Advanced Catalytic Hydrogenation for Benzhydrol: Scalable Solutions for Global Pharma Supply Chains
Advanced Catalytic Hydrogenation for Benzhydrol: Scalable Solutions for Global Pharma Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking more efficient, environmentally benign, and cost-effective pathways for synthesizing critical intermediates like benzhydrol and its derivatives. A significant technological advancement in this domain is detailed in patent CN102241566B, which discloses a novel preparation method utilizing transition metal complexes for catalytic hydrogenation. This approach represents a paradigm shift from stoichiometric reduction methods, offering a robust solution for the production of high-purity benzhydrol. By leveraging specific nitrogen-phosphine transition metal catalysts, this method achieves exceptional conversion rates exceeding 98% while minimizing byproduct formation. For R&D Directors and Procurement Managers alike, understanding the nuances of this technology is crucial for optimizing supply chains and reducing manufacturing costs in the competitive landscape of API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benzhydrol and its derivatives has relied heavily on stoichiometric reduction methods that pose significant environmental and operational challenges. Traditional processes often utilize zinc powder or aluminum powder in alkaline environments, which necessitates the handling and disposal of large quantities of metal waste, creating a substantial burden on waste management systems and increasing overall production costs. Alternatively, the use of sodium borohydride or potassium borohydride, while effective, frequently results in the formation of boric acid ester byproducts that complicate downstream purification and can negatively impact the overall yield. Furthermore, conventional catalytic hydrogenation methods using palladium carbon or Raney nickel often suffer from issues such as over-reduction to diphenylmethane or require prolonged reaction times, as seen in some prior art where reactions extend up to 48 hours. These inefficiencies create bottlenecks in production schedules and elevate the cost reduction in pharmaceutical intermediate manufacturing, making it difficult to maintain competitive pricing without sacrificing quality or compliance.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a specialized transition metal complex catalyst that dramatically enhances reaction efficiency and selectivity. This method operates under milder conditions with significantly reduced catalyst loading, yet it maintains a stable reaction process that consistently delivers high conversion rates. The use of a nitrogen-phosphine transition metal complex allows for precise control over the hydrogenation process, effectively suppressing the formation of unwanted byproducts like diphenylmethane that plague older catalytic systems. Moreover, the post-reaction treatment is remarkably simple, eliminating the need for complex filtration of metal sludges or extensive purification steps to remove boron residues. This streamlined workflow not only shortens the entire process cycle but also aligns perfectly with modern green chemistry principles, offering a sustainable pathway for the commercial scale-up of complex pharmaceutical intermediates. 
Mechanistic Insights into Transition Metal Complex Catalyzed Hydrogenation
The core of this technological breakthrough lies in the unique structure of the transition metal complex catalyst, which follows the general formula MLnL′XY. In this system, the transition metal M is typically Ruthenium or Rhodium, coordinated with ligands that possess specific NH2-N(SP2) or NH2-NH2 structural features. These ligands play a critical role in facilitating the hydrogen transfer mechanism, ensuring that the hydrogenation of the carbonyl group in benzophenone proceeds with high stereoselectivity and chemoselectivity. The presence of phosphine ligands such as BINAP or TolBINAP further stabilizes the metal center, allowing the reaction to proceed efficiently even at relatively low catalyst concentrations ranging from 1:20,000 to 1:1,000,000 molar ratios. This high turnover number is a testament to the robustness of the catalytic cycle, which minimizes metal leaching and ensures that the final product meets stringent purity specifications required for pharmaceutical applications. 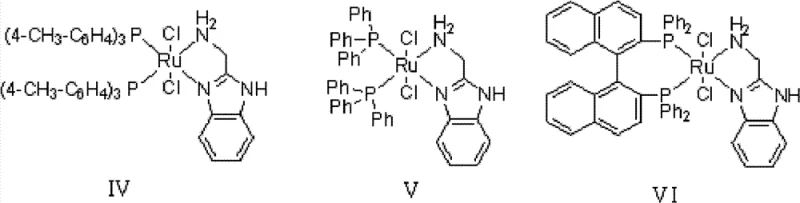
From an impurity control perspective, this catalytic system offers distinct advantages over traditional reducing agents by fundamentally altering the reaction pathway. The specific coordination environment created by the diamine and phosphine ligands prevents the over-reduction of the alcohol product to the corresponding hydrocarbon, a common side reaction in less selective catalytic systems. Additionally, the absence of stoichiometric metal reductants means there is no risk of metal contamination from zinc or aluminum, which can be notoriously difficult to remove to trace levels. The reaction conditions, which typically involve temperatures between 25°C to 100°C and hydrogen pressures from 0.5MPa to 10MPa, are optimized to maximize the formation of the desired benzhydrol while minimizing thermal degradation or polymerization of sensitive functional groups. This level of control is essential for producing high-purity benzhydrol derivatives, such as 4,4'-difluoro diphenyl methanol, which are critical precursors for calcium channel blockers and antihistamines.
