Scalable Amantadine Production: A Green Polyionic Liquid Catalytic Route for High-Purity Pharmaceutical Intermediates
Scalable Amantadine Production: A Green Polyionic Liquid Catalytic Route for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with environmental sustainability, particularly for critical antiviral intermediates like amantadine. Patent CN112939798A introduces a transformative preparation method that leverages a novel polyionic liquid (PIL-1) catalyst to streamline the conversion of adamantane into high-purity amantadine. This technology addresses longstanding inefficiencies in traditional Ritter reactions by replacing excessive liquid acid usage with a recoverable solid acid system. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the commercial scale-up of complex pharmaceutical intermediates while adhering to stricter environmental regulations. The process not only achieves impressive yields exceeding 90% under optimized conditions but also simplifies downstream processing, making it an ideal candidate for reliable pharmaceutical intermediates supplier networks aiming to secure stable, cost-effective supply chains for antiviral drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for amantadine and its derivatives have historically relied on harsh reaction conditions that pose significant challenges for industrial scalability and environmental compliance. Conventional methods often utilize large excesses of concentrated sulfuric acid or require expensive pre-functionalized starting materials like adamantanol. For instance, comparative data in the patent highlights that methods employing copper catalysts or aluminum salts with adamantanol result in lower yields and higher raw material costs. Furthermore, processes relying solely on liquid mineral acids generate substantial quantities of waste acid, necessitating complex neutralization and disposal procedures that inflate operational expenditures. The use of volatile organic solvents and the difficulty in separating homogeneous catalysts from the product mixture further complicate the purification process, often leading to metal impurities that are unacceptable for high-grade API intermediate applications. These factors collectively hinder the ability to achieve cost reduction in pharmaceutical intermediates manufacturing while maintaining the rigorous quality standards required by global regulatory bodies.
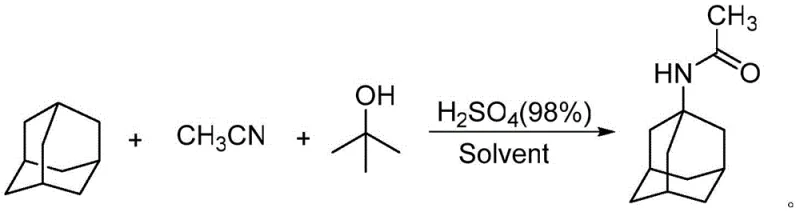
The Novel Approach
The innovative methodology disclosed in CN112939798A overcomes these barriers by introducing a polyionic liquid catalyst (PIL-1) that functions as a highly efficient, heterogeneous solid acid. This approach enables the direct amidation of inexpensive adamantane with acetonitrile, bypassing the need for costly adamantanol precursors. The PIL-1 catalyst, characterized by its cross-linked polymer structure containing sulfonic acid groups, provides a high density of active sites while remaining insoluble in the reaction medium. This unique property allows for the drastic reduction of sulfuric acid co-catalyst usage, shifting the process towards a greener chemical profile. The reaction proceeds smoothly in 1,2-dichloroethane at moderate temperatures, yielding 1-acetamidoadamantane with exceptional selectivity. By eliminating the need for complex chromatographic purification and enabling catalyst recovery through simple filtration, this novel route offers a streamlined pathway for reducing lead time for high-purity pharmaceutical intermediates, ensuring a more agile response to market demands for antiviral therapeutics.
