Advanced Phosphite Ester Acrylate Synthesis for Commercial Scale-Up and Supply Chain Optimization
The photocuring industry has long grappled with the persistent challenge of surface oxygen inhibition, a phenomenon that severely compromises the mechanical integrity and aesthetic quality of high-performance coatings and inks. Patent CN109880103B introduces a groundbreaking phosphite ester acrylate compound that fundamentally alters the landscape of radical photopolymerization by integrating a low surface tension polysiloxane chain directly into the monomer structure. This innovative molecular design allows the active species to spontaneously migrate to the air-interface of the coating, where oxygen inhibition is most critical, while simultaneously functioning as a redox-active auxiliary that regenerates free radicals to sustain the curing process. For R&D directors and procurement managers seeking a reliable phosphite ester acrylate supplier, this technology represents a significant leap forward in achieving complete surface curing without the detrimental side effects associated with traditional additives, such as yellowing or unpleasant odors, thereby unlocking new possibilities for transparent and light-colored material applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies to mitigate oxygen inhibition in free radical photopolymerization have historically relied on the addition of external auxiliaries such as tertiary amines or thiol compounds, both of which carry significant drawbacks that limit their utility in premium applications. Tertiary amines, while effective hydrogen donors, are notorious for causing severe yellowing in cured films, which renders them unsuitable for clear coats, optical materials, or any application where aesthetic clarity is paramount. Similarly, thiol-based aids suffer from poor processing stability and emit strong, pungent odors that create hazardous working environments and complicate regulatory compliance for consumer-facing products. Furthermore, these conventional additives often remain as separate phases within the coating matrix, leading to potential migration issues over time and failing to integrate permanently into the cross-linked network, which can ultimately degrade the long-term weather resistance and mechanical durability of the final polymer product.
The Novel Approach
The novel approach detailed in the patent data circumvents these historical limitations by chemically grafting the oxygen-scavenging phosphite functionality directly onto an acrylate monomer backbone that is further modified with a polysiloxane segment. This structural integration ensures that the anti-oxygen inhibition agent is not merely a transient additive but an intrinsic part of the curing network, permanently locked into the polymer matrix upon polymerization. The polysiloxane chain imparts a remarkably low surface energy to the molecule, driving it to enrich at the coating-air interface precisely where oxygen concentration is highest, thus providing targeted protection exactly where it is needed most. This dual-action mechanism not only eliminates the risk of yellowing associated with amine synergists but also enhances the surface properties of the coating, resulting in superior antifouling, wiping resistance, and overall firmness without compromising the transparency or visual appeal of the material.
Mechanistic Insights into Phosphite-Mediated Radical Regeneration
The core chemical innovation lies in the redox capability of the phosphite ester group, which interacts dynamically with peroxy radicals and alkoxy radicals generated during the initial stages of photopolymerization. When oxygen interferes with the growing polymer chain to form a peroxy radical, the phosphite moiety undergoes an oxidation-reduction reaction that consumes the inhibitory oxygen species and simultaneously releases a new active free radical. This regenerated radical is capable of re-initiating the polymerization process, effectively creating a self-sustaining cycle that overcomes the kinetic barrier imposed by atmospheric oxygen. The stability of the resulting phosphorus-oxygen double bond (P=O) ensures that the oxidation product is chemically inert and colorless, preventing the formation of chromophores that typically lead to material degradation and discoloration under UV exposure.
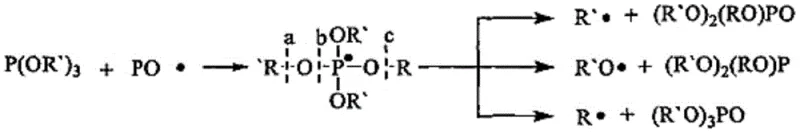
Furthermore, the incorporation of the polysiloxane chain segment plays a critical role in the spatial distribution of the active species within the curing film. Due to the shielding of the high-energy polysiloxane backbone by abundant alkyl groups on the silicon atoms, the compound exhibits weak intermolecular forces and minimal surface energy. This physicochemical property drives the monomer to float to the surface of the coating or ink formulation during the leveling process, creating a high concentration of oxygen-scavenging groups at the critical interface. This surface enrichment significantly increases the probability of the siloxane segments being incorporated into the cross-linked network, thereby improving the adhesion and firmness of the surface layer while simultaneously providing a hydrophobic barrier that enhances weather resistance and durability against environmental stressors.
