Advanced Low-Temperature Synthesis of 2-Bromomalonaldehyde for High-Performance Liquid Crystal and API Manufacturing
The landscape of fine chemical manufacturing for heterocyclic precursors is undergoing a significant transformation driven by the need for higher purity and process stability. Patent CN115894192A introduces a groundbreaking synthetic methodology for 2-bromomalonaldehyde, a critical building block extensively utilized in the production of pyrimidine-based liquid crystals and various active pharmaceutical ingredients. This innovation addresses the longstanding challenge of intermediate instability that has historically plagued the commercial production of this valuable compound. By shifting from conventional room-temperature deprotection to a rigorously controlled low-temperature protocol coupled with in-situ salt formation, the disclosed technology effectively mitigates the kinetic drivers of polymerization. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a pivotal shift towards more robust and scalable manufacturing protocols that ensure consistent quality.
The implementation of this advanced synthesis route offers substantial implications for the supply chain continuity of high-performance materials. Traditional processes often suffer from batch-to-batch variability due to the unpredictable nature of malonaldehyde polymerization, leading to supply bottlenecks and increased costs associated with waste disposal and reprocessing. The new method described in the patent stabilizes the reactive intermediate as a sodium salt, allowing for safer handling and storage prior to the final bromination step. This strategic modification not only enhances the overall process safety profile but also significantly streamlines the downstream purification requirements. As the demand for high-purity OLED material and liquid crystal components continues to surge, adopting such stabilized synthetic pathways becomes essential for maintaining competitive advantage in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-bromomalonaldehyde has relied on the direct deprotection of 1,1,3,3-tetramethoxypropane followed immediately by bromination and concentration. In these legacy processes, the deprotection step is typically conducted at ambient temperatures, which inadvertently triggers the rapid enolization and subsequent polymerization of the generated free malonaldehyde. This uncontrolled side reaction creates a complex mixture of oligomers and tars, drastically reducing the availability of the monomeric species required for the subsequent bromination. Furthermore, the necessity to remove methanol byproducts via heating under negative pressure exacerbates the thermal stress on the molecule, accelerating degradation and resulting in a deeply colored, low-purity crude product. Consequently, traditional yields rarely exceed 60%, imposing a heavy economic burden on manufacturers through inefficient raw material utilization and extensive chromatographic purification needs.
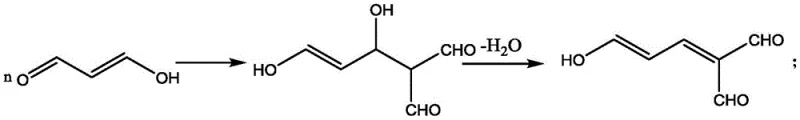
The visual representation of the polymerization pathway highlights the inherent instability of the free aldehyde form under standard processing conditions. The conjugated system formed during polymerization is thermodynamically favored at higher temperatures, making the conventional concentration step a critical failure point in the process. This instability not only compromises the yield but also introduces difficult-to-remove impurities that can interfere with downstream coupling reactions in liquid crystal synthesis. For procurement managers focused on cost reduction in liquid crystal material manufacturing, the inefficiency of the old method translates directly into higher unit costs and unreliable delivery schedules due to the frequent need for re-runs or extensive rework.
The Novel Approach
In stark contrast, the novel approach detailed in the patent employs a strategic intervention by converting the unstable free malonaldehyde into its corresponding sodium salt immediately after generation. By conducting the initial acid hydrolysis at temperatures not exceeding 5°C, the kinetic energy available for polymerization is minimized, preserving the integrity of the carbon backbone. The subsequent neutralization with sodium hydroxide locks the molecule into a stable ionic form that is resistant to the thermal stresses of vacuum concentration. This allows for the efficient removal of methanol without the risk of product degradation, a feat impossible with the free aldehyde. The isolation of the solid sodium salt via acetone crystallization provides a pure, stable intermediate that can be stored or transported before the final bromination step, decoupling the two most sensitive stages of the synthesis.
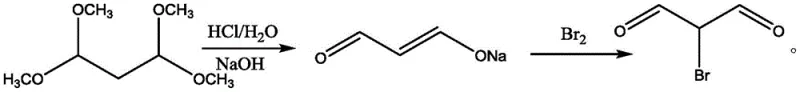
The final bromination of the isolated sodium salt proceeds under mild conditions without generating methanol, thereby eliminating the need for a second concentration step that could induce polymerization. The product precipitates directly from the reaction mixture upon cooling, allowing for simple filtration to obtain an off-white solid with exceptional purity levels reaching 99.9%. This streamlined workflow not only boosts yields to over 90% but also ensures a consistent physical form that is easier to handle in automated dosing systems. For supply chain heads concerned with the commercial scale-up of complex heterocyclic intermediates, this decoupled process offers superior flexibility and risk mitigation, ensuring that production targets can be met with predictable reliability.
