Industrial Scale-Up of Ziprasidone: A Technical Analysis of Solvent Engineering and Purity Control
Industrial Scale-Up of Ziprasidone: A Technical Analysis of Solvent Engineering and Purity Control
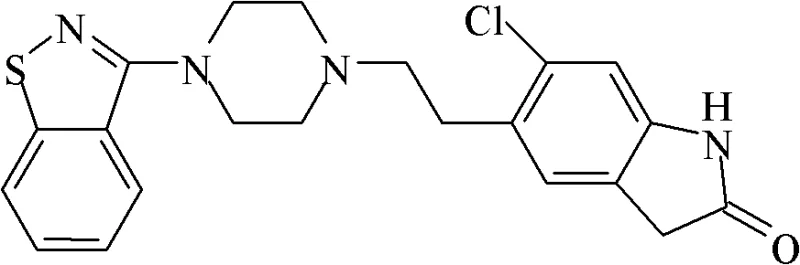
The pharmaceutical industry constantly seeks robust synthetic routes for atypical antipsychotic agents, and Ziprasidone stands out as a critical molecule in this therapeutic class. The patent CN102250083B introduces a transformative methodology for the preparation of Ziprasidone that addresses longstanding inefficiencies in prior art. This technical disclosure outlines a process utilizing a mixed solvent system comprising a water-soluble polar nonprotonic solvent and an aqueous solution of an inorganic base. By operating at moderate temperatures between 60 and 70 degrees Celsius, the reaction achieves completion within a concise timeframe of 3 to 6 hours. This represents a paradigm shift from traditional methods that often suffered from prolonged reaction times and cumbersome purification requirements. The strategic selection of solvents not only enhances the solubility of the key intermediates but also optimizes the acid-binding capacity of the base, leading to superior reaction kinetics. For R&D directors and process chemists, this patent offers a blueprint for achieving high yields and exceptional purity profiles essential for regulatory compliance. As a reliable API intermediate supplier, understanding these mechanistic nuances is vital for ensuring supply chain continuity and product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Ziprasidone have been plagued by significant operational drawbacks that hindered their industrial viability. For instance, Patent US431031 describes a method involving reflux reactions for up to 40 hours, which is energetically inefficient and time-consuming. Furthermore, this legacy technique necessitates the use of column chromatography to isolate the pure Ziprasidone base, a step that is notoriously difficult to scale and results in low yields of approximately 20 percent. Other methods, such as those disclosed in Patent US5388846 and WO2005040160A2, rely heavily on aqueous phases or alcohol-water mixtures. While these attempts aimed to improve environmental safety, they often resulted in heterogeneous systems due to the poor solubility of the chloroacetyl indolone precursor. This heterogeneity leads to incomplete reactions, requiring extensive post-reaction processing such as recrystallization from tetrahydrofuran or ethanol to achieve acceptable purity levels. Consequently, these conventional routes increase the overall production cost and extend the manufacturing lead time, creating bottlenecks for procurement managers seeking cost-effective sourcing strategies.
The Novel Approach
The methodology presented in CN102250083B overcomes these historical barriers through the ingenious application of a homogeneous mixed solvent system. By dissolving the reactants, specifically 5-(2-chloroacetyl)-6-chloro-1,3-dihydro-indol-2-(2H)-one and 3-piperazinyl-1,2-benzisothiazole hydrochloride, in a blend of polar aprotic solvents like N,N-dimethylformamide (DMF) or acetone with an aqueous inorganic base, the process ensures a single-phase reaction environment. This homogeneity is crucial for maximizing molecular collisions and driving the nucleophilic substitution to completion efficiently. The reaction conditions are remarkably mild, requiring heating only to 60 to 70 degrees Celsius, which reduces energy consumption and thermal stress on the equipment. Post-reaction workup is simplified to a straightforward precipitation by adding purified water, followed by filtration and drying. This eliminates the need for complex extraction or chromatographic purification steps, directly yielding a product with purity exceeding 99 percent. Such streamlining is a game-changer for commercial scale-up of complex pharmaceutical intermediates, offering a clear path to reduced operational expenditures.

Mechanistic Insights into Nucleophilic Substitution in Mixed Solvents
The core chemical transformation in this synthesis is a nucleophilic substitution where the piperazine nitrogen attacks the chloroacetyl group of the indolone derivative. In traditional aqueous systems, the poor solubility of the organic halide limits the availability of the electrophile, slowing down the reaction rate significantly. However, the introduction of a water-miscible polar aprotic solvent fundamentally alters the solvation shell around the reactants. These solvents, characterized by their high dielectric constants and ability to solvate cations without hydrogen bonding to anions, leave the nucleophilic nitrogen of the piperazine ring "naked" and highly reactive. This enhancement in nucleophilicity allows the reaction to proceed rapidly even at moderate temperatures. Furthermore, the presence of the inorganic base, such as potassium carbonate or sodium carbonate, serves to neutralize the hydrochloric acid salt of the piperazine derivative in situ, generating the free base necessary for the attack. The synergy between the solvent's solubilizing power and the base's deprotonation efficiency creates an optimal kinetic environment that minimizes side reactions and byproduct formation.
