Scalable Synthesis of 1-Chloro-3,5-di-O-acyl-2-deoxy-L-ribofuranoside Derivatives for Antiviral APIs
The pharmaceutical industry's relentless pursuit of potent antiviral agents has placed L-series nucleosides at the forefront of medicinal chemistry research, driven by their superior biological activity and reduced toxicity profiles compared to their D-series counterparts. Patent CN1878782A introduces a groundbreaking industrial methodology for the preparation of 1-chloro-3,5-di-O-acyl-2-deoxy-L-ribofuranoside derivatives, which serve as critical precursors in the synthesis of these high-value therapeutic compounds. This invention addresses the longstanding bottlenecks in carbohydrate chemistry by establishing a robust, scalable route that begins with the readily available 2-deoxy-D-galactose rather than the prohibitively expensive 2-deoxy-L-ribose. By leveraging a strategic sequence of glycosylation, oxidative cleavage, and stereoselective reduction, the process achieves high yields while maintaining stringent control over stereochemical integrity. For R&D directors and process chemists, this patent represents a paradigm shift from laboratory-scale curiosity to viable commercial manufacturing, offering a clear pathway to secure the supply chain for next-generation antiviral drugs.
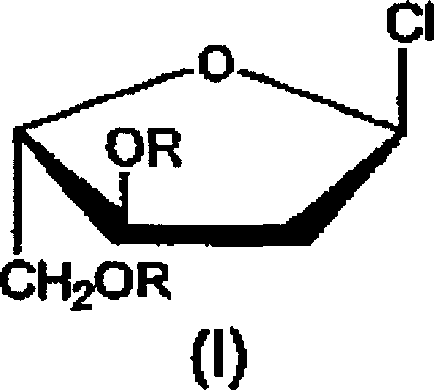
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described by W. Zhang et al., have historically relied on synthetic routes that are fundamentally unsuited for large-scale industrial application due to severe safety and economic constraints. These conventional processes typically initiate with 2-deoxy-L-ribose, a starting material that is not only costly but also difficult to source in the bulk quantities required for commercial API production. Furthermore, the synthetic steps involved often necessitate the use of pyridine in large excesses, creating significant challenges regarding worker safety, environmental compliance, and solvent recovery costs. The reliance on silica gel column chromatography for purification in multiple steps creates a throughput bottleneck that makes scaling to metric ton levels practically impossible without exorbitant capital expenditure. Additionally, the final chlorination steps in older methods frequently utilize diethyl ether, a solvent with extreme flammability risks that complicate plant safety protocols and increase insurance liabilities for manufacturing facilities.
The Novel Approach
In stark contrast, the process disclosed in CN1878782A engineers a solution that systematically dismantles these barriers by reimagining the synthetic trajectory from the ground up. By switching the starting material to 2-deoxy-D-galactose, the method taps into a much more abundant and economically favorable feedstock, immediately lowering the baseline cost of goods sold. The reaction design cleverly avoids the use of pyridine, opting instead for triethylamine in toluene, a solvent system that is far more amenable to industrial distillation and recycling. Crucially, the invention eliminates the need for chromatographic purification in the early stages, replacing it with efficient crystallization and precipitation techniques that are standard in bulk chemical manufacturing. The final chlorination is conducted in toluene at controlled low temperatures, removing the fire hazard associated with ether and allowing for direct isolation of the product as a solid, thereby streamlining the downstream processing workflow significantly.
Mechanistic Insights into Stereoselective Inversion and Functionalization
The core chemical innovation of this patent lies in its elegant manipulation of stereochemistry to convert a D-sugar into an L-sugar derivative through a precise oxidative-reductive sequence. The process begins with the acid-catalyzed glycosylation of 2-deoxy-D-galactose in methanol, where strict temperature control below 3°C is maintained to favor the formation of the furanoside ring over the pyranoside isomer, ensuring high regioselectivity from the outset. Following this, the critical transformation occurs via oxidation with sodium metaperiodate, which selectively cleaves the vicinal diol to generate an aldehyde intermediate. This step is highly exothermic and requires careful thermal management below 10°C to prevent degradation or side reactions that could compromise the purity of the intermediate. The subsequent reduction with sodium borohydride is the stereochemical pivot point, where the hydride attack is directed to establish the L-ribose configuration, effectively inverting the chirality relative to the starting galactose scaffold.