How to Synthesize Benzhydrol Efficiently
Implementing this synthesis route in a production environment requires careful attention to the preparation of the reaction mixture and the control of hydrogenation parameters. The process begins with the charging of benzophenone and a suitable solvent, such as toluene or tetrahydrofuran, into a reactor under an inert atmosphere to prevent oxidation. Following this, the transition metal complex catalyst and a base, which can be a mineral alkali like potassium hydroxide or an organic amine like triethylamine, are added to initiate the catalytic cycle. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and conditions required to achieve the reported >98% conversion efficiency. Adhering to these protocols ensures that the benefits of the novel catalyst system are fully realized, providing a reliable and reproducible method for manufacturing this key intermediate.
- Charge benzophenone and a suitable solvent such as toluene or tetrahydrofuran into a stainless steel reactor under an inert nitrogen atmosphere.
- Add the specific transition metal complex catalyst along with a base such as potassium tert-butoxide or triethylamine to the reaction mixture.
- Pressurize with hydrogen gas and maintain reaction temperature between 25°C to 100°C until completion, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic hydrogenation technology translates into tangible operational benefits that extend beyond simple chemical yield. The elimination of stoichiometric metal reductants like zinc or aluminum powder removes a major source of hazardous waste, thereby reducing the costs associated with waste disposal and environmental compliance. Furthermore, the simplified post-reaction workup, which avoids complex filtration of metal sludges or boron residues, significantly reduces the processing time and labor required per batch. This efficiency gain allows for faster turnover of production equipment, effectively increasing the overall capacity of the manufacturing facility without the need for capital investment in new hardware. By reducing lead time for high-purity benzhydrol, manufacturers can respond more agilely to market demands and ensure a continuous supply of critical intermediates to downstream API producers.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in raw material consumption and waste treatment expenses. By utilizing a catalyst with a high turnover number, the amount of expensive transition metal required per kilogram of product is minimized, leading to substantial cost savings compared to methods requiring excess reductants. Additionally, the high conversion rate of over 98% means that less starting material is wasted, and the yield of the desired product is maximized, further improving the overall cost efficiency of the manufacturing process. The qualitative improvement in process simplicity also reduces utility consumption, as shorter reaction cycles and simpler workups require less energy for heating, cooling, and separation operations.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system contributes significantly to supply chain stability by reducing the risk of batch failures due to impurity issues or incomplete reactions. The use of readily available solvents and bases, combined with a catalyst that tolerates a range of reaction conditions, ensures that production can continue smoothly even if minor variations in raw material quality occur. This reliability is crucial for maintaining long-term contracts with pharmaceutical clients who require consistent quality and on-time delivery. Moreover, the scalability of the process, demonstrated in 100L reactor embodiments, assures buyers that the supply can be ramped up quickly to meet surges in demand without compromising on the quality or purity of the benzhydrol supplied.
- Scalability and Environmental Compliance: As regulatory pressures on chemical manufacturing intensify, the environmental profile of a synthesis route becomes a key factor in supplier selection. This hydrogenation method generates minimal waste and avoids the use of toxic heavy metals in stoichiometric quantities, making it easier to comply with strict environmental regulations. The ability to scale this process from laboratory to commercial production without significant re-engineering demonstrates its maturity and readiness for industrial application. This scalability ensures that the supply of benzhydrol can grow in tandem with the needs of the pharmaceutical industry, providing a sustainable and compliant source of this vital intermediate for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of benzhydrol using this advanced catalytic method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions about integrating this technology into their supply chains.
Q: What are the primary advantages of this catalytic method over traditional zinc powder reduction?
A: Traditional zinc or aluminum powder reduction generates significant amounts of metal waste and requires complex post-treatment filtration. This catalytic hydrogenation method utilizes minimal catalyst loading, produces very few byproducts, and simplifies the workup process significantly, leading to a cleaner environmental profile and reduced disposal costs.
Q: What conversion efficiency can be expected using this transition metal complex catalyst?
A: According to the technical data in patent CN102241566B, the process consistently achieves a conversion rate of more than 98%. In specific embodiments, conversion efficiencies reached 99.2% and 99.7%, demonstrating high reaction stability and effectiveness across different substrate derivatives.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the patent explicitly details embodiments conducted in 100L stainless steel reactors, proving the feasibility of scale-up. The process uses stable reaction conditions, common solvents, and robust catalysts, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates without significant engineering barriers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzhydrol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for key pharmaceutical intermediates like benzhydrol. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest industry standards. By leveraging advanced technologies such as the transition metal catalyzed hydrogenation described in patent CN102241566B, we are able to offer high-purity benzhydrol that supports the complex needs of modern drug development and manufacturing.
We invite you to collaborate with us to optimize your supply chain and achieve significant operational efficiencies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate how our capabilities can enhance your manufacturing process. Partnering with us means gaining access to a reliable benzhydrol supplier dedicated to driving innovation and value in the pharmaceutical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
