Advanced Synthesis of 7-Amino-5-Azaspiro Heptane Quinolone Intermediates for Commercial Scale
The pharmaceutical industry continuously seeks enhancements in antibiotic efficacy, particularly regarding oral bioavailability and therapeutic index. Patent CN1036193C introduces a groundbreaking approach utilizing 7-unsubstituted or substituted amino-5-azaspiro(2,4)heptane structures to address these critical needs. These novel compounds effectively solve the long-standing limitation of poor absorption found in many high-activity quinolone derivatives currently on the market. By integrating a spiro-cyclic amine moiety at the 7-position of the quinolone nucleus, the invention significantly alters the physicochemical properties of the final active pharmaceutical ingredient. This structural modification enhances lipophilicity, thereby facilitating better membrane permeability and systemic distribution after oral administration. The patent details comprehensive synthetic pathways that ensure high purity and stereochemical control, which are paramount for regulatory compliance and clinical success in treating bacterial infections across human and veterinary sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthesis methods for quinolone antibiotics often rely on simple cyclic amines that fail to provide the necessary steric bulk and lipophilic balance required for optimal pharmacokinetics in modern treatments. Traditional routes may involve multiple protection and deprotection steps that increase waste generation and reduce overall yield efficiency significantly. Furthermore, older methodologies frequently struggle with the resolution of racemic mixtures, leading to products with inconsistent biological activity profiles and potential safety concerns. The lack of specific spiro-structural integration in legacy processes limits the ability to fine-tune the interaction between the drug molecule and bacterial DNA gyrase effectively. Consequently, manufacturers face challenges in scaling these processes without incurring substantial costs related to solvent recovery and impurity management. These limitations necessitate a shift towards more sophisticated molecular architectures that can be produced reliably and economically.
The Novel Approach
The novel approach described in the patent leverages the unique reactivity of spiro-containing cyclic amines to overcome these historical barriers in antibiotic synthesis. By employing specific precursors like ethyl acetoacetate and utilizing controlled cyclization conditions, the process generates the 5-azaspiro[2,4]heptane core with high fidelity and purity. The method incorporates strategic use of protecting groups such as tert-butoxycarbonyl to manage reactivity during the coupling phase efficiently. This ensures that the nitrogen atom at the 7-position of the quinolone is substituted selectively without affecting other sensitive functional groups on the molecule. The resulting intermediates exhibit superior stability and can be purified through standard crystallization techniques, reducing the need for complex chromatographic separations. This streamlined workflow not only improves the chemical purity but also aligns with modern green chemistry principles by minimizing hazardous waste streams.
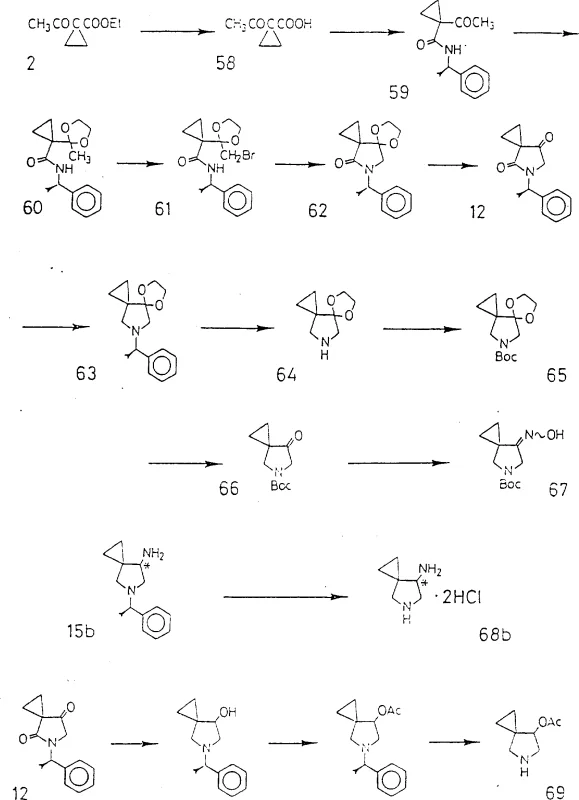
Mechanistic Insights into Spiro-Amine Coupling and Quinolone Functionalization
The core mechanistic insight involves the nucleophilic displacement of a halogen atom, typically fluorine or chlorine, at the 7-position of the quinolone ring system. The spiro-cyclic amine acts as a potent nucleophile, attacking the electron-deficient carbon center to form a stable carbon-nitrogen bond. This reaction is typically conducted in polar aprotic solvents like dimethyl sulfoxide or acetonitrile at elevated temperatures ranging from 80°C to 130°C. The presence of an acid binder, such as triethylamine or inorganic carbonates, is crucial to neutralize the hydrogen halide byproduct and drive the equilibrium towards product formation. The steric environment created by the spiro-ring system influences the transition state energy, favoring the formation of the desired regioisomer over potential side products. Understanding this kinetic profile is essential for optimizing reaction times and minimizing the formation of des-fluoro impurities.
