Advanced Catalytic Synthesis of Chiral Acyl Ammonium Salts for Commercial Scale-up and Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the construction of chiral building blocks, which serve as the foundational elements for complex active pharmaceutical ingredients. Patent CN102659633A introduces a significant advancement in this domain by detailing a novel synthetic method for chiral acyl ammonium salts, specifically focusing on the preparation of (S)-3-methyl-butyl-1-hydroxymethyl carbamic acid-(S)-3-methyl-butyl-1-hydroxymethyl ammonium salt. This technology leverages the unique reactivity of 7,7,8,8-tetracyanoquinodimethane (TCNQ) in the presence of a zinc chloride catalyst and L-leucinol to achieve a direct condensation reaction. The strategic importance of this patent lies in its ability to generate complex chiral ammonium structures through a streamlined one-step process, bypassing the need for multi-step protection and deprotection sequences that traditionally plague chiral synthesis. For R&D directors and procurement specialists, this represents a pivotal shift towards more efficient access to high-value chiral intermediates, potentially reducing the overall timeline for drug development pipelines while maintaining rigorous stereochemical control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral acyl ammonium salts has been fraught with significant technical hurdles that impede efficient commercial production. Conventional routes often rely on the resolution of racemic mixtures, a process that inherently limits the maximum theoretical yield to 50% and generates substantial chemical waste in the form of the unwanted enantiomer. Furthermore, existing methodologies frequently necessitate the use of expensive chiral auxiliaries or precious metal catalysts, which not only drive up the raw material costs but also introduce complex purification challenges to remove trace metal contaminants from the final product. The multi-step nature of these traditional pathways increases the operational burden on manufacturing facilities, requiring extensive time for intermediate isolation and quality control testing at each stage. These inefficiencies create bottlenecks in the supply chain, leading to longer lead times and higher volatility in the pricing of critical pharmaceutical intermediates, thereby affecting the overall cost structure of downstream drug manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN102659633A offers a transformative one-step synthetic route that dramatically simplifies the production landscape. By utilizing L-leucinol, a readily available chiral pool material, the process inherently embeds stereochemistry into the product without the need for external resolution steps. The reaction proceeds through a direct condensation mechanism facilitated by zinc chloride, which activates the nitrile groups of the TCNQ precursor for nucleophilic attack. This approach eliminates the need for expensive precious metal catalysts and reduces the number of unit operations required, thereby lowering both capital expenditure and operational costs. The simplicity of the work-up procedure, involving standard column chromatography and solvent evaporation, further enhances the practicality of this method for industrial adoption. This novel pathway not only improves the atom economy of the synthesis but also aligns with green chemistry principles by minimizing waste generation and energy consumption associated with prolonged multi-step processing.

Mechanistic Insights into Zinc Chloride-Catalyzed Condensation
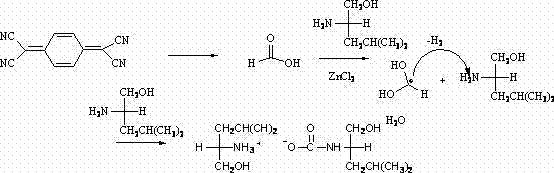
The core of this technological breakthrough lies in the intricate mechanistic pathway driven by the Lewis acidity of zinc chloride. Under the reflux conditions in chlorobenzene, the zinc chloride catalyst initiates the hydrolysis of the unstable 7,7,8,8-tetracyanoquinodimethane, converting the cyano groups into formic acid intermediates in situ. This generated formic acid then undergoes a condensation reaction with the excessive L-leucinol present in the reaction matrix. The carbonium ion within the formic acid intermediate is susceptible to nucleophilic attack by the amino group of the leucinol, leading to the formation of a critical C-N bond. Simultaneously, the hydroxyl groups participate in the stabilization of the transition state, facilitating the elimination of water molecules and driving the equilibrium towards the formation of the carbamate linkage. This cascade of reactions culminates in the assembly of the chiral acyl ammonium salt structure, where the stereochemical information from the L-leucinol is preserved and transferred to the final ammonium salt product.
Understanding the impurity profile is crucial for R&D teams aiming to replicate this process at scale. The mechanism suggests that the primary side reactions could involve incomplete hydrolysis of the nitrile groups or over-alkylation of the amino centers if the stoichiometry is not strictly controlled. However, the use of a significant excess of L-leucinol (90mmol relative to 4.90mmol of TCNQ) ensures that the equilibrium is pushed heavily towards the desired product, minimizing the presence of unreacted starting materials. The zinc chloride catalyst also plays a role in suppressing side reactions by coordinating with the nitrogen atoms, thereby modulating their nucleophilicity and preventing polymerization or degradation of the sensitive quinodimethane core. This precise control over the reaction environment allows for the isolation of monocrystalline products with high purity, as evidenced by the X-diffraction analysis data provided in the patent documentation, ensuring that the material meets the stringent specifications required for pharmaceutical applications.
