Advanced One-Step Synthesis of Chiral Oxazolines for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust methodologies for constructing chiral heterocycles, which serve as critical scaffolds in drug discovery and development. Patent CN102643246B introduces a groundbreaking approach to synthesizing chiral 2-carbonyl oxazolines, specifically targeting the production of (S)-4-isopropyl-oxazolin-2-one. This technology leverages the unique reactivity of 7,7,8,8-tetracyanoquinodimethane (TCNQ) in the presence of a zinc chloride catalyst to achieve a direct, one-step cyclization with L-valinol. For R&D directors and procurement specialists, this represents a significant shift from traditional multi-step protections and deprotections towards a more convergent synthetic strategy. The ability to generate the necessary carbonyl functionality in situ from the cyano groups of TCNQ simplifies the supply chain requirements and reduces the handling of hazardous reagents. As a reliable pharmaceutical intermediates supplier, understanding such novel pathways is essential for evaluating the long-term viability and cost-efficiency of key building blocks used in oncology and other therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral oxazolidinones and related oxazoline derivatives has relied heavily on the reaction of amino alcohols with activated carbonates, phosgene equivalents, or carboxylic acids. These conventional routes often suffer from significant drawbacks, including the requirement for strictly anhydrous conditions, the use of toxic reagents like phosgene or triphosgene, and the necessity for protecting group strategies to prevent side reactions. Furthermore, many existing methods involve multiple isolation steps, leading to cumulative yield losses and increased waste generation. The reliance on pre-functionalized carbonyl sources adds complexity to the supply chain, as these reagents can be unstable or expensive to procure in bulk quantities. For manufacturing teams, the disposal of halogenated byproducts and the management of exothermic risks associated with traditional activation agents pose continuous operational challenges that drive up the overall cost of goods sold.
The Novel Approach
In stark contrast, the methodology disclosed in CN102643246B offers a streamlined alternative by utilizing TCNQ as a latent source of formic acid. Under the catalytic influence of zinc chloride in chlorobenzene, the cyano groups of TCNQ undergo transformation to generate the requisite carbonyl species directly within the reaction mixture. This allows for a direct condensation with L-valinol, followed by spontaneous cyclization to form the target oxazoline ring system. This one-pot protocol eliminates the need for external formic acid addition and avoids the handling of volatile, corrosive acids. The process operates under reflux conditions, which are thermally efficient and easy to control in standard reactor vessels. By consolidating the generation of the electrophile and the nucleophilic attack into a single operational step, this novel approach significantly reduces processing time and solvent consumption, aligning perfectly with the goals of cost reduction in pharmaceutical intermediates manufacturing.
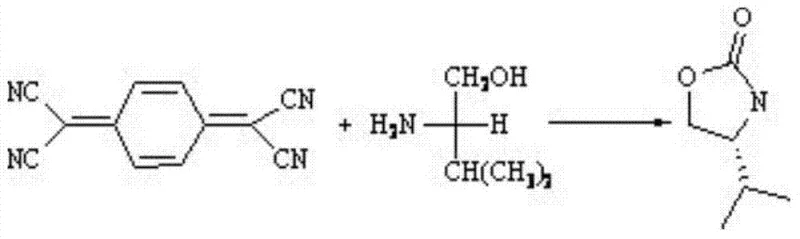
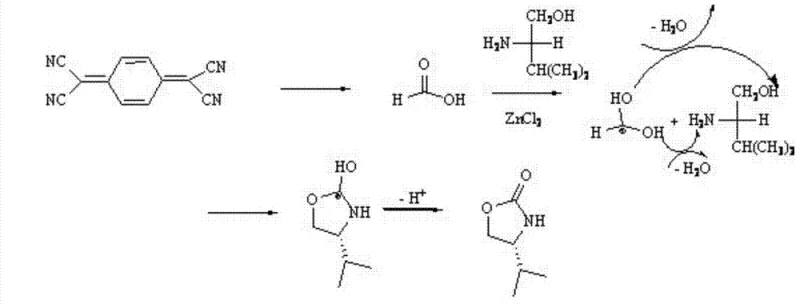
Mechanistic Insights into ZnCl2-Catalyzed Cyclization
The core of this synthetic innovation lies in the Lewis acid-mediated activation of the nitrile groups present in the TCNQ molecule. Zinc chloride, acting as a strong Lewis acid, coordinates with the nitrogen atoms of the cyano groups, increasing their electrophilicity and rendering them susceptible to hydrolysis or alcoholysis in the presence of trace moisture or the hydroxyl group of the amino alcohol. The patent suggests a mechanism where the cyano groups are converted into formic acid equivalents in situ. This generated formic acid then engages in a condensation reaction with the amine functionality of L-valinol. The proximity of the hydroxyl group in the amino alcohol facilitates an intramolecular nucleophilic attack on the carbonyl carbon, leading to the formation of the five-membered oxazolidinone ring. This cascade sequence is driven by the thermodynamic stability of the resulting heterocycle and the removal of water molecules during the condensation phases.