How to Synthesize Phosphite Ester Acrylate Efficiently
The synthesis of this high-value intermediate involves a controlled multi-step substitution reaction that requires precise management of reaction conditions to ensure high purity and yield. The process begins with the reaction of a dihalogenated phosphorus compound with a hydroxyl-containing acrylate in the presence of an acid-binding agent, forming a reactive halogenated phosphite ester intermediate. This intermediate is then subjected to a second substitution reaction with a monohydroxy-terminated polysiloxane, linking the low surface tension chain to the phosphorus core. The entire procedure is typically conducted under an inert nitrogen atmosphere at controlled temperatures ranging from -10°C to 120°C to prevent premature polymerization or side reactions, followed by filtration and solvent removal to isolate the final product.
- React dihalogenated phosphorus compound with hydroxyl-containing acrylate in the presence of an acid-binding agent to form a halogenated phosphite ester intermediate.
- Add polysiloxane with a hydroxyl terminal group to the intermediate mixture to perform a secondary substitution reaction.
- Maintain reaction temperature between 30-120°C under nitrogen atmosphere, then filter and purify to obtain the final phosphite ester acrylate compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this integrated phosphite-siloxane monomer offers substantial advantages for procurement and supply chain stakeholders by simplifying the formulation process and reducing the total cost of ownership for coating manufacturers. By eliminating the need for separate, potentially hazardous oxygen inhibition additives like amines or thiols, formulators can streamline their raw material inventory and reduce the complexity of their supply chains. The inherent stability and non-yellowing nature of the compound also reduce the risk of batch rejection due to aesthetic defects, leading to higher first-pass yields and less waste in production. Additionally, the improved weather resistance and surface durability of the final coating can extend the service life of the end product, providing a strong value proposition for downstream customers in the automotive, furniture, and printing industries.
- Cost Reduction in Manufacturing: The integration of the oxygen inhibition function directly into the monomer structure eliminates the need for purchasing and handling separate auxiliary agents, which simplifies the batching process and reduces raw material handling costs. Furthermore, the elimination of expensive heavy metal catalysts or complex purification steps often required to remove amine residues results in significant cost savings in the overall manufacturing process. The improved curing efficiency also allows for faster line speeds in industrial coating applications, increasing throughput and maximizing the utilization of capital equipment without requiring additional energy input.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including dihalogenated phosphorus compounds and hydroxyl-terminated polysiloxanes, are commercially available from established chemical suppliers, ensuring a robust and continuous supply chain. The synthetic route utilizes standard substitution reactions that are well-understood and easily scalable, reducing the risk of production bottlenecks or technical failures during scale-up. This reliability allows procurement managers to secure long-term supply agreements with confidence, knowing that the production process is not dependent on exotic or single-source reagents that could be subject to market volatility.
- Scalability and Environmental Compliance: The synthesis process operates under relatively mild conditions and generates byproducts such as triethylamine hydrochloride that are easily removed by filtration, minimizing the burden on waste treatment facilities. The absence of volatile, odorous thiols or amines in the final formulation improves the working environment for plant operators and helps manufacturers meet increasingly stringent environmental and safety regulations. The scalability of the reaction from laboratory to commercial tonnage is supported by the use of common solvents and standard reactor configurations, facilitating a smooth transition from R&D to full-scale industrial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphite ester acrylate technology in industrial coating systems. These insights are derived directly from the patent specifications and are intended to assist technical teams in evaluating the feasibility of integrating this material into their existing product lines. Understanding the specific mechanistic advantages and processing requirements is essential for maximizing the performance benefits of this novel monomer.
Q: How does this phosphite ester acrylate solve surface oxygen inhibition?
A: The phosphite group structure undergoes a redox reaction with peroxides or oxygen, releasing active free radicals that re-initiate polymerization, effectively consuming oxygen at the coating surface.
Q: Does this compound cause yellowing in transparent coatings?
A: No, unlike amine or thiol-based auxiliaries, the oxidation product of the phosphite group forms a stable P=O bond which is colorless, ensuring excellent yellowing resistance for light-colored systems.
Q: What is the role of the polysiloxane chain in this molecule?
A: The polysiloxane chain provides low surface tension, causing the monomer to migrate to the coating surface where oxygen inhibition is most severe, thereby maximizing its protective effect.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphite Ester Acrylate Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like this phosphite-siloxane integration are executed with precision and consistency. Our rigorous QC labs and stringent purity specifications guarantee that every batch of high-purity phosphite ester acrylate meets the exacting standards required for advanced photocuring applications. We understand the critical importance of supply continuity for global coating manufacturers and have established robust logistics networks to deliver materials on time, every time, supporting your production schedules without interruption.
We invite you to engage with our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific formulation needs. By collaborating with us, you can access specific COA data and route feasibility assessments that will help you optimize your supply chain and reduce manufacturing costs. Whether you are looking to improve the surface properties of your current products or develop next-generation low-VOC coatings, our expertise in fine chemical synthesis positions us as the ideal partner to support your innovation goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
