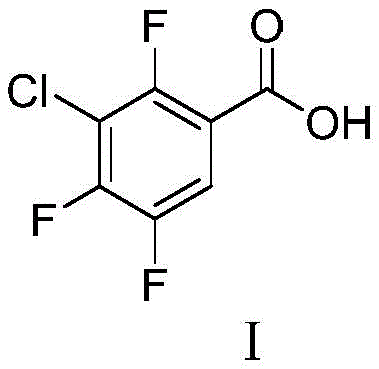
Advanced Synthesis of 2,4,5-Trifluoro-3-Chlorobenzoic Acid for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antibiotic intermediates, particularly those serving next-generation fluoroquinolones. Patent CN109293503B introduces a highly efficient preparation method for 2,4,5-trifluoro-3-chlorobenzoic acid, a pivotal building block in the synthesis of Sitafloxacin. This broad-spectrum antibacterial agent has demonstrated superior activity against Streptococcus pneumoniae and Enterobacteriaceae compared to earlier generations like ciprofloxacin. The disclosed technology addresses long-standing challenges in process chemistry by utilizing 1,2,4-trifluorobenzene as a cost-effective starting material. By streamlining the reaction sequence through nitration, chlorination, reduction, and Sandmeyer cyanidation, the method achieves high yields while maintaining operational simplicity. For R&D Directors and Procurement Managers, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. The structural integrity of the final product is paramount for downstream drug efficacy, and this patent provides a verified roadmap to achieve consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2,4,5-trifluoro-3-chlorobenzoic acid has relied on several distinct synthetic routes, each plagued by specific inefficiencies that hinder large-scale commercial adoption. One common approach initiates with 3-chloro-4-fluoroaniline, necessitating a cumbersome ten-step sequence that includes amino protection, nitration, deprotection, and subsequent chlorination. This excessive step count not only accumulates material losses at each stage but also drastically increases labor and solvent costs, making it economically unviable for competitive API manufacturing. Another existing method starts from 2,4-dichloro-5-fluorobenzoic acid, requiring six steps involving reduction and diazotization; however, the initial raw materials for this route are often difficult to source reliably, creating supply chain bottlenecks. Direct chlorination of 2,4,5-trifluorobenzoic acid offers fewer steps but suffers from inherently low yields due to poor regioselectivity, leading to significant waste generation. Furthermore, routes utilizing 3,4,5,6-tetrachlorophthalic acid involve complex fluorination and ammoniation steps that pose safety risks and environmental compliance challenges. These conventional limitations collectively result in higher production costs and inconsistent batch quality, which are critical pain points for supply chain heads managing global inventory.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages 1,2,4-trifluorobenzene to establish a mature and stable process route that significantly enhances industrial value. By strategically sequencing nitration and chlorination reactions, the method ensures high regioselectivity, minimizing the formation of unwanted isomers that complicate purification. The process operates under relatively mild conditions compared to high-energy alternatives, reducing the demand for specialized high-pressure equipment and lowering overall capital expenditure. The simplicity of operation allows for easier scale-up from laboratory to commercial production, ensuring that the high yields observed in examples can be replicated in large reactors. This route eliminates the need for complex protecting group strategies, thereby reducing the consumption of auxiliary reagents and solvents. For a reliable pharmaceutical intermediates supplier, adopting this methodology translates to a more resilient production capability that can withstand market fluctuations. The robustness of this chemistry supports the consistent delivery of high-purity intermediates, essential for meeting the stringent regulatory requirements of global drug manufacturers.
Mechanistic Insights into the Multi-Step Synthetic Route
The core of this synthesis lies in the precise control of electrophilic aromatic substitution and functional group transformations. The process begins with the nitration of 1,2,4-trifluorobenzene using a mixture of nitric and sulfuric acid, where the electron-withdrawing fluorine atoms direct the incoming nitro group to the 6-position, yielding 1,2,6-trifluoronitrobenzene with exceptional efficiency. Subsequent chlorination is performed under pressure, introducing the chlorine atom at the 3-position relative to the nitro group, a step that is critical for establishing the final substitution pattern. The second nitration further functionalizes the ring, setting the stage for the reduction of the nitro group to an amine using hydrogen and a catalyst such as Raney Ni or Pd/C. This reduction step must be carefully monitored to prevent over-reduction or dehalogenation, ensuring the integrity of the fluorine and chlorine substituents. The resulting aniline derivative then undergoes diazotization at low temperatures to form the diazonium salt, a highly reactive intermediate that is immediately consumed in the Sandmeyer cyanidation reaction. This sequence transforms the amino group into a nitrile, which is subsequently hydrolyzed under alkaline conditions to yield the target carboxylic acid. Each transformation is optimized to maximize atom economy and minimize byproduct formation.
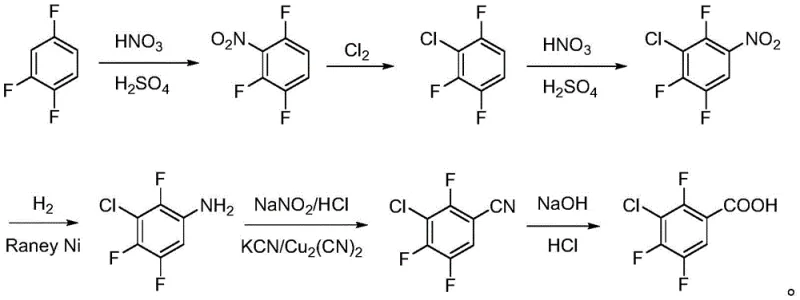
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this route incorporates several mechanisms to ensure a clean impurity profile. The use of specific molar equivalents of reagents, such as controlling the ratio of nitric acid to sulfuric acid, prevents over-nitration which could lead to dinitro impurities that are difficult to remove. During the chlorination step, maintaining the reaction temperature between 60-150°C and controlling chlorine pressure helps avoid polychlorinated byproducts. The reduction step utilizes catalytic hydrogenation, which is generally cleaner than metal-acid reductions, reducing the risk of metal contamination in the final product. In the Sandmeyer reaction, the precise addition of the diazonium salt to the cyanide solution minimizes the formation of phenolic byproducts resulting from hydrolysis of the diazonium group. Finally, the hydrolysis of the nitrile is conducted under controlled alkaline conditions, followed by acidification to precipitate the product, which serves as an effective purification step to remove inorganic salts and water-soluble impurities. This rigorous control over reaction parameters ensures that the final 2,4,5-trifluoro-3-chlorobenzoic acid meets the stringent purity specifications required for API synthesis.
