Advanced Copper Catalyst Technology for Scalable Production of High-Purity Pharmaceutical Intermediates
Advanced Copper Catalyst Technology for Scalable Production of High-Purity Pharmaceutical Intermediates
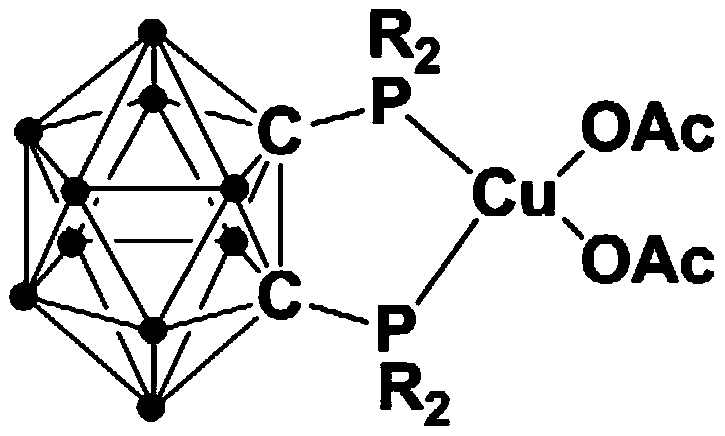
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN111635435A, which details the invention of a novel divalent copper complex containing a bisphosphine ortho-carborane ligand. This technology represents a paradigm shift for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering high-purity compounds through green chemistry principles. The core innovation lies in the unique structural integration of the robust carborane cage with phosphine ligands, creating a catalyst that is not only highly active but also remarkably stable in air. This stability addresses a critical pain point in industrial catalysis, where sensitive organometallic reagents often require inert atmosphere handling, thereby inflating operational costs and complexity.
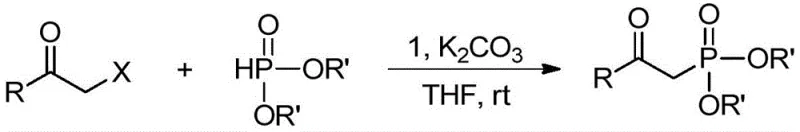
For R&D directors and process chemists, the implications of this patent are profound. The described copper complexes facilitate the one-step synthesis of beta-carbonylphosphine oxides, a class of compounds with extensive applications in medicinal chemistry and materials science. Unlike traditional methods that rely on harsh conditions or multi-step sequences involving expensive reagents like alkynes, this new approach utilizes inexpensive alpha-halo ketones and phosphites. The ability to achieve high yields and excellent selectivity under mild, room-temperature conditions positions this technology as a superior alternative for the commercial scale-up of complex polymer additives and electronic chemical precursors. By leveraging this intellectual property, chemical enterprises can streamline their production workflows while adhering to increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-carbonylphosphine oxides has been fraught with challenges that hinder large-scale adoption. Traditional pathways, such as the Arbuzov reaction or acylation of alkyl phosphonates, typically necessitate extreme reaction conditions, including strong acidic or basic environments that are difficult to control and pose safety risks. Furthermore, modern radical-based approaches, while innovative, often suffer from procedural complexity, requiring two distinct steps involving addition and hydration. These methods frequently depend on costly starting materials like polysubstituted alkenes or allenes and mandate the use of external oxidants. Such dependencies not only drive up the raw material costs but also generate significant waste streams, complicating downstream purification and waste treatment processes. For procurement managers, these inefficiencies translate into volatile supply chains and unpredictable production timelines.
The Novel Approach
In stark contrast, the methodology outlined in CN111635435A offers a streamlined, one-pot solution that elegantly bypasses these historical bottlenecks. The novel approach utilizes the newly developed bisphosphine ortho-carborane copper complexes to catalyze the direct coupling of alpha-halo ketones and phosphites. This reaction proceeds efficiently at room temperature, eliminating the energy expenditure associated with heating or cooling cycles. Crucially, the process does not require the participation of acids, bases, or oxidants, thereby simplifying the reaction matrix and reducing the formation of by-products. The result is a cleaner reaction profile with higher atom economy. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates, as the simplified workup allows for faster turnover from reactor to finished goods inventory.
Mechanistic Insights into Bisphosphine Carborane Copper Catalysis
The exceptional performance of this catalytic system can be attributed to the unique electronic and steric properties imparted by the ortho-carborane scaffold. The carborane cage acts as a robust, three-dimensional anchor that stabilizes the phosphine ligands against oxidation, a common degradation pathway for standard phosphines. This structural integrity ensures that the copper center remains active throughout the catalytic cycle, maintaining high turnover numbers even over extended reaction times. Mechanistically, the copper(II) center likely facilitates the activation of the carbon-halogen bond in the alpha-halo ketone, generating a reactive intermediate that readily undergoes nucleophilic attack by the phosphite. The rigidity of the ligand framework may also enforce a specific geometry around the metal center, enhancing the stereoselectivity of the transformation. This level of control is vital for producing chiral intermediates required in advanced drug synthesis.