Furthermore, the patent highlights the importance of stereochemistry in the biological activity of the final compounds for clinical applications. The synthesis allows for the resolution of optical isomers, specifically the 7-(S)-amino configuration, which has been demonstrated to possess higher antibacterial potency compared to its enantiomer. This selectivity is achieved through the use of chiral auxiliaries or resolution agents during the intermediate synthesis stages. The mechanism ensures that the spatial arrangement of the amino group relative to the spiro-ring is preserved throughout the coupling process. This level of control is vital for meeting stringent regulatory requirements regarding chiral purity in pharmaceutical manufacturing. The ability to produce single enantiomers reduces the metabolic load on patients and enhances the therapeutic index of the antibiotic.
How to Synthesize 7-Amino-5-Azaspiro Heptane Efficiently
The synthesis of these high-value intermediates requires precise adherence to the patented reaction conditions to ensure reproducibility and safety in a commercial setting. Operators must manage exothermic steps carefully, particularly during the reduction phases involving lithium aluminum hydride to prevent thermal runaways. The detailed standardized synthesis steps see the guide below provide a roadmap for transitioning from laboratory scale to pilot plant operations smoothly. Each stage, from the initial cyclization to the final deprotection, demands rigorous quality control checks to verify identity and purity. Implementing these protocols ensures that the supply chain remains robust against variations in raw material quality.
- Cyclize ethyl acetoacetate derivatives with amines to form the spiro-core structure under controlled basic conditions.
- Protect the amino group using tert-butoxycarbonyl reagents to ensure selectivity during subsequent coupling reactions.
- Couple the protected spiro-amine with 7-halo-quinolone derivatives in polar aprotic solvents at elevated temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
Commercial adoption of this technology offers distinct advantages for procurement and supply chain teams managing antibiotic portfolios globally. The streamlined synthesis reduces the number of unit operations, which directly correlates to lower operational expenditures and reduced facility occupancy time. This efficiency translates into a more competitive cost structure for the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps in favor of crystallization significantly lowers solvent consumption and waste disposal costs. By utilizing readily available starting materials like ethyl acetoacetate and benzylamine, the process avoids reliance on exotic or supply-constrained reagents. The high yield of the cyclization steps ensures that material throughput is maximized, reducing the cost per kilogram of the active intermediate. Additionally, the robustness of the reaction conditions allows for the use of standard stainless-steel equipment without the need for specialized lining, further decreasing capital expenditure requirements for production lines. This economic efficiency is compounded by the reduced need for specialized labor to manage complex separation units, allowing operational resources to be allocated more effectively across the manufacturing site.
- Enhanced Supply Chain Reliability: The synthetic route is designed to be resilient against fluctuations in raw material availability, utilizing commodity chemicals that are widely sourced from multiple global suppliers. The intermediate stability allows for extended storage periods without significant degradation, enabling manufacturers to build strategic inventory buffers. This flexibility mitigates the risk of production stoppages due to logistics delays or geopolitical supply disruptions. Furthermore, the scalability of the process from grams to metric tons ensures that supply can be ramped up quickly to meet sudden surges in demand for critical antibiotic therapies. The consistent quality of the output reduces the need for re-processing, ensuring a steady flow of materials to downstream formulation units.
- Scalability and Environmental Compliance: The process generates fewer hazardous byproducts compared to traditional quinolone synthesis methods, simplifying effluent treatment and reducing environmental compliance burdens. The use of catalytic hydrogenation for deprotection steps replaces stoichiometric chemical reductions, aligning with sustainability goals. Waste streams are primarily composed of organic solvents that can be recovered and recycled, minimizing the overall environmental footprint. This compliance advantage facilitates faster regulatory approvals in regions with strict environmental standards, ensuring uninterrupted market access for the final pharmaceutical products. Moreover, the energy efficiency of the reaction temperatures contributes to a lower carbon footprint per unit of production.
Frequently Asked Questions (FAQ)
Addressing technical queries is essential for stakeholders evaluating the feasibility of integrating this intermediate into their existing manufacturing frameworks. The following answers are derived directly from the experimental data and claims within the patent documentation. They cover critical aspects regarding stability, purity, and regulatory alignment.
Q: How does the spiro-structure improve oral absorption?
A: The spiro-cyclic amine moiety increases the lipophilicity of the quinolone derivative, facilitating better membrane permeability and systemic distribution after oral administration compared to non-spiro analogs.
Q: Is stereochemical control possible in this synthesis?
A: Yes, the patent describes methods to resolve optical isomers, specifically favoring the 7-(S)-amino configuration which demonstrates higher antibacterial potency.
Q: What are the scalability advantages of this route?
A: The process utilizes commodity chemicals and standard equipment, allowing for easy scale-up from grams to metric tons without requiring specialized reactor linings or complex purification units.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Amino-5-Azaspiro Heptane Supplier
Partnering with NINGBO INNO PHARMCHEM provides access to this advanced technology through our experienced CDMO capabilities and technical expertise. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the highest international standards for pharmaceutical intermediates. We are committed to supporting your development timelines with consistent quality and transparent communication throughout the project lifecycle.
We invite you to request a Customized Cost-Saving Analysis to understand how this route can optimize your specific budget and operational efficiency. Contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us help you secure a stable and cost-effective supply chain for your next-generation antibiotic products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
