Advanced Manufacturing of 2,4'-Difluorobenzophenone: A Green Route for Flutriafol Intermediates
Advanced Manufacturing of 2,4'-Difluorobenzophenone: A Green Route for Flutriafol Intermediates
The chemical industry is constantly seeking more efficient and environmentally benign pathways for producing critical agrochemical intermediates, and the technology disclosed in patent CN113956141A represents a significant leap forward in this domain. This patent details a novel production method for 2,4'-Difluorobenzophenone, a pivotal precursor in the synthesis of the fungicide Flutriafol, addressing long-standing issues regarding high production costs and environmental pollution associated with traditional manufacturing processes. By utilizing o-fluorotoluene as a starting material and employing a sophisticated sequence of photochlorination, catalytic hydrolysis, and Friedel-Crafts acylation, the process achieves exceptional product purity while maintaining a zero-wastewater discharge profile. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for securing a reliable agrochemical intermediate supplier capable of delivering high-quality materials at a competitive operational cost.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2,4'-Difluorobenzophenone has been plagued by significant technical and economic hurdles that hinder large-scale industrial adoption. Conventional synthetic routes often rely on expensive starting materials such as o-fluorobenzoyl chloride directly, which drives up the raw material costs substantially and creates a heavy economic burden for downstream manufacturers. Furthermore, traditional methods frequently necessitate the use of complex solvent systems that are difficult to recover, leading to increased waste generation and higher disposal costs. The presence of numerous byproducts in these older processes complicates the separation and purification stages, often requiring energy-intensive recrystallization steps that still fail to guarantee the stringent purity levels required for modern pharmaceutical and agrochemical applications. These inefficiencies not only inflate the final price of the intermediate but also pose significant challenges for supply chain heads who must manage the risks associated with volatile raw material markets and strict environmental regulations.
The Novel Approach
In stark contrast to these legacy methods, the innovative approach outlined in the patent leverages a streamlined three-step synthesis starting from the readily available and cost-effective o-fluorotoluene. This new route introduces a side reaction inhibitor during the initial photochlorination stage, which effectively suppresses the formation of unwanted isomers and poly-chlorinated byproducts, thereby simplifying the downstream purification workload. A key feature of this methodology is the strategic use of fluorobenzene not just as a reactant but also as a recyclable solvent, which drastically reduces the consumption of auxiliary chemicals and minimizes solvent waste. Moreover, the process incorporates a closed-loop water management system where washing water is recycled for the quenching reaction, ensuring that the entire production cycle generates no wastewater. This holistic design not only lowers the cost reduction in fungicide intermediate manufacturing but also aligns perfectly with global sustainability goals, making it an attractive option for companies aiming to green their supply chains.
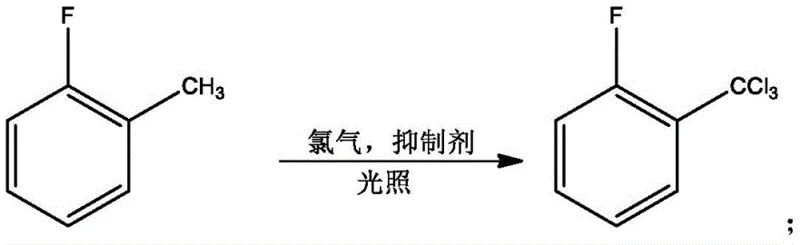
Mechanistic Insights into AlCl3/LiCl Catalyzed Friedel-Crafts Acylation
The heart of this synthesis lies in the final acylation step, where o-fluorobenzoyl chloride reacts with fluorobenzene under the influence of a specialized mixed catalyst system comprising anhydrous aluminum trichloride and lithium chloride. The addition of lithium chloride to the traditional Lewis acid catalyst is a critical innovation that enhances the catalytic activity and selectivity of the reaction, leading to a marked increase in yield and a significant reduction in the formation of the 2,2'-difluorobenzophenone isomer. Mechanistically, the Lewis acid activates the carbonyl group of the acid chloride, generating a highly reactive acylium ion that attacks the electron-rich aromatic ring of the fluorobenzene. The presence of the inhibitor in the earlier chlorination steps ensures that the acid chloride precursor is of high quality, which further propagates purity through to the final coupling reaction. This precise control over the reaction environment allows for the production of high-purity 2,4'-Difluorobenzophenone with minimal impurity profiles, satisfying the rigorous demands of R&D teams focused on impurity control.
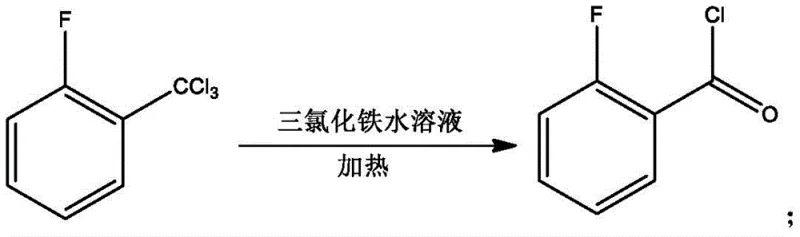
Furthermore, the purification strategy employed in this patent is meticulously designed to remove trace impurities that could affect the performance of the final agrochemical product. Following the acylation reaction, the crude product undergoes a combination of vacuum rectification and melt crystallization, a dual-purification technique that is highly effective at separating compounds with similar boiling points or solubilities. The melt crystallization step, in particular, exploits the differences in freezing points between the target 2,4'-isomer and the undesired 2,2'-isomer, allowing for the isolation of the target molecule with a purity content reaching up to 99.97%. This level of purity is crucial for the subsequent synthesis of Flutriafol, as impurities can interfere with the biological activity of the fungicide or cause stability issues in the final formulation. By integrating these advanced purification techniques with a robust catalytic system, the process ensures consistent quality and reliability, which are paramount for commercial scale-up of complex agrochemical intermediates.