How to Synthesize Chiral Acyl Ammonium Salt Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and purity. The process begins with the precise weighing of anhydrous zinc chloride, which must be handled under inert conditions to prevent premature hydrolysis before the reaction starts. The solvent system, chlorobenzene, is chosen for its high boiling point, which allows the reaction to proceed at elevated temperatures necessary for the activation of the sterically hindered TCNQ molecule. Operators must maintain a consistent reflux temperature for a duration of 72 hours to ensure complete conversion, as the reaction kinetics are relatively slow due to the stability of the starting nitrile groups. Following the reaction, the work-up involves a standard extraction protocol using dichloromethane and water, followed by drying over anhydrous sodium sulfate to remove trace moisture that could affect the stability of the ammonium salt. The final purification step utilizes column chromatography with a specific ratio of petroleum ether to dichloromethane, which is critical for separating the product from any residual zinc salts or organic byproducts.
- Prepare the reaction mixture by combining 150 mol% zinc chloride catalyst with 7,7,8,8-tetracyanoquinodimethane and L-leucinol in chlorobenzene solvent.
- Maintain the reaction under reflux conditions for 72 hours to ensure complete condensation and formation of the ammonium salt structure.
- Purify the crude product using column chromatography with petroleum ether and dichloromethane elution to isolate the monocrystalline salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The primary benefit is the substantial reduction in raw material costs achieved by replacing expensive chiral resolution agents with commodity-grade L-leucinol. This shift not only lowers the direct material cost but also stabilizes the supply chain by relying on widely available feedstocks rather than specialized, low-volume reagents. Furthermore, the elimination of precious metal catalysts removes the need for costly metal scavenging steps and the associated regulatory testing for heavy metal residues, which significantly reduces the analytical burden and time-to-market for new drug candidates. The simplified one-step process also translates to lower energy consumption and reduced solvent usage per kilogram of product, contributing to a more sustainable and cost-effective manufacturing footprint that aligns with modern environmental compliance standards.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the strategic selection of reagents that minimize waste and maximize atom economy. By utilizing a catalytic amount of zinc chloride instead of stoichiometric amounts of expensive chiral reagents, the process drastically reduces the cost of goods sold. Additionally, the one-step nature of the synthesis eliminates the need for multiple isolation and purification stages, which are typically the most expensive parts of chemical manufacturing in terms of labor and equipment time. This streamlined approach allows for a significant reduction in operational expenditures, making the final chiral intermediate more price-competitive in the global market without compromising on quality or stereochemical purity.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the reliance on L-leucinol, a bulk chemical with a stable and robust global supply network. Unlike specialized chiral catalysts that may have long lead times or single-source risks, L-leucinol is produced at scale for various industries, ensuring consistent availability and price stability. The use of common solvents like chlorobenzene and dichloromethane further mitigates supply risks, as these are standard inventory items for most chemical manufacturing facilities. This accessibility ensures that production schedules can be maintained without interruption, providing downstream pharmaceutical clients with a reliable source of critical intermediates even during periods of market volatility or raw material shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor equipment and conditions that are easily transferable from laboratory to pilot and commercial scales. The absence of hazardous reagents or extreme pressure conditions simplifies the safety profile of the manufacturing process, reducing the need for specialized containment infrastructure. From an environmental standpoint, the reduced solvent usage and the elimination of heavy metal waste streams facilitate easier waste treatment and disposal, helping manufacturers meet increasingly stringent environmental regulations. This compliance advantage not only avoids potential fines but also enhances the corporate sustainability profile, which is becoming a key factor in supplier selection for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These answers are derived directly from the experimental data and mechanistic insights provided in the patent literature, offering a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows or for procurement teams assessing the long-term viability of the supply chain.
Q: What is the primary advantage of using zinc chloride in this synthesis?
A: Zinc chloride acts as a Lewis acid catalyst that facilitates the hydrolysis of the tetracyanoquinodimethane precursor and promotes the subsequent condensation with L-leucinol, enabling a one-step formation of the chiral salt.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of commodity chemicals like L-leucinol and standard solvents like chlorobenzene makes the process highly adaptable for commercial scale-up, provided that reflux conditions are strictly controlled.
Q: What is the expected purity of the final product?
A: Through rigorous column chromatography purification and recrystallization, the process yields monocrystalline products with high stereochemical integrity, suitable for sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Acyl Ammonium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the zinc chloride-catalyzed synthesis can be successfully translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify stereochemical integrity and impurity profiles. Our dedication to technical excellence ensures that every batch of chiral acyl ammonium salt we supply is consistent, reliable, and ready for use in sensitive pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our advanced synthetic capabilities can enhance your operational efficiency. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable network of chemical expertise and manufacturing capacity that is dedicated to supporting your success in the competitive global pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
