Revolutionizing NH-Sulfoximine Production: Electrochemical Synthesis for Commercial Scale
Introduction to Next-Generation Sulfoximine Synthesis
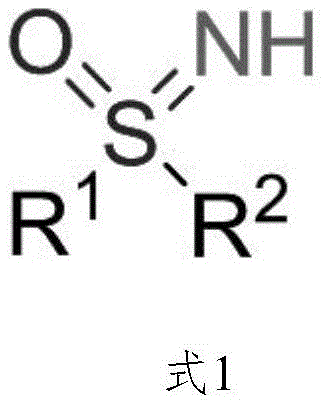
The pharmaceutical and agrochemical industries are constantly seeking more efficient and sustainable pathways to access complex nitrogen-sulfur heterocycles, particularly NH-sulfoximines, which serve as critical bioisosteres in modern drug design. Patent CN113402434A introduces a groundbreaking electrochemical methodology that fundamentally shifts the paradigm from stoichiometric chemical oxidation to catalytic electron-driven synthesis. This innovation addresses the long-standing challenges associated with the preparation of NH-sulfoximine scaffolds, which are pivotal components in high-value therapeutic agents such as the cancer treatment candidate BAY 1000394 and various enzyme inhibitors. By leveraging electricity as a traceless oxidant, this technology not only enhances the atom economy of the reaction but also aligns perfectly with the global push towards greener manufacturing processes in the fine chemical sector.
The core breakthrough lies in the ability to generate high-valent iodine species in situ using a catalytic amount of aryl iodide and a constant electric current, rather than relying on pre-formed, expensive hypervalent iodine reagents. This approach drastically simplifies the reaction setup and workup procedures, making it an attractive option for both laboratory research and industrial scale-up. For R&D directors and process chemists, this represents a significant opportunity to streamline the synthesis of sulfoximine-containing drug candidates, reducing both the environmental footprint and the overall cost of goods sold (COGS). The versatility of this method allows for the functionalization of a wide range of thioether substrates, expanding the chemical space available for medicinal chemistry exploration.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct synthesis of NH-sulfoximines from sulfides has been plagued by significant economic and environmental inefficiencies. Traditional protocols typically mandate the use of stoichiometric or even excess amounts of hypervalent iodine reagents, such as diacetoxyiodobenzene (PIDA) or bis(trifluoroacetoxy)iodobenzene (PIFA), to drive the oxidation and nitrogen transfer steps. These reagents are not only costly to procure in bulk quantities but also generate substantial amounts of iodobenzene waste, which complicates downstream purification and disposal. Furthermore, the solubility profiles of these high-valent iodine species often necessitate the use of large volumes of organic solvents, leading to increased operational expenses and safety hazards associated with solvent handling and recovery.
From a supply chain perspective, the reliance on stoichiometric oxidants creates a bottleneck, as the procurement of high-purity hypervalent iodine compounds can be subject to market volatility and lead time fluctuations. Additionally, the separation of the desired sulfoximine product from the reduced iodine byproducts often requires rigorous chromatographic purification, which is difficult to translate into continuous manufacturing processes. The accumulation of iodine-containing waste streams also poses regulatory challenges regarding environmental compliance, forcing manufacturers to invest heavily in waste treatment infrastructure. Consequently, these conventional methods limit the commercial viability of sulfoximine-based APIs, particularly for indications where cost sensitivity is high.
The Novel Approach
In stark contrast, the electrochemical strategy detailed in the patent data offers a streamlined, catalytic alternative that circumvents the need for stoichiometric chemical oxidants. By employing a simple undivided cell setup with a graphite anode and a platinum cathode, the system utilizes electrons to oxidize a catalytic amount of aryl iodide (e.g., p-iodoanisole) directly within the reaction mixture. This in situ generation of the active hypervalent iodine species ensures that the oxidant is produced only as needed, minimizing side reactions and maximizing resource efficiency. The reaction proceeds under mild conditions, typically at ambient temperatures between 20-30°C, which preserves the integrity of sensitive functional groups on the substrate.
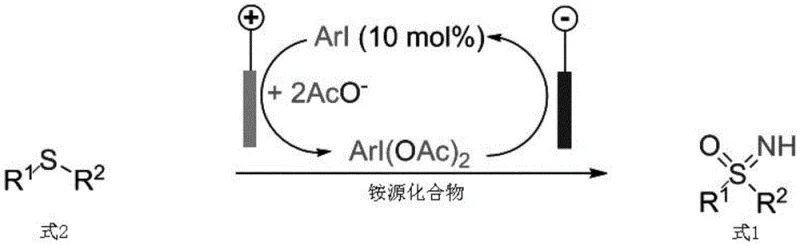
The operational simplicity of this novel approach is a major advantage for process development teams. The reaction utilizes readily available ammonium salts, such as ammonium acetate, as the nitrogen source, avoiding the handling of hazardous ammonia gas or unstable amine derivatives. The use of mixed solvent systems, such as hexafluoroisopropanol (HFIP) and methanol, facilitates the solubility of both organic substrates and ionic electrolytes, ensuring homogeneous reaction kinetics. Most importantly, the elimination of bulk hypervalent iodine reagents translates to a cleaner reaction profile, reducing the burden on purification units and enabling higher throughput. This method effectively decouples the synthesis of high-value sulfoximines from the constraints of traditional stoichiometric oxidation, paving the way for more sustainable and cost-effective manufacturing.
