Advanced Synthesis and Commercial Scale-Up of Octahydro Cyclopenta Pyrrole Carboxylic Acid Derivatives
The pharmaceutical industry continuously seeks novel scaffolds that offer improved metabolic stability and binding affinity, and the octahydro cyclopenta[c]pyrrole carboxylic acid derivative represents a significant advancement in this domain. As disclosed in patent CN101463001A, this innovative chemical entity provides a robust foundation for designing compounds with diverse biological activities, particularly addressing the limitations of monocyclic proline analogs which often suffer from rapid enzymatic hydrolysis in vivo. The structural rigidity introduced by the fused bicyclic system enhances resistance against peptidase degradation, thereby potentially improving the bioavailability and half-life of therapeutic agents incorporating this motif. This technical breakthrough is not merely an academic exercise but a tangible solution for developing next-generation antiviral and cardiovascular medications where metabolic stability is paramount. By leveraging natural methionine esters as chiral starting materials, the synthesis ensures inherent optical purity, which is a critical quality attribute for any active pharmaceutical ingredient intended for human use. The ability to introduce various functional groups such as hydroxyl or fluorine at specific positions further expands the chemical space available for medicinal chemists to optimize drug-target interactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing bicyclic proline analogs often rely on non-selective cyclization reactions that yield complex mixtures of stereoisomers, necessitating costly and inefficient separation processes downstream. Many conventional methods utilize harsh reaction conditions, including extreme temperatures or highly toxic reagents, which pose significant safety risks and environmental challenges during large-scale manufacturing. Furthermore, the reliance on racemic starting materials in older methodologies frequently results in a maximum theoretical yield of only 50% for the desired enantiomer, leading to substantial material waste and increased production costs. The lack of precise control over the stereochemistry at the bridgehead carbons can also compromise the biological activity of the final drug candidate, as receptors often exhibit high specificity for a single enantiomer. Additionally, some legacy processes involve multiple protection and deprotection steps that add unnecessary complexity and reduce the overall atom economy of the synthesis. These inefficiencies create bottlenecks in the supply chain, making it difficult to secure reliable quantities of high-purity intermediates for clinical trials and commercial launch.
The Novel Approach
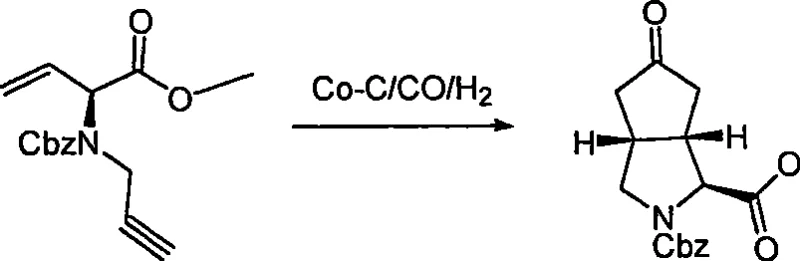
In stark contrast, the novel approach detailed in the patent utilizes a cobalt-carbonyl catalytic cyclization strategy that dramatically streamlines the construction of the octahydro cyclopenta[c]pyrrole core. This method capitalizes on the unique reactivity of cobalt complexes to facilitate the formation of carbon-carbon bonds under relatively mild conditions, preserving the integrity of sensitive functional groups present in the molecule. By starting from naturally occurring chiral pools like methionine, the synthesis bypasses the need for early-stage resolution, thereby locking in the desired stereochemistry from the outset. The process allows for the direct introduction of key functionalities such as ketones, which can be subsequently modified into hydroxyl or fluoro groups with high regioselectivity. This modular approach not only simplifies the synthetic pathway but also enhances the flexibility to generate a library of derivatives for structure-activity relationship studies. The robustness of the catalytic system ensures consistent performance across different batches, which is essential for maintaining quality standards in a regulated manufacturing environment.