Mechanistic Insights into PIL-1 Catalyzed Ritter Reaction
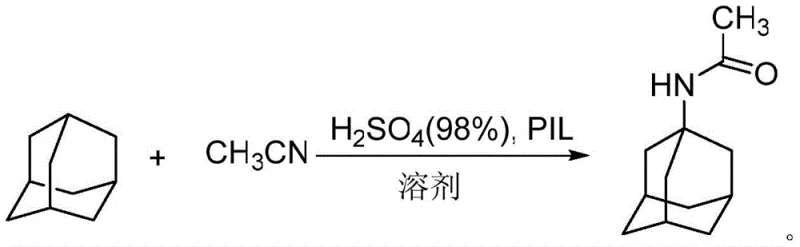
The core of this technological advancement lies in the synergistic interaction between the polyionic liquid catalyst and the reaction substrates during the Ritter-type amidation. The PIL-1 catalyst, synthesized via the copolymerization of vinylpyridine sulfonate monomers and divinylbenzene, creates a robust three-dimensional network rich in Brønsted acid sites. In the presence of a catalytic amount of sulfuric acid, the PIL-1 facilitates the protonation of acetonitrile, generating a highly reactive nitrilium ion intermediate. Simultaneously, the acidic environment promotes the formation of the 1-adamantyl carbocation from the adamantane substrate. The nucleophilic attack of the nitrile on the carbocation leads to the formation of the N-(1-adamantyl)acetamide intermediate with high regioselectivity. The heterogeneous nature of the catalyst ensures that the reaction occurs primarily at the solid-liquid interface, minimizing side reactions such as polymerization or over-oxidation that are common in homogeneous acid systems. This precise control over the reaction microenvironment is crucial for maintaining the structural integrity of the adamantane cage and achieving the high purity levels demanded by high-purity pharmaceutical intermediates specifications.
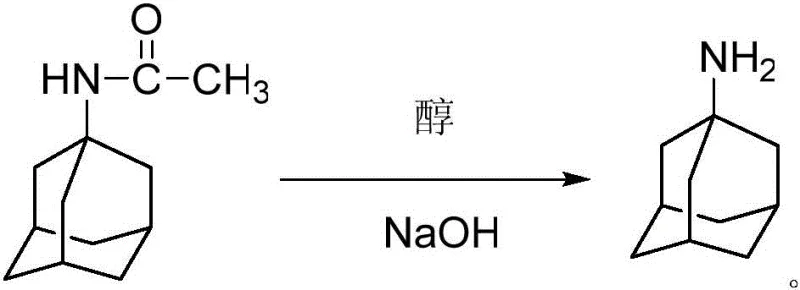
Following the amidation step, the process employs a straightforward alkaline hydrolysis to liberate the free amine. The use of ethylene glycol as a solvent in this stage is particularly advantageous due to its high boiling point and ability to dissolve both the organic intermediate and the inorganic base. Under elevated temperatures, the amide bond is cleaved efficiently by sodium hydroxide, releasing ammonia and forming the target amantadine molecule. The choice of ethylene glycol also aids in suppressing potential degradation pathways that might occur in aqueous systems at similar temperatures. Subsequent acidification with hydrochloric acid converts the free base into the stable hydrochloride salt, which precipitates readily from the organic phase. This multi-step sequence demonstrates a sophisticated understanding of solubility parameters and reaction kinetics, ensuring that impurities are effectively partitioned away from the desired product. The result is a final product with GC purity exceeding 99%, validating the efficacy of this mechanistic approach for producing commercial scale-up of complex pharmaceutical intermediates with minimal impurity profiles.
How to Synthesize Amantadine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating these high-yield results in a laboratory or pilot plant setting. The process is divided into three distinct operational phases: the catalytic amidation, the alkaline hydrolysis, and the final salt formation. Each step has been rigorously optimized regarding temperature, molar ratios, and reaction time to maximize throughput while minimizing resource consumption. The use of the PIL-1 catalyst is central to the success of the first step, where careful control of the acid-to-substrate ratio is critical. Detailed standard operating procedures for scaling this chemistry from gram to kilogram quantities are essential for ensuring batch-to-batch consistency. For technical teams looking to implement this route, the following guide summarizes the critical parameters derived from the experimental data, serving as a foundation for process validation and technology transfer.
- Amidation Reaction: React adamantane with acetonitrile in 1,2-dichloroethane using polyionic liquid PIL-1 and sulfuric acid at 60°C for 19 hours to form 1-acetamidoadamantane.
- Hydrolysis: Hydrolyze the intermediate 1-acetamidoadamantane in ethylene glycol with sodium hydroxide at 135°C for 12 hours to yield free amantadine.