Mechanistic Insights into Electrochemical Iodine Catalysis
The mechanistic elegance of this transformation relies on a sophisticated catalytic cycle driven by anodic oxidation. Initially, the low-valent aryl iodide catalyst undergoes a two-electron oxidation at the anode surface to generate a highly reactive cationic iodine(III) species. In the presence of acetate ions from the electrolyte or solvent, this species is stabilized as a hypervalent iodine(III) diacetate intermediate, which serves as the active oxidant in the cycle. This in situ formation mimics the reactivity of externally added PIDA but avoids the associated logistical and waste issues. The electrochemical potential is carefully controlled to ensure selective oxidation of the iodine catalyst without over-oxidizing the organic substrate or the solvent system.
Subsequently, the generated iodine(III) species reacts with the ammonium source to form a key nitrogen-iodine intermediate, which acts as a nitrene precursor. This electrophilic nitrogen species is then captured by the nucleophilic sulfur atom of the thioether substrate, forming a sulfilimine intermediate. The final oxidation step, mediated by another equivalent of the electrochemically generated iodine(III) species or via nucleophilic attack by solvent molecules, converts the sulfilimine into the target NH-sulfoximine. Throughout this cycle, the reduced iodine(I) species is regenerated at the anode, closing the catalytic loop. This mechanism ensures high chemoselectivity, as the oxidation power is tuned specifically for the sulfur-nitrogen bond formation, minimizing the formation of sulfone or other over-oxidized impurities.
How to Synthesize NH-Sulfoximine Compounds Efficiently
Implementing this electrochemical protocol requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of a homogeneous solution containing the thioether substrate, ammonium acetate, a supporting electrolyte like tetrabutylammonium acetate, and a catalytic loading of p-iodoanisole in a mixed solvent of HFIP and methanol. The reaction vessel is equipped with a graphite anode and a platinum cathode, connected to a constant current power supply set to deliver 7-10 mA. Maintaining the temperature between 20-30°C is crucial to prevent thermal degradation of the intermediates while ensuring sufficient reaction kinetics. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by adding thioether substrate, ammonium acetate, electrolyte (e.g., tetrabutylammonium acetate), and catalytic aryl iodide (e.g., p-iodoanisole) into a solvent system of hexafluoroisopropanol and methanol.
- Insert graphite anode and platinum cathode into the solution, then apply a constant current of 7-10 mA while stirring at 20-30°C for 5-8 hours to facilitate anodic oxidation.
- Upon completion, quench the reaction with sodium thiosulfate, extract with dichloromethane, dry over sodium sulfate, and purify the crude product via column chromatography to obtain high-purity NH-sulfoximine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical synthesis route offers compelling strategic advantages that extend beyond mere technical feasibility. The most significant impact is observed in the reduction of raw material costs, driven by the substitution of expensive stoichiometric oxidants with inexpensive electricity and catalytic aryl iodides. This shift not only lowers the direct material cost per kilogram of the product but also stabilizes the supply chain by reducing dependency on specialized reagent suppliers who may have limited production capacity. Furthermore, the simplified workup procedure reduces the consumption of solvents and silica gel for purification, contributing to substantial operational expenditure (OPEX) savings.
- Cost Reduction in Manufacturing: The elimination of stoichiometric hypervalent iodine reagents represents a major cost saving opportunity, as these chemicals are among the most expensive reagents in organic synthesis. By replacing them with a catalytic system powered by electricity, the process drastically reduces the cost of goods sold (COGS). Additionally, the mild reaction conditions minimize energy consumption related to heating or cooling, further enhancing the economic efficiency of the manufacturing process. The reduction in waste disposal costs, due to the absence of heavy iodine byproducts, adds another layer of financial benefit to the overall production economics.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as thioethers, ammonium acetate, and common solvents ensures a robust and resilient supply chain. Unlike specialized oxidants that may face supply disruptions, these starting materials are widely available from multiple global vendors, mitigating the risk of production stoppages. The scalability of electrochemical reactors also allows for flexible production scheduling, enabling manufacturers to respond quickly to fluctuating market demands without the need for extensive inventory buffering. This reliability is critical for maintaining continuity in the supply of key pharmaceutical intermediates.
- Scalability and Environmental Compliance: Electrochemical synthesis is inherently scalable, particularly when adapted to continuous flow reactors, which offer superior mass and heat transfer characteristics compared to batch processes. This scalability facilitates the transition from pilot plant to commercial production with minimal process re-engineering. Moreover, the green nature of the process, characterized by the use of electrons as reagents and the generation of minimal waste, aligns with stringent environmental regulations and corporate sustainability goals. This compliance reduces the regulatory burden and enhances the brand reputation of the manufacturer as a responsible supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of NH-sulfoximines, based on the detailed specifications provided in the patent literature. These insights are designed to clarify the operational feasibility and strategic value of adopting this technology for large-scale production. Understanding these nuances is essential for stakeholders evaluating the integration of this method into their existing manufacturing portfolios.
Q: What are the primary advantages of this electrochemical method over traditional oxidation?
A: Unlike traditional methods requiring stoichiometric amounts of expensive hypervalent iodine reagents (like PIDA), this patent utilizes electricity as the sole oxidant with only catalytic amounts of aryl iodide. This significantly reduces reagent costs and minimizes hazardous waste generation.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the process operates under mild conditions (20-30°C) and uses readily available starting materials like thioethers. The electrochemical nature allows for potential scalability through flow chemistry setups, offering a robust pathway for commercial production.
Q: What is the typical purity and yield achievable with this protocol?
A: According to the patent data, the method consistently achieves yields exceeding 80%, with nuclear magnetic resonance (NMR) purity greater than 95% after standard chromatographic purification, ensuring suitability for sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NH-Sulfoximine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value pharmaceutical intermediates like NH-sulfoximines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We are committed to delivering high-purity NH-sulfoximine compounds that meet the exacting standards of the pharmaceutical industry.
We invite procurement teams and R&D leaders to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of this green technology. By leveraging our expertise in process development and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your specific project requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your next project benefits from the highest levels of quality and efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