How to Synthesize 2,4,5-Trifluoro-3-Chlorobenzoic Acid Efficiently
Implementing this synthesis requires a systematic approach to reaction engineering and process safety. The patent outlines a seven-step sequence that transforms simple starting materials into a complex, highly functionalized benzoic acid derivative. Operators must pay close attention to temperature control during the exothermic nitration steps and manage gas evolution during the chlorination and reduction phases. The transition from the nitro compound to the amine and subsequently to the nitrile involves handling hazardous reagents such as cyanides and diazonium salts, necessitating strict safety protocols and containment measures. Detailed standard operating procedures should be established for each unit operation, from reagent dosing to workup and isolation. The following guide provides a structured overview of the critical process parameters derived from the patent examples, serving as a foundation for process development teams aiming to replicate this high-yield route.
- Nitration of 1,2,4-trifluorobenzene using nitric and sulfuric acid at 30-90°C to form 1,2,6-trifluoronitrobenzene.
- Chlorination under pressure (1-5 atm) at 60-150°C to introduce the chlorine atom at the 3-position.
- Second nitration, followed by catalytic reduction, diazotization, Sandmeyer cyanidation, and final hydrolysis to yield the target benzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the selection of 1,2,4-trifluorobenzene as the starting material, which is commercially available in large quantities and at a lower cost compared to the specialized anilines or benzoic acids required by older methods. This shift in raw material sourcing significantly reduces the cost reduction in pharmaceutical intermediates manufacturing by lowering the baseline material input costs. Furthermore, the mature nature of the process route implies a lower risk of batch failure, enhancing supply chain reliability and ensuring consistent availability of the intermediate for downstream API production. The simplicity of the operation reduces the need for highly specialized labor and complex equipment maintenance, contributing to overall operational efficiency. By minimizing the number of purification steps and improving overall yield, the process also reduces waste disposal costs and environmental compliance burdens. These factors collectively create a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive protecting group chemistry and the use of commodity chemicals like 1,2,4-trifluorobenzene drastically lower the raw material expenditure. The high yields reported in the patent examples indicate that less starting material is wasted per unit of product, directly improving the cost of goods sold. Additionally, the simplified workup procedures reduce solvent consumption and energy usage during distillation and drying phases. This economic efficiency allows for more competitive pricing strategies in the global market for antibiotic intermediates.
- Enhanced Supply Chain Reliability: Relying on easily obtained raw materials mitigates the risk of supply disruptions caused by shortages of niche precursors. The robustness of the reaction conditions means that production can be maintained across different facilities without significant re-validation, supporting a distributed manufacturing model. This flexibility is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that drug manufacturers can meet their production schedules without delay. The consistent quality of the output further reduces the need for extensive incoming quality control testing by customers.
- Scalability and Environmental Compliance: The process is designed for commercial scale-up of complex pharmaceutical intermediates, with reaction conditions that are manageable in standard stainless steel reactors. The reduction in step count and the avoidance of hazardous reagents where possible contribute to a smaller environmental footprint. Efficient waste management is facilitated by the cleaner reaction profile, making it easier to comply with increasingly strict environmental regulations. This scalability ensures that supply can be ramped up quickly to meet surges in demand for Sitafloxacin and related antibiotics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,4,5-trifluoro-3-chlorobenzoic acid. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation. They are intended to clarify the feasibility and advantages of this specific synthetic route for industry stakeholders. Understanding these details is essential for making informed decisions about sourcing and process adoption.
Q: What is the primary starting material for this synthesis route?
A: The process utilizes 1,2,4-trifluorobenzene as the initial raw material, which is noted for being cheap and easily obtained compared to alternatives like 3-chloro-4-fluoroaniline.
Q: How does this method improve upon conventional preparation techniques?
A: Unlike conventional methods that suffer from low yields or complex multi-step protections, this route offers a mature process with simple operations and significantly higher industrial value.
Q: What are the critical reaction conditions for the Sandmeyer step?
A: The Sandmeyer cyanidation reaction requires precise temperature control between 20-80°C and specific molar equivalents of cuprous cyanide and potassium cyanide to ensure high conversion to the nitrile intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trifluoro-3-Chlorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving antibiotics. Our technical team has extensively analyzed the pathway described in CN109293503B and is fully equipped to execute this synthesis with precision and scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are designed to handle the specific safety requirements of nitration and cyanidation reactions, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering 2,4,5-trifluoro-3-chlorobenzoic acid that meets the highest industry standards, supporting your drug development timelines and commercial manufacturing goals.
We invite you to collaborate with us to optimize your supply chain for Sitafloxacin intermediates. Our team can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By leveraging our expertise in fine chemical synthesis, we can help you secure a stable and cost-effective source of this critical building block. Let us support your mission to bring advanced antibiotic therapies to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
