Advanced Fluoroethylene Carbonate Production for High-Performance Battery Electrolyte Manufacturing
Advanced Fluoroethylene Carbonate Production for High-Performance Battery Electrolyte Manufacturing
The landscape of lithium-ion battery electrolyte additives is undergoing a significant transformation driven by the demand for higher safety and cycle stability, with patent CN114621177B introducing a groundbreaking preparation method for fluoroethylene carbonate. This specific technical disclosure addresses the critical limitations of traditional fluorination processes by utilizing a unique liquid-phase reaction system that employs divalent metal fluoride salts as both the fluorine source and the catalyst. Unlike conventional approaches that rely on hazardous gaseous fluorine or expensive phase transfer agents, this innovation enables a milder reaction environment that drastically reduces the formation of thermal decomposition byproducts. The strategic implementation of this synthesis route allows manufacturers to achieve superior conversion rates while maintaining the structural integrity of the sensitive carbonate ring during the fluorination process. Furthermore, the inherent ability to regenerate the metal fluoride salt from the chloride byproduct creates a closed-loop system that aligns perfectly with modern sustainable manufacturing goals. This report analyzes the technical merits and commercial implications of this advanced methodology for global supply chain decision-makers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for fluoroethylene carbonate often involve direct fluorination using elemental fluorine gas, which presents severe safety hazards and requires specialized equipment to handle the highly reactive and corrosive nature of the reagent. 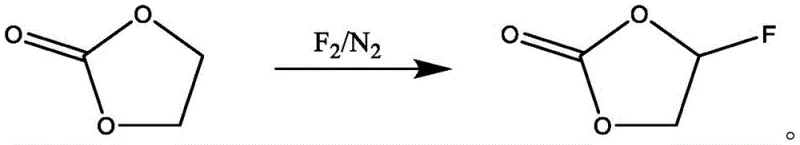 As illustrated in the reaction scheme, the use of fluorine gas necessitates rigorous inert gas protection and precise temperature control to prevent runaway reactions that can lead to catastrophic equipment failure or product degradation. Additionally, electrochemical fluorination methods, while avoiding direct gas handling, often suffer from complex downstream processing requirements and lower overall selectivity due to side reactions at the electrode surface. The reliance on alkali metal fluorides like potassium fluoride in liquid-phase methods typically mandates the addition of expensive crown ethers as phase transfer catalysts to solubilize the fluoride ions effectively. These catalysts are not only costly but also difficult to remove completely from the final product, potentially introducing ionic impurities that degrade battery performance over time. Consequently, the industry has long sought a method that balances safety, cost, and purity without compromising on reaction efficiency or scalability.
As illustrated in the reaction scheme, the use of fluorine gas necessitates rigorous inert gas protection and precise temperature control to prevent runaway reactions that can lead to catastrophic equipment failure or product degradation. Additionally, electrochemical fluorination methods, while avoiding direct gas handling, often suffer from complex downstream processing requirements and lower overall selectivity due to side reactions at the electrode surface. The reliance on alkali metal fluorides like potassium fluoride in liquid-phase methods typically mandates the addition of expensive crown ethers as phase transfer catalysts to solubilize the fluoride ions effectively. These catalysts are not only costly but also difficult to remove completely from the final product, potentially introducing ionic impurities that degrade battery performance over time. Consequently, the industry has long sought a method that balances safety, cost, and purity without compromising on reaction efficiency or scalability.
The Novel Approach
The patented methodology introduces a paradigm shift by utilizing divalent metal fluorides, such as cobalt or zinc fluoride, which function effectively in liquid-phase systems without the need for auxiliary phase transfer catalysts. This approach leverages the specific chemical affinity of divalent metals for chlorine atoms, facilitating a smooth halogen exchange reaction that proceeds under significantly milder thermal conditions compared to gas-phase alternatives. By eliminating the requirement for crown ethers, the process simplifies the purification workflow and reduces the risk of metal ion contamination in the final electrolyte additive. The reaction system is designed to operate at temperatures that prevent the thermal decomposition of the fluoroethylene carbonate product, thereby preserving the high selectivity required for premium battery applications. Moreover, the solubility characteristics of the divalent metal salts allow for the easy precipitation of the regenerated fluoride salt after the byproduct chloride is treated, enabling a practical recycling loop. This novel strategy effectively resolves the long-standing trade-off between reaction activity and process safety in fluorine chemistry manufacturing.
Mechanistic Insights into Divalent Metal Fluoride Catalyzed Fluorination
The core mechanism of this synthesis relies on the unique interaction between the divalent metal cation and the organic chloride substrate, which lowers the activation energy barrier for the fluorine-chlorine exchange reaction. Unlike monovalent alkali fluorides that require complexation to release fluoride ions, divalent metal fluorides possess an intrinsic ability to coordinate with the chlorine atom on the chloroethylene carbonate molecule. This coordination weakens the carbon-chlorine bond, allowing the fluoride ion to displace the chlorine atom more readily even in the absence of strong phase transfer catalysts. The reaction proceeds through a transition state where the metal center temporarily holds both the incoming fluoride and the outgoing chloride, ensuring a controlled substitution that minimizes random radical attacks. This controlled mechanism is crucial for preventing over-fluorination, which is a common issue in high-energy fluorination processes that leads to the formation of useless polyfluorinated impurities. Understanding this mechanistic nuance is essential for R&D teams aiming to optimize reaction parameters for maximum yield and minimal waste generation in industrial settings.