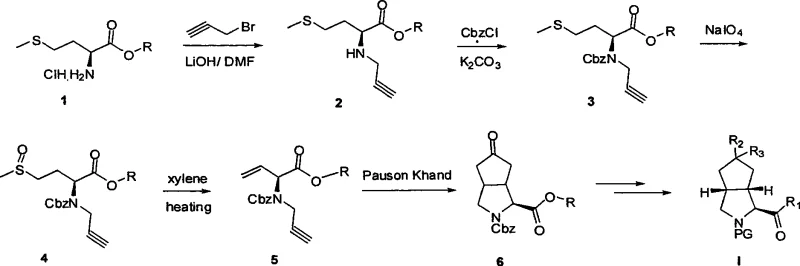
Mechanistic Insights into Cobalt-Carbonyl Catalytic Cyclization
The heart of this synthetic innovation lies in the intricate mechanism of the cobalt-carbonyl catalyzed ring-closing reaction, which effectively transforms a linear enyne precursor into the rigid bicyclic framework. This transformation typically proceeds through the coordination of the alkyne and alkene moieties to the cobalt center, followed by the insertion of carbon monoxide to form a metallacycle intermediate. The subsequent reductive elimination releases the cyclic enone product while regenerating the active catalyst species, allowing the cycle to continue with high turnover numbers. Understanding this mechanistic pathway is crucial for optimizing reaction parameters such as carbon monoxide pressure and temperature, which directly influence the rate of cyclization and the ratio of diastereomers formed. The use of specific ligands or additives, such as molecular sieves, can further enhance the efficiency of the reaction by sequestering water or other impurities that might deactivate the catalyst. This level of mechanistic control enables chemists to fine-tune the process to maximize yield and minimize the formation of side products, ensuring a cleaner reaction profile that simplifies downstream purification.
Impurity control within this mechanistic framework is achieved through a combination of kinetic control during the cyclization and thermodynamic control during subsequent processing steps. The chiral information encoded in the starting methionine derivative directs the facial selectivity of the cyclization, favoring the formation of one diastereomer over the other. However, minor amounts of the undesired isomer may still form, necessitating a robust chiral separation strategy to meet stringent purity specifications. The patent describes the use of advanced chromatographic techniques, such as supercritical fluid chromatography with chiral stationary phases, to effectively resolve these diastereomers. This ensures that the final product possesses the high optical purity required for pharmaceutical applications, preventing potential toxicity or reduced efficacy associated with the wrong enantiomer. Furthermore, the ability to convert the ketone intermediate into various derivatives allows for the masking or removal of impurities through selective chemical transformations, adding another layer of quality control to the overall process.
How to Synthesize Octahydro Cyclopenta Pyrrole Efficiently
The synthesis of these valuable intermediates begins with the preparation of a chiral propargyl amine from natural methionine, followed by a series of oxidation and elimination steps to generate the requisite enyne substrate. This precursor is then subjected to the critical cobalt-catalyzed cyclization under controlled carbon monoxide pressure to establish the bicyclic core. Subsequent functionalization steps, including reduction and fluorination, allow for the diversification of the scaffold to meet specific drug design requirements. The detailed standardized synthesis steps see below guide.
- Preparation of chiral propargyl amine intermediates from natural methionine ester via substitution and oxidation.
- Execution of cobalt-carbonyl catalytic cyclization under controlled CO pressure to form the bicyclic core structure.
- Functionalization of the core scaffold through reduction, fluorination, and chiral separation to achieve target optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented synthesis route offers compelling advantages that translate directly into cost efficiencies and risk mitigation for pharmaceutical manufacturers. The utilization of natural methionine as a starting material leverages a widely available and cost-effective chiral pool, reducing dependency on expensive synthetic chiral auxiliaries or resolution agents. This foundational choice significantly lowers the raw material costs and stabilizes the supply chain against fluctuations in the availability of specialized reagents. Moreover, the streamlined nature of the synthetic sequence reduces the total number of unit operations required, which in turn decreases labor costs, energy consumption, and waste generation. The robustness of the cobalt-catalyzed step ensures high reproducibility, minimizing the risk of batch failures that can disrupt production schedules and delay time-to-market for critical therapies. By adopting this efficient methodology, companies can achieve substantial cost savings in pharmaceutical intermediates manufacturing while maintaining the highest standards of quality and compliance.
