifying the workup procedure. The absence of metal catalysts means there is no risk of heavy metal contamination in the final API intermediate, a crucial factor for regulatory approval in pharmaceutical manufacturing. Moreover, the reaction conditions are mild enough to tolerate a wide range of substituents on the nitrogen atom and the alkene chain, allowing for the rapid generation of diverse chemical libraries. This operational simplicity translates directly into reduced processing time and lower energy consumption, making it a superior choice for cost-effective manufacturing.
![General reaction scheme for catalyst-free thermal synthesis of 1,5-azaspiro[2,4]heptane from vinyl azides](/insights/img/1-5-azaspiro-heptane-synthesis-pharma-supplier-20260308125248-03.webp)
Mechanistic Insights into Thermal Alder-Ene Cyclization
The success of this synthesis relies on a sophisticated yet elegant mechanistic pathway involving the formation of a reactive azapropenidine intermediate. Upon heating, the vinyl azide moiety loses nitrogen gas to generate a highly reactive nitrene species, which rapidly rearranges into the azapropenidine intermediate M. This transient species is key to the reaction's success, as it positions the nitrogen atom and the pendant alkene chain in close proximity for the subsequent cyclization step. The reaction then proceeds via an intramolecular Alder-ene reaction, a concerted pericyclic process that forms two new sigma bonds and shifts a pi bond simultaneously. This specific mechanistic trajectory is responsible for the observed high stereoselectivity, as the transition state geometry favors the formation of the cis-configured product exclusively. Understanding this mechanism allows chemists to predict the outcome of various substrate modifications and optimize reaction parameters such as temperature and solvent polarity to maximize efficiency.
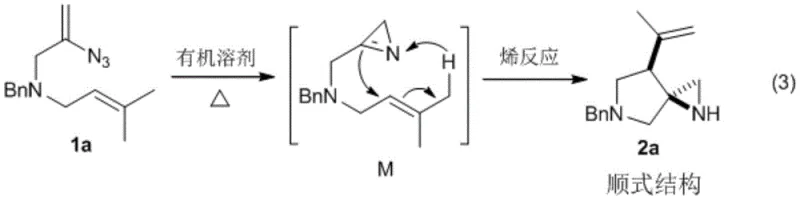
Furthermore, the mechanistic pathway provides inherent advantages regarding impurity control and product purity. Because the reaction is driven by thermal activation rather than chemical reagents, the potential for reagent-derived impurities is virtually eliminated. The concerted nature of the Alder-ene reaction minimizes the formation of regioisomers or stereoisomers that often plague stepwise ionic cyclizations. The exclusive formation of the cis-isomer simplifies downstream purification, often removing the need for complex chiral separation techniques or extensive recrystallization steps. This high level of intrinsic selectivity ensures that the final product meets stringent purity specifications required for pharmaceutical applications. Additionally, the byproduct of the initial azide decomposition is merely nitrogen gas, which escapes the reaction mixture, driving the equilibrium forward and preventing the accumulation of toxic or difficult-to-remove side products. This clean reaction profile is a testament to the power of pericyclic reactions in modern organic synthesis.
How to Synthesize 1,5-Azaspiro[2,4]heptane Efficiently
To implement this synthesis effectively, operators must adhere to precise thermal parameters and solvent choices outlined in the patent examples. The process begins with the dissolution of the specific vinyl azide precursor in dry toluene under an inert nitrogen atmosphere to prevent oxidation or moisture interference. The reaction mixture is then heated to a controlled temperature range, typically between 60°C and 120°C, depending on the specific substitution pattern of the substrate. Monitoring the reaction progress is essential, with typical reaction times ranging from 6 to 15 hours to ensure complete conversion of the starting material. Once the reaction is deemed complete, the solvent is removed under reduced pressure, and the crude residue is purified using standard silica gel column chromatography. Detailed standardized synthetic steps and specific optimization parameters for various substrates are provided in the guide below.
- Dissolve the vinyl azide precursor in an aprotic organic solvent such as toluene under a nitrogen atmosphere.
- Heat the reaction mixture to a temperature between 60°C and 120°C and maintain for 6 to 15 hours to facilitate cyclization.
- Remove the solvent under reduced pressure and purify the residue via silica gel column chromatography to isolate the pure cis-configured product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this catalyst-free thermal synthesis offers profound advantages for procurement managers and supply chain directors seeking to optimize their manufacturing budgets. The most significant benefit is the drastic reduction in raw material costs associated with the elimination of expensive transition metal catalysts and stoichiometric amounts of strong acids or bases. By removing these costly reagents from the bill of materials, manufacturers can achieve substantial cost savings per kilogram of produced intermediate. Furthermore, the simplified workup procedure reduces the consumption of solvents and consumables required for neutralization and extraction, further driving down operational expenditures. The robustness of the supply chain is also enhanced because the starting materials, such as primary amines and allyl bromides, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. This availability ensures consistent production schedules and mitigates the risk of delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The elimination of catalysts and harsh reagents directly lowers the cost of goods sold by reducing both material expenses and waste disposal fees. Without the need for specialized catalyst recovery systems or extensive neutralization steps, the overall process becomes significantly more economical. The high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted, which is a critical factor in maintaining competitive pricing in the fine chemical market.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available vinyl azide precursors ensures a reliable supply chain that is less susceptible to geopolitical or logistical disruptions. Since the synthesis does not require exotic or highly regulated reagents, procurement teams can source materials from a broader network of qualified vendors. This flexibility allows for better negotiation leverage and ensures continuity of supply even during market fluctuations, providing a strategic advantage for long-term production planning.
- Scalability and Environmental Compliance: The thermal nature of the reaction makes it inherently scalable, as heat transfer can be easily managed in large-scale reactors without the complexities of mixing heterogeneous catalysts. The absence of heavy metals and strong acids simplifies environmental compliance, reducing the burden on wastewater treatment facilities and lowering the cost of regulatory reporting. This green chemistry profile aligns perfectly with modern sustainability goals, making the facility more attractive to eco-conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation. They are designed to provide clarity on the operational feasibility and strategic benefits of adopting this new route for 1,5-azaspiro[2,4]heptane production.
Q: What is the primary advantage of the vinyl azide cyclization method over traditional acid-catalyzed routes?
A: The primary advantage is the elimination of harsh reagents. Traditional methods require strong acids like sulfuric acid and strong bases for neutralization, generating significant waste. The new thermal method operates catalyst-free, simplifying purification and reducing environmental impact.
Q: Does this synthesis method control the stereochemistry of the final spiro compound?
A: Yes, the method exhibits excellent stereocontrol. The reaction proceeds through a specific azapropenidine intermediate that undergoes an intramolecular Alder-ene reaction, resulting exclusively in the single cis-configuration of the 1,5-azaspiro[2,4]heptane scaffold.
Q: Are the raw materials for this synthesis readily available for large-scale production?
A: Yes, the vinyl azide precursors are synthesized from commercially available primary amines, propargyl bromide, and allyl bromides. The preparation involves standard nucleophilic substitutions and a silver-catalyzed azidation, making the supply chain robust and scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Azaspiro[2,4]heptane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalyst-free synthesis technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle thermal cyclization processes with precision, maintaining stringent purity specifications and rigorous QC labs to guarantee product quality. We understand the critical importance of timeline and consistency in the pharmaceutical supply chain, and our dedicated technical team is ready to support your specific needs with customized solutions.
We invite you to collaborate with us to leverage this innovative synthesis route for your next drug development program. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time to market with our reliable and efficient manufacturing capabilities.