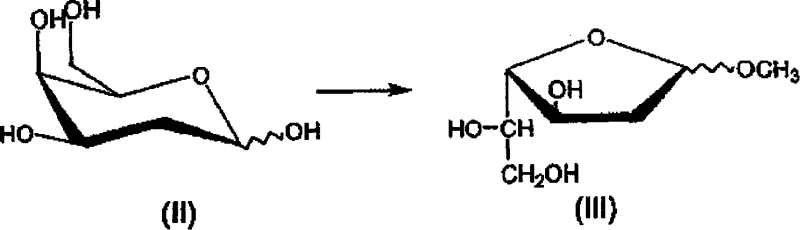
Following the establishment of the correct L-configuration, the molecule undergoes selective acylation at the 3 and 5 positions using acyl chlorides in the presence of a tertiary amine base. The choice of toluene as the solvent here is strategic, as it facilitates the solubility of the intermediates while allowing for easy removal of the amine hydrochloride salts formed during the reaction. The final step involves the introduction of the chlorine atom at the anomeric position using gaseous hydrogen chloride. This reaction is performed in the presence of acetyl chloride, which serves a dual purpose: it acts as a dehydrating agent to scavenge the methanol byproduct by converting it into methyl acetate, thus driving the equilibrium towards the desired 1-chloro product. Throughout this entire sequence, the control of impurities is managed through physical means such as temperature regulation and solvent selection rather than relying on post-reaction purification, which is a hallmark of a mature, scalable chemical process designed for high-purity output.
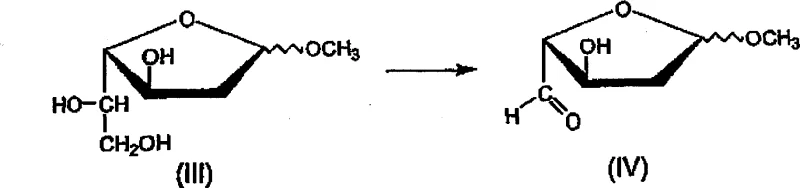
How to Synthesize 1-Chloro-3,5-di-O-acyl-2-deoxy-L-ribofuranoside Efficiently
The synthesis of these critical antiviral intermediates requires a disciplined approach to reaction conditions to ensure both high yield and stereochemical purity. The patented procedure outlines a logical progression where each step builds upon the purity of the previous one, minimizing the carryover of impurities that could affect the final API quality. Operators must pay close attention to the exothermic nature of the oxidation and reduction steps, utilizing external cooling and controlled addition rates to maintain the internal temperatures within the narrow windows specified in the patent documentation. The workup procedures, involving salt removal and solvent exchanges, are designed to be robust and forgiving, allowing for consistent results even when scaling up reactor volumes. For a detailed breakdown of the specific molar ratios, addition times, and isolation techniques, please refer to the standardized synthesis guide below.
- Glycosylation of 2-deoxy-D-galactose with methanol and acid catalyst at low temperature (<3°C) to form 1-O-methyl-2-deoxy-D-galactofuranoside.
- Oxidative cleavage using sodium metaperiodate followed by stereoselective reduction with sodium borohydride to invert configuration to L-ribose.