Furthermore, the stereochemical integrity of the final product is preserved throughout this rigorous thermal process. Since the chirality is inherent in the L-valinol starting material, the reaction conditions must be mild enough to prevent racemization at the alpha-carbon. The use of zinc chloride appears to facilitate the cyclization without inducing epimerization, ensuring that the final (S)-4-isopropyl-oxazolin-2-one retains high optical purity. The mechanism also accounts for the formation of byproducts, which are effectively separated via column chromatography using a petroleum ether and dichloromethane system. Understanding this mechanistic pathway allows process chemists to fine-tune parameters such as catalyst loading and temperature to maximize the 40% yield reported in the embodiment, potentially improving upon it through optimization of the workup procedure.
How to Synthesize (S)-4-isopropyl-oxazolin-2-one Efficiently
To implement this synthesis effectively, precise control over the stoichiometry and reaction environment is required. The process begins with the suspension of anhydrous zinc chloride in chlorobenzene, followed by the addition of TCNQ and a substantial excess of L-valinol to drive the equilibrium forward. The mixture is subjected to prolonged reflux, typically lasting around 72 hours, to ensure complete conversion of the stable TCNQ precursor. Following the reaction, the workup involves solvent removal and extraction, with the final purification achieved through silica gel chromatography.
- Combine 7,7,8,8-tetracyanoquinodimethane (TCNQ) and L-valinol in chlorobenzene solvent with 105 mol% zinc chloride catalyst.
- Perform reflux reaction for 72 hours (3 days) to facilitate the in situ generation of formic acid and subsequent condensation.
- Purify the crude product via column chromatography using petroleum ether and dichloromethane to isolate the target chiral oxazoline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this TCNQ-based route offers distinct strategic advantages over legacy manufacturing processes. The primary benefit stems from the simplification of the raw material portfolio; by using TCNQ to generate the carbonyl component internally, manufacturers can reduce their dependency on external suppliers of activated carboxylic acid derivatives. This consolidation of reagents not only simplifies inventory management but also mitigates the risks associated with the transportation and storage of hazardous acylating agents. Additionally, the use of chlorobenzene as a solvent is advantageous for large-scale operations due to its high boiling point, which allows for vigorous reflux without the need for pressurized equipment, thereby enhancing operational safety and reducing capital expenditure on specialized reactors.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of discrete steps for carbonyl activation and the reduction in reagent costs. By generating the reactive formic acid species in situ from TCNQ, the process avoids the purchase of expensive coupling reagents or protected precursors. Although the catalyst loading is relatively high at 105 mol%, zinc chloride is an inexpensive and widely available commodity chemical. The overall reduction in unit operations—from multiple reaction vessels to a single pot—translates to lower labor costs, reduced energy consumption for heating and cooling cycles, and decreased solvent waste disposal fees, leading to substantial cost savings in the final production budget.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes significantly to supply chain continuity. Both TCNQ and L-valinol are commercially available fine chemicals with established global supply networks, reducing the risk of raw material shortages. The tolerance of the reaction to standard industrial conditions means that production can be easily transferred between different manufacturing sites without requiring highly specialized infrastructure. This flexibility ensures that production schedules can be maintained even during periods of logistical disruption, providing a reliable source of high-purity pharmaceutical intermediates for downstream drug synthesis.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns well with green chemistry principles by minimizing the number of synthetic steps and avoiding the use of phosgene or its equivalents. The waste stream is primarily composed of organic solvents and zinc salts, which are manageable through standard wastewater treatment protocols. The simplicity of the workup, involving extraction and chromatography, is amenable to scale-up using simulated moving bed (SMB) chromatography or crystallization techniques for larger batches. This scalability ensures that the process can meet the demands of commercial production ranging from pilot plant studies to multi-ton annual outputs without compromising on quality or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and mechanistic proposals found in the patent documentation, providing a clear framework for evaluating the feasibility of this route for your specific applications.
Q: What is the primary advantage of using TCNQ in this oxazoline synthesis?
A: The use of 7,7,8,8-tetracyanoquinodimethane (TCNQ) serves a dual purpose: it acts as a precursor that decomposes under Lewis acid catalysis to generate formic acid in situ, which then reacts with the amino alcohol. This eliminates the need for handling separate corrosive formic acid reagents and streamlines the atom economy of the reaction sequence.
Q: How does the zinc chloride catalyst influence the stereoselectivity?
A: Zinc chloride acts as a potent Lewis acid that coordinates with the nitrogen and oxygen atoms of the reactants, facilitating the cyclization process. While the chirality is primarily derived from the L-valinol starting material, the catalyst ensures efficient ring closure to form the stable oxazolidinone structure without racemization under the specified reflux conditions.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes standard industrial solvents like chlorobenzene and operates under reflux conditions, which are easily scalable. The simplicity of the one-pot reaction design reduces unit operations, making it highly attractive for reducing lead time for high-purity pharmaceutical intermediates in a commercial setting.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-isopropyl-oxazolin-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that chiral oxazolines play in the synthesis of advanced therapeutic agents. Our team of expert process chemists is fully equipped to adapt the methodology described in CN102643246B to meet your specific volume and purity requirements. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities include rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the enantiomeric excess of the final product. We are committed to delivering high-quality intermediates that accelerate your drug development timelines.
We invite you to collaborate with us to explore the full potential of this efficient synthetic route. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your project's specific constraints. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive pricing for your upcoming projects. Let us be your partner in transforming innovative patent chemistry into commercial reality.