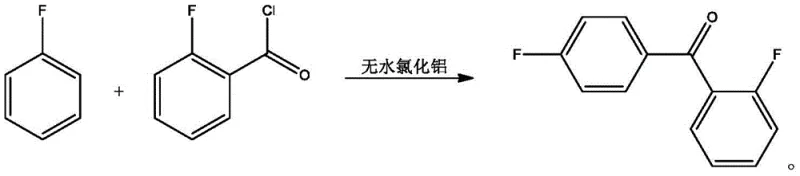
How to Synthesize 2,4'-Difluorobenzophenone Efficiently
The synthesis protocol described in the patent offers a clear and reproducible pathway for manufacturing this valuable intermediate, beginning with the controlled photochlorination of o-fluorotoluene to generate the necessary acid chloride precursor. The process requires careful attention to temperature control and the timing of inhibitor addition to maximize the yield of the desired chlorinated species while minimizing side reactions. Once the precursor is prepared via hydrolysis, the final coupling reaction is conducted under mild conditions using the mixed catalyst system, followed by a straightforward workup procedure that facilitates solvent recovery and waste minimization. For technical teams looking to implement this route, the detailed standardized synthesis steps provided below outline the specific parameters and operational sequences required to achieve optimal results in a pilot or production setting.
- Perform photochlorination of o-fluorotoluene under illumination with a side reaction inhibitor to obtain chlorinated intermediates, followed by rectification.
- Hydrolyze the chlorinated mixture using an aqueous ferric trichloride solution under heating to generate o-fluorobenzoyl chloride.
- Conduct Friedel-Crafts acylation with fluorobenzene using an AlCl3/LiCl catalyst, followed by quenching, washing, and melt crystallization for purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly address the pain points of procurement managers and supply chain leaders in the fine chemical sector. The shift from expensive, specialized starting materials to commodity chemicals like o-fluorotoluene fundamentally alters the cost structure of the production process, enabling significant savings on raw material procurement without compromising on quality. Additionally, the ability to recycle fluorobenzene solvent and reuse wash water for quenching creates a circular economy within the plant, drastically reducing the volume of hazardous waste that requires treatment and disposal. These operational efficiencies translate into a more resilient supply chain that is less susceptible to fluctuations in raw material prices and regulatory changes regarding environmental compliance. For organizations seeking a reliable agrochemical intermediate supplier, adopting this technology means securing a source of high-purity materials that is both economically viable and environmentally sustainable.
- Cost Reduction in Manufacturing: The elimination of complex solvent systems and the use of inexpensive, widely available raw materials significantly lower the overall production costs. By recycling the fluorobenzene solvent and repurposing the aluminum trichloride solution generated during quenching for water treatment applications, the process minimizes waste disposal fees and maximizes resource utilization. This lean manufacturing approach ensures that the final product remains cost-competitive in the global market, providing a distinct advantage for buyers looking to optimize their supply chain expenses.
- Enhanced Supply Chain Reliability: Utilizing o-fluorotoluene as the primary feedstock ensures a stable and continuous supply of raw materials, as it is a commodity chemical produced on a large scale by multiple vendors. The simplicity of the synthetic route, which avoids sensitive reagents or extreme reaction conditions, reduces the risk of production delays caused by equipment failures or safety incidents. This robustness allows suppliers to maintain consistent delivery schedules and meet the demanding lead times required by downstream agrochemical manufacturers, thereby strengthening the overall reliability of the supply network.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, featuring unit operations such as rectification and crystallization that are easily adapted to large-scale production facilities. The zero-wastewater discharge capability is a major compliance advantage, as it eliminates the need for complex effluent treatment plants and ensures adherence to increasingly strict environmental regulations. This eco-friendly profile not only reduces regulatory risk but also enhances the corporate social responsibility standing of the manufacturer, making it a preferred partner for multinational corporations with stringent sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions and answers address common technical and commercial inquiries regarding the production of 2,4'-Difluorobenzophenone using this patented method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the technology's capabilities and limitations. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their existing supply chains or product portfolios.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method utilizes cheap and easily available raw materials like o-fluorotoluene, features a simple synthetic route, and achieves zero wastewater discharge by recycling wash water for quenching.
Q: How is high purity achieved in the final 2,4'-Difluorobenzophenone product?
A: High purity up to 99.97% is attained through the use of a mixed AlCl3/LiCl catalyst which reduces byproducts, combined with a final purification step involving vacuum rectification and melt crystallization.
Q: Is the solvent used in this process environmentally sustainable?
A: Yes, fluorobenzene is used as both a reactant and a solvent, and it can be recovered and recycled after the reaction, significantly reducing resource consumption and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4'-Difluorobenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of agrochemical formulations, and we are committed to delivering products that meet the highest standards of purity and consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the development phase or full-scale manufacturing. We operate stringent purity specifications and maintain rigorous QC labs to verify that every batch of 2,4'-Difluorobenzophenone conforms to the exacting needs of your synthesis processes, giving you confidence in the reliability of your supply chain.
We invite you to contact our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our optimized production route. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the global fine chemical market.