Impurity control is further enhanced by the specific solubility differences between the reactant metal fluoride and the byproduct metal chloride in the chosen solvent system. During the reaction, the metal chloride byproduct tends to remain in solution or form a distinct phase that can be easily separated from the organic product layer through filtration or distillation. This physical separation capability is vital for maintaining the high purity standards required for electronic grade chemicals used in sensitive lithium-ion battery formulations. The process parameters, such as reaction temperature and time, are tightly optimized to prevent the thermal degradation of the product, which can occur if the system is heated excessively during the exchange process. By maintaining the temperature within a narrow window, the formation of polymeric side products is suppressed, ensuring a clean impurity profile that simplifies downstream refining. This level of control over the reaction environment directly translates to a more consistent product quality that meets the stringent specifications of top-tier battery manufacturers.
How to Synthesize Fluoroethylene Carbonate Efficiently
The operational protocol for this synthesis involves a straightforward sequence of mixing, reacting, and separating steps that are designed for easy integration into existing chemical manufacturing infrastructure. Detailed standard operating procedures regarding specific molar ratios, solvent choices, and regeneration conditions are critical for replicating the high performance described in the technical data.
- Mix divalent metal fluoride salt with a polar aprotic solvent in a reaction vessel to form a homogeneous mixture.
- Add chloroethylene carbonate to the mixture under inert gas protection and maintain specific temperature conditions for fluorination.
- Separate the crude fluoroethylene carbonate via distillation and regenerate the metal fluoride salt byproduct for recycling.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this manufacturing route offers substantial advantages by removing the dependency on expensive and hard-to-source phase transfer catalysts that traditionally inflate the bill of materials for fluoroethylene carbonate production. The elimination of crown ethers not only reduces direct material costs but also simplifies the supply chain by reducing the number of specialized reagents that need to be sourced and managed. Furthermore, the ability to regenerate the fluorine source from the byproduct salt creates a circular economy model within the plant, significantly lowering the consumption of raw fluorine-containing materials over the long term. This reduction in raw material turnover decreases the frequency of procurement cycles and mitigates the risk associated with price volatility in the global fluorine chemical market. Supply chain managers can benefit from a more predictable cost structure and reduced exposure to external market fluctuations for critical reagents.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the elimination of noble or complex catalysts and the implementation of a regenerable fluorine source system that minimizes waste disposal costs. By avoiding the use of crown ethers, manufacturers save on high-value additives that contribute significantly to the overall production expense without adding value to the final product quality. The regeneration of the metal fluoride salt from the chloride byproduct further extends the utility of the initial fluorine investment, effectively lowering the cost per kilogram of the final active ingredient. Additionally, the milder reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility bills and a smaller carbon footprint for the manufacturing facility. These combined factors result in a leaner production cost structure that enhances competitiveness in the global electrolyte additive market.
- Enhanced Supply Chain Reliability: The reliance on readily available divalent metal salts and common organic solvents ensures a robust supply chain that is less susceptible to disruptions compared to processes requiring specialized fluorine gas handling infrastructure. The liquid-phase nature of the reaction allows for the use of standard chemical reactors, reducing the need for custom-engineered equipment that often has long lead times for procurement and installation. This flexibility enables manufacturers to scale production capacity more rapidly in response to market demand without being bottlenecked by equipment availability or specialized operator training requirements. The stability of the raw materials also simplifies storage and logistics, as there is no need for high-pressure gas cylinders or cryogenic storage systems that pose safety and regulatory challenges. Consequently, the supply of fluoroethylene carbonate becomes more resilient and capable of meeting the continuous demands of large-scale battery production lines.
- Scalability and Environmental Compliance: The liquid-phase process is inherently easier to scale from laboratory to commercial production volumes due to the well-understood engineering principles governing stirred tank reactors and distillation columns. The absence of hazardous fluorine gas eliminates the need for complex scrubbing systems and emergency containment protocols, simplifying environmental compliance and reducing the regulatory burden on the manufacturing site. Waste generation is minimized through the recycling of the metal fluoride salt, aligning the process with green chemistry principles and reducing the volume of hazardous waste requiring treatment or disposal. The lower operating temperatures also reduce the risk of thermal runaway incidents, enhancing overall plant safety and reducing insurance premiums associated with high-risk chemical operations. This combination of scalability and safety makes the technology highly attractive for investment and long-term industrial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this divalent metal fluoride synthesis route for fluoroethylene carbonate. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for technical evaluation.
Q: Why are divalent metal fluorides preferred over alkali metal fluorides in this synthesis?
A: Divalent metal fluorides exhibit lower solubility compared to their chloride byproducts, facilitating easier separation and regeneration without requiring expensive phase transfer catalysts like crown ethers.
Q: How does the regeneration process contribute to cost efficiency?
A: The byproduct metal chloride salt can be fluorinated back into the metal fluoride salt using hydrogen fluoride, allowing the fluorine source to be recycled multiple times and significantly reducing raw material waste.
Q: What impact does this method have on product purity for battery applications?
A: By operating at milder temperatures and avoiding high-activity catalysts that cause decomposition, this method minimizes polyfluorinated byproducts, resulting in higher selectivity and purity essential for stable SEI film formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroethylene Carbonate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting this advanced synthesis technology to deliver high-purity fluoroethylene carbonate that meets the rigorous demands of the modern energy storage industry. Our engineering team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of electrolyte additive performs consistently in high-performance lithium-ion cells. Our commitment to process excellence means that we can leverage the benefits of this catalyst-free, regenerable route to offer a product with superior impurity profiles and competitive pricing structures. Clients can trust in our ability to manage the complexities of fluorine chemistry while delivering a reliable supply of critical battery materials.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain resilience and product performance. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how our manufacturing efficiencies translate into value for your battery production operations. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your specific application requirements. Our team is ready to provide the technical support and commercial flexibility needed to secure your position in the rapidly evolving energy market. Let us collaborate to drive the next generation of battery technology forward with superior materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