- Acylation with acyl chloride in toluene/triethylamine, followed by chlorination with gaseous HCl to yield the final 1-chloro derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented process translates directly into tangible operational efficiencies and risk mitigation strategies that strengthen the overall supply network. The shift away from expensive and scarce starting materials like 2-deoxy-L-ribose to commodity chemicals like 2-deoxy-D-galactose drastically reduces the volatility of raw material costs, providing greater predictability in budgeting and long-term planning. Furthermore, the elimination of silica gel chromatography removes a major bottleneck in production throughput, allowing manufacturing facilities to produce larger batches in shorter cycle times without the need for specialized, low-capacity purification equipment. This improvement in process intensity means that the same physical plant infrastructure can support significantly higher output volumes, effectively increasing asset utilization rates without requiring heavy capital investment in new hardware.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the substitution of high-cost reagents with inexpensive alternatives and the simplification of unit operations. By avoiding the use of pyridine and diethyl ether, the process reduces the costs associated with hazardous waste disposal and solvent recovery, as toluene and triethylamine are easier to recycle using standard distillation columns. The ability to isolate intermediates through crystallization rather than chromatography significantly lowers the consumption of consumables like silica gel and elution solvents, which are major cost drivers in fine chemical synthesis. Additionally, the high yields reported in the patent examples indicate minimal material loss, ensuring that the maximum amount of input mass is converted into valuable product, thereby optimizing the overall material balance and reducing the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: Relying on 2-deoxy-D-galactose as the primary feedstock enhances supply security because this chemical is produced on a much larger industrial scale than its L-isomer counterpart, reducing the risk of shortages or price spikes due to supply constraints. The robustness of the synthetic route, characterized by its tolerance to standard industrial conditions and lack of sensitive purification steps, ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding production delays. Moreover, the use of common solvents like toluene and methanol ensures that solvent availability will not become a limiting factor, as these are staple chemicals in the global chemical trade with stable and diversified supply sources.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process offers a cleaner profile that aligns with modern green chemistry principles and stringent regulatory standards. The removal of highly flammable ethers and toxic pyridines simplifies the safety case for the manufacturing plant, potentially lowering insurance premiums and reducing the burden on environmental health and safety teams. The process generates less hazardous waste volume due to the absence of chromatography fractions, making waste treatment more straightforward and cost-effective. This scalability ensures that the transition from pilot plant to full commercial production can be achieved with minimal re-engineering, allowing for rapid response to market demand surges for antiviral medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, drawing directly from the experimental data and claims within the patent specification. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this chemistry into their existing manufacturing portfolios. The answers provided reflect the specific advantages and operational parameters defined in CN1878782A, ensuring accuracy and relevance for decision-makers.
Q: Why is 2-deoxy-D-galactose preferred over 2-deoxy-L-ribose as a starting material?
A: 2-deoxy-D-galactose is significantly more commercially available and cost-effective than 2-deoxy-L-ribose. The patented process utilizes an oxidative cleavage and reduction strategy to invert the stereochemistry from the D-series to the required L-series configuration, bypassing the need for expensive chiral starting materials.
Q: How does this process improve safety compared to conventional methods?
A: Conventional methods often rely on highly toxic solvents like pyridine and highly flammable solvents like diethyl ether. This novel route utilizes toluene and triethylamine, which are easier to handle and recover on an industrial scale, and eliminates the need for hazardous ether precipitation steps.
Q: Is silica gel chromatography required for purification in this method?
A: No. A major advantage of this invention is the elimination of silica gel column chromatography in the early stages. Purification is achieved through controlled crystallization, precipitation, and solvent exchanges, making the process highly suitable for multi-kilogram or ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Chloro-3,5-di-O-acyl-2-deoxy-L-ribofuranoside Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antiviral therapies depends on a secure and high-quality supply of critical intermediates like 1-chloro-3,5-di-O-acyl-2-deoxy-L-ribofuranoside derivatives. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the exacting standards required for API synthesis, providing you with the confidence needed to advance your drug development programs. Our commitment to technical excellence means we can adapt the patented conditions to fit your specific facility requirements while maintaining the high yields and selectivity demonstrated in the literature.
We invite you to engage with our technical procurement team to discuss how we can support your specific project needs with a Customized Cost-Saving Analysis tailored to your volume requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that will help you optimize your supply chain strategy. Contact us today to request samples or to schedule a technical discussion on how we can deliver high-purity pharmaceutical intermediates that drive your success in the competitive antiviral market.