From an impurity control perspective, the air stability of the catalyst is a game-changer. In conventional copper catalysis, trace oxygen can lead to the formation of inactive copper-oxo species or oxidized phosphine oxides, which act as catalyst poisons. The carborane-modified ligand resists this oxidation, ensuring that the active catalytic species persists. Consequently, the reaction mixture contains fewer metal-containing impurities and oxidized by-products, simplifying the purification burden. For quality assurance teams, this means the final product meets stringent purity specifications with less intensive chromatographic separation. The broad substrate scope demonstrated in the patent, accommodating various electron-donating and electron-withdrawing groups on the aromatic rings, further underscores the versatility of this mechanistic pathway, making it applicable to a wide array of fine chemical targets.
How to Synthesize Beta-Carbonylphosphine Oxides Efficiently
The practical implementation of this technology involves a straightforward protocol that is easily adaptable to existing manufacturing infrastructure. The process begins with the preparation of the catalyst itself, which is achieved through a simple lithiation-phosphination sequence followed by metallation with copper acetate. Once the catalyst is secured, the synthetic application involves dissolving the catalyst, the alpha-halo ketone substrate, and the phosphite ester in a common organic solvent. The detailed standardized synthesis steps see the guide below for precise molar ratios and timing optimization. This simplicity allows for rapid technology transfer from the laboratory to the pilot plant, minimizing the risk of scale-up failures.
- Dissolve the divalent copper complex catalyst, alpha-halo ketone substrate, and phosphite ester in an organic solvent such as THF or acetone.
- Add an inorganic base like potassium carbonate to the reaction mixture and stir at room temperature for 5 to 8 hours to ensure complete conversion.
- Concentrate the reaction mixture and purify the crude product via column chromatography using a petroleum ether and ethyl acetate eluent system.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this catalytic technology offers substantial strategic benefits for procurement and supply chain operations, primarily driven by the simplification of the manufacturing process. The elimination of harsh reagents and the ability to run reactions at ambient temperatures directly correlate to lower utility costs and reduced equipment wear and tear. Furthermore, the high stability of the catalyst means it can be stored and handled without specialized inert atmosphere equipment, lowering the barrier to entry for contract manufacturing organizations. These factors combine to create a more resilient supply chain capable of responding quickly to market demands for key intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is significant, driven by the removal of expensive reagents and energy-intensive conditions. By avoiding the need for strong acids, bases, or oxidants, the cost of goods sold (COGS) is drastically reduced. Additionally, the high yields reported in the patent minimize raw material waste, ensuring that every kilogram of input translates effectively into output. The simplified purification process further reduces solvent consumption and labor hours associated with chromatography. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain or retained as improved margin.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on sensitive reagents that have short shelf lives or require cold chain logistics. The air-stable nature of the bisphosphine carborane copper complexes mitigates these risks, allowing for bulk purchasing and long-term storage without degradation. This stability ensures that production schedules are not disrupted by reagent spoilage. Moreover, the use of commercially available and inexpensive starting materials like alpha-halo ketones and phosphites reduces dependency on niche suppliers, diversifying the sourcing strategy and enhancing overall supply security.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding chemical waste and emissions, this green chemistry approach provides a clear compliance advantage. The reaction generates minimal hazardous waste, and the absence of heavy metal oxidants simplifies effluent treatment. The one-pot nature of the synthesis reduces the number of unit operations required, making the process inherently easier to scale from kilograms to metric tons. This scalability ensures that the technology can meet the demands of global markets without compromising on environmental standards or safety protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic system. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into your current production portfolio.
Q: What are the stability characteristics of the bisphosphine carborane copper complex?
A: According to patent CN111635435A, the synthesized copper complexes exhibit high stability and can exist stably in air, which significantly simplifies storage and handling requirements compared to air-sensitive traditional catalysts.
Q: Does this catalytic method require harsh reaction conditions?
A: No, the process operates efficiently at room temperature without the need for strong acids, strong bases, or additional oxidants, offering a greener alternative to conventional Arbuzov reactions.
Q: What is the substrate scope for this catalytic system?
A: The catalyst demonstrates broad applicability, successfully facilitating the reaction of various alpha-halo ketones, including those with electron-donating and electron-withdrawing groups, with both diethyl and diphenyl phosphites.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Carbonylphosphine Oxide Supplier
The technological potential of the bisphosphine carborane copper complex is immense, offering a pathway to superior chemical intermediates with enhanced purity and sustainability profiles. At NINGBO INNO PHARMCHEM, we pride ourselves on being at the forefront of such innovations, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch meets the exacting standards required by the global pharmaceutical and agrochemical industries. We understand that consistency is key, and our robust quality management systems guarantee supply continuity.
We invite you to explore how this advanced catalytic technology can optimize your specific manufacturing needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your production volume and target specifications. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access not just to a product, but to a comprehensive solution that drives efficiency and value across your entire supply chain.