- Salt Formation: Dissolve amantadine in dichloromethane and react with 5N hydrochloric acid at 50°C for 3 hours to obtain amantadine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this PIL-1 catalyzed process offers compelling advantages that directly impact the bottom line and supply chain resilience. The shift from expensive, pre-oxidized raw materials to bulk commodity chemicals like adamantane and acetonitrile significantly lowers the entry barrier for production. Furthermore, the reduction in hazardous waste generation translates to lower disposal fees and reduced regulatory burden, which are critical factors in the total cost of ownership for chemical manufacturing. The ability to recover and reuse the solid catalyst adds another layer of economic efficiency, decoupling production costs from the volatility of catalyst pricing. These factors combined create a robust framework for cost reduction in pharmaceutical intermediates manufacturing, allowing suppliers to offer more competitive pricing without compromising on quality or delivery reliability.
- Cost Reduction in Manufacturing: The elimination of expensive metal catalysts and the drastic reduction in sulfuric acid consumption directly lower the variable costs associated with each production batch. By utilizing a heterogeneous catalyst system, the need for energy-intensive distillation or chromatography steps to remove catalyst residues is removed, simplifying the unit operations required. This streamlined workflow reduces utility consumption and labor hours, contributing to substantial operational savings. Additionally, the use of cheaper starting materials like adamantane instead of adamantanol ensures that the raw material cost base remains stable and predictable, shielding the supply chain from price fluctuations in specialized reagents.
- Enhanced Supply Chain Reliability: The simplicity of the workup procedure, which relies on filtration and crystallization rather than complex extraction sequences, enhances the robustness of the manufacturing process. This reliability minimizes the risk of batch failures and ensures consistent output volumes, which is vital for maintaining uninterrupted supply to downstream API manufacturers. The recyclability of the PIL-1 catalyst further secures the supply chain by reducing dependency on external catalyst vendors. With a process that is less sensitive to minor variations in reagent quality, manufacturers can source raw materials from a broader range of qualified suppliers, thereby mitigating the risk of single-source bottlenecks.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method, such as waste minimization and catalyst reuse, align perfectly with modern environmental, social, and governance (ESG) goals. The reduced generation of acidic wastewater simplifies effluent treatment, making it easier to comply with increasingly stringent environmental regulations. This compliance advantage future-proofs the manufacturing site against regulatory changes that could otherwise halt production. Moreover, the demonstrated scalability of the reaction, evidenced by successful amplification experiments, confirms that the process can be transferred to large-scale reactors without losing efficiency, ensuring that supply can meet growing global demand for antiviral medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this method into their existing production portfolios. The answers highlight the balance between technical performance and economic viability that defines this innovative approach.
Q: How does the PIL-1 catalyst improve the environmental profile of amantadine synthesis?
A: The polyionic liquid PIL-1 acts as a solid acid catalyst that significantly reduces the required dosage of concentrated sulfuric acid compared to traditional liquid acid methods. This reduction minimizes waste acid generation and simplifies post-reaction workup through simple filtration, thereby lowering the environmental burden and disposal costs associated with hazardous waste treatment.
Q: Why is adamantane preferred over adamantanol as a starting material in this process?
A: Adamantane is a more economically viable starting material compared to adamantanol, which is often more expensive and requires prior oxidation steps. The patented method utilizes a direct Ritter-type reaction on adamantane, bypassing the need for pre-functionalized alcohols, which streamlines the supply chain and reduces overall raw material procurement costs for large-scale manufacturing.
Q: Is the polyionic liquid catalyst reusable in this synthesis?
A: Yes, the heterogeneous nature of the polyionic liquid PIL-1 allows it to be recovered via simple filtration after the reaction is complete. The patent data indicates that the catalyst maintains its structural integrity and acidic properties, enabling multiple reuse cycles which contributes to substantial long-term operational cost savings and process sustainability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amantadine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes for key pharmaceutical building blocks like amantadine. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of amantadine or its intermediates performs consistently in your downstream applications. By leveraging advanced catalytic technologies such as the PIL-1 system described in CN112939798A, we continue to push the boundaries of what is possible in fine chemical manufacturing, offering our partners a distinct competitive edge in the global marketplace.
We invite you to collaborate with us to explore how this optimized synthesis route can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this greener methodology can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why NINGBO INNO PHARMCHEM is the preferred partner for high-quality, cost-effective pharmaceutical intermediates.