Impurity control is another critical aspect where this mechanism excels. In heterogeneous systems, localized high concentrations of base or incomplete mixing can lead to hydrolysis of the chloroacetyl group or degradation of the sensitive indolone ring. The homogeneous nature of the mixed solvent system ensures uniform distribution of heat and reagents throughout the reaction vessel. This uniformity prevents hot spots that could trigger decomposition pathways. Additionally, the specific temperature range of 60 to 70 degrees Celsius is carefully selected to be high enough to overcome the activation energy barrier for the substitution but low enough to prevent thermal degradation of the product. The result is a clean reaction profile where the primary impurity is unreacted starting material, which is easily removed during the water precipitation step due to solubility differences. This inherent selectivity reduces the burden on downstream purification processes, ensuring that the final API meets stringent pharmacopeial standards with minimal additional processing.
How to Synthesize Ziprasidone Efficiently
Implementing this synthesis route requires precise control over stoichiometry and solvent ratios to maximize efficiency. The patent specifies a molar ratio of the indolone derivative to the piperazine salt ranging from 1:1 to 1:1.3, with a preferred range of 1:1.05 to 1:1.15 to ensure complete consumption of the limiting reagent. The volume ratio of the polar aprotic solvent to the aqueous base solution is maintained between 7:3 and 8:2, providing the ideal balance between solubility and base strength. Detailed standardized synthesis steps see the guide below.
- Dissolve 5-(2-chloroacetyl)-6-chloro-1,3-dihydro-indol-2-(2H)-one and 3-piperazinyl-1,2-benzisothiazole hydrochloride in a mixed system of water-soluble polar nonprotonic solvent and aqueous inorganic base.
- Heat the reaction mixture to a temperature between 60 and 70 degrees Celsius and maintain insulation for 3 to 6 hours to ensure complete conversion.
- Upon completion, add purified water to precipitate the product, followed by suction filtration, washing, and drying to obtain high-purity Ziprasidone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible economic and logistical benefits that extend beyond simple yield improvements. The elimination of column chromatography, a resource-intensive and solvent-heavy process, translates directly into significant cost reduction in pharmaceutical manufacturing. By removing this step, manufacturers save on the costs of silica gel, large volumes of elution solvents, and the associated waste disposal fees. Furthermore, the shortened reaction time from days to mere hours increases the throughput of existing reactor infrastructure, allowing for more batches to be produced within the same timeframe. This efficiency gain is crucial for meeting tight delivery schedules and maintaining inventory levels without the need for excessive capital investment in new equipment. The use of common, commercially available solvents like acetone and DMF also mitigates supply chain risks associated with specialty reagents, ensuring a stable and reliable supply of raw materials.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive purification techniques like column chromatography, which are both labor-intensive and solvent-consuming. By achieving high purity directly through precipitation, the method drastically reduces the consumption of organic solvents and adsorbents. This simplification of the workflow lowers the overall variable cost per kilogram of the final product. Additionally, the high yield observed in the examples, consistently above 89 percent, means less raw material is wasted, further enhancing the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: The reliance on standard industrial chemicals such as potassium carbonate, sodium carbonate, and common polar solvents ensures that the supply chain is robust and less susceptible to disruptions. Unlike processes requiring exotic catalysts or hard-to-source reagents, this method utilizes commodities that are readily available from multiple global suppliers. This diversification of the supply base reduces the risk of single-source dependency and price volatility. Moreover, the simplicity of the operation reduces the likelihood of batch failures due to operator error, contributing to a more predictable and consistent output schedule for downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of hazardous heavy metal catalysts make this process inherently safer and easier to scale from pilot plant to commercial production. The reduced solvent usage and the ability to recover and recycle the aqueous mother liquor contribute to a smaller environmental footprint. This aligns with increasingly stringent global environmental regulations regarding volatile organic compound (VOC) emissions and waste generation. Companies adopting this green chemistry approach can benefit from lower compliance costs and an improved sustainability profile, which is becoming a key differentiator in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ziprasidone synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. They are intended to provide clarity for technical teams evaluating the feasibility of this route for their specific manufacturing contexts. Understanding these details is essential for making informed decisions about process adoption and technology transfer.
Q: What are the primary advantages of the mixed solvent system in Ziprasidone synthesis?
A: The mixed solvent system significantly increases the solubility of reactants compared to pure aqueous systems, enhances the acid-binding effect of the inorganic base, and drastically shortens reaction times from over 30 hours to just 3-6 hours.
Q: How does this method improve product purity compared to prior art?
A: By optimizing the solvent polarity and reaction temperature, this method achieves HPLC purity levels exceeding 99.0% directly after filtration, eliminating the need for complex column chromatography purification steps required in older patents.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial solvents like DMF or acetone and mild temperatures (60-70°C), making it highly scalable, safe, and economically viable for multi-ton production without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ziprasidone Supplier
The technical potential of this mixed-solvent synthesis route underscores the importance of partnering with a CDMO that possesses deep expertise in process optimization and scale-up. NINGBO INNO PHARMCHEM stands ready to leverage this advanced methodology to deliver high-quality Ziprasidone and its intermediates to the global market. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch meets the highest international standards. Our commitment to quality assurance ensures that our clients receive products that are fully compliant with regulatory requirements, minimizing the risk of delays in drug development and approval.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains and reduce manufacturing costs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how this innovative synthesis method can be integrated into your production portfolio. Let us help you navigate the complexities of API manufacturing with solutions that are both scientifically robust and commercially sound.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