- Cost Reduction in Manufacturing: The elimination of complex resolution steps early in the synthesis significantly reduces material loss and processing time, leading to a more economical production process. By avoiding the use of stoichiometric amounts of expensive chiral reagents and instead relying on catalytic processes, the overall cost of goods sold is drastically optimized. The high atom economy of the cyclization reaction ensures that a greater proportion of the starting material is incorporated into the final product, minimizing waste disposal costs. Additionally, the ability to perform reactions under milder conditions reduces the energy burden on the manufacturing facility, contributing to lower operational expenditures. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality or purity of the delivered intermediates.
- Enhanced Supply Chain Reliability: Sourcing strategies are strengthened by the use of commodity chemicals like methionine and cobalt salts, which are less susceptible to supply disruptions compared to bespoke fine chemicals. The scalability of the process means that production volumes can be ramped up quickly to meet surging demand during clinical trials or commercial launch phases without requiring significant capital investment in new equipment. The robustness of the chemistry also reduces the likelihood of technical queries or deviations during manufacturing, ensuring a steady and predictable flow of materials to the customer. This reliability is crucial for maintaining continuous production lines for downstream API manufacturing, preventing costly downtime and ensuring patient access to essential medications.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing catalytic amounts of metal rather than stoichiometric toxic reagents, which simplifies waste treatment and environmental compliance. The reduction in solvent usage and the potential for solvent recycling further enhance the sustainability profile of the manufacturing operation. Scalability is inherently built into the design, as the reaction conditions are compatible with standard industrial reactors, facilitating a seamless transition from pilot plant to full commercial scale. This alignment with environmental regulations and sustainability goals not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these octahydro cyclopenta pyrrole derivatives. These answers are derived directly from the patented technology and practical manufacturing experience, providing clarity on process capabilities and quality standards. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their drug development pipelines. The responses cover aspects ranging from stereochemical control to scalability, ensuring a comprehensive overview of the technology's value proposition.
Q: What is the primary advantage of using cobalt-carbonyl catalysis for this synthesis?
A: The cobalt-carbonyl catalytic cyclization allows for the construction of the complex bicyclic octahydro cyclopenta pyrrole core under relatively mild conditions compared to traditional high-temperature methods. This approach significantly enhances stereocontrol and reduces the formation of unwanted byproducts, ensuring higher optical purity which is critical for pharmaceutical applications.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patented process is specifically designed with scalability in mind. By utilizing readily available starting materials like natural methionine and employing robust catalytic systems, the route avoids exotic reagents that hinder scale-up. The reaction conditions, including pressure and temperature parameters, are manageable within standard industrial reactor setups, facilitating a smooth transition from laboratory to commercial manufacturing.
Q: How does this method address impurity control in chiral compounds?
A: Impurity control is achieved through a combination of stereoselective synthesis starting from chiral pools and subsequent chiral separation techniques. The process includes specific steps for diastereomer separation using advanced chromatography, ensuring that the final product meets stringent enantiomeric excess requirements necessary for regulatory compliance in drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Octahydro Cyclopenta Pyrrole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs, and we are committed to delivering excellence in every batch. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from preclinical studies to market launch. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every shipment meets the exacting standards required by global regulatory agencies. Our expertise in cobalt-catalyzed transformations allows us to optimize this specific route for maximum efficiency and yield, providing you with a competitive edge in your manufacturing costs. We understand the complexities of supply chain management and work proactively to mitigate risks, ensuring continuity of supply even in volatile market conditions.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our capabilities align with your project goals. By partnering with us, you gain access to a wealth of chemical expertise and manufacturing capacity dedicated to accelerating your path to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