Mechanistic Insights into Low-Temperature Salt Stabilization
The core mechanistic advantage of this synthesis lies in the manipulation of the equilibrium between the free aldehyde and its enol forms through pH and temperature control. At low temperatures (0-5°C) and acidic pH, the hydrolysis of the acetal protecting groups proceeds selectively to release malonaldehyde, but the low thermal energy prevents the molecules from overcoming the activation barrier for nucleophilic attack on neighboring carbonyls. Once the free aldehyde is generated, the immediate addition of a stoichiometric amount of sodium hydroxide shifts the equilibrium towards the enolate anion. This anionic species is resonance-stabilized and lacks the electrophilic character of the neutral aldehyde, rendering it inert to self-condensation reactions. This chemical 'masking' strategy is a sophisticated application of physical organic chemistry principles to solve a practical engineering problem.
Furthermore, the choice of acetone as an anti-solvent for crystallizing the sodium salt is mechanistically significant due to its ability to reduce the dielectric constant of the medium, promoting the precipitation of the ionic species while keeping organic impurities in solution. This step acts as a powerful purification checkpoint before the final halogenation. When the solid salt is redissolved for bromination, the reaction occurs cleanly via electrophilic substitution at the alpha-position. Since the byproduct is sodium bromide rather than volatile methanol, there is no driving force or requirement for thermal concentration. The entire sequence is designed to keep the reactive centers protected or stabilized until the very last moment, ensuring that the final product emerges with minimal structural defects or oligomeric contaminants.
How to Synthesize 2-Bromomalonaldehyde Efficiently
The operational protocol for this synthesis requires precise control over reaction parameters to fully realize the benefits of the patented method. The process begins with the careful addition of dilute hydrochloric acid to the acetal precursor under vigorous stirring to ensure uniform heat dissipation. Maintaining the temperature below 5°C throughout the deprotection phase is critical, as even slight deviations can initiate the formation of polymeric byproducts that are difficult to reverse. Following neutralization, the vacuum concentration step must be monitored to ensure that the temperature does not exceed 50°C, preserving the stability of the sodium salt. The crystallization step serves as a key quality gate, where the purity of the intermediate can be visually assessed by the color of the crystals, with off-white indicating high quality.
- Hydrolyze 1,1,3,3-tetramethoxypropane using dilute acid (0.1-1M HCl) at temperatures not exceeding 5°C to generate free malonaldehyde while suppressing polymerization.
- Neutralize the reaction mixture with sodium hydroxide to form the stable malonaldehyde sodium salt, followed by vacuum concentration to remove methanol byproducts without thermal degradation.
- Induce crystallization of the sodium salt by adding acetone to the concentrate, then filter to isolate the stable solid intermediate.
- React the isolated malonaldehyde sodium salt with elemental bromine in an aqueous medium at controlled low temperatures (0-25°C) to obtain the final 2-bromomalonaldehyde product via filtration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this stabilized synthesis route offers profound advantages for organizations managing the procurement of high-value intermediates. The primary benefit is the drastic reduction in process variability, which translates to more reliable lead times and consistent inventory planning. By eliminating the unpredictable polymerization side reactions, manufacturers can guarantee higher throughput per batch without the need for oversized reactors to compensate for low yields. This efficiency gain directly impacts the cost structure, allowing for more competitive pricing models without sacrificing margin. Additionally, the simplified workup procedure reduces the consumption of solvents and energy, aligning with modern sustainability goals and reducing the environmental compliance burden associated with waste treatment.
- Cost Reduction in Manufacturing: The elimination of thermal degradation steps and the significant increase in yield mean that less raw material is required to produce the same amount of final product. The avoidance of complex purification techniques such as column chromatography, which are often necessary to remove polymeric tars in conventional methods, further lowers the operational expenditure. The ability to isolate a stable solid intermediate also reduces the risk of batch loss, ensuring that capital invested in raw materials is fully converted into saleable product.
- Enhanced Supply Chain Reliability: The decoupling of the deprotection and bromination steps allows for greater flexibility in production scheduling. Manufacturers can stockpile the stable sodium salt intermediate, creating a buffer against supply chain disruptions for the final bromination reagents. This inventory strategy ensures continuous availability of the final API intermediate even during periods of raw material volatility. The robustness of the process also reduces the likelihood of failed batches, providing customers with a dependable source of supply for their critical manufacturing lines.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of high-temperature concentration steps make this process inherently safer and easier to scale from pilot plant to full commercial production. The reduction in solvent usage and the generation of simpler aqueous waste streams facilitate easier treatment and disposal, helping companies meet increasingly stringent environmental regulations. This green chemistry approach not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key factor in vendor selection for major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing workflows. The answers are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: Why is the traditional room-temperature synthesis of 2-bromomalonaldehyde problematic for industrial scale-up?
A: Traditional methods involve deprotecting 1,1,3,3-tetramethoxypropane at room temperature, which generates free malonaldehyde. This intermediate is highly unstable and prone to rapid polymerization, leading to yields below 60% and significant impurity profiles that complicate downstream purification.
Q: How does the sodium salt intermediate strategy improve product purity and yield?
A: By converting the unstable free malonaldehyde into its sodium salt form immediately after generation, the reactive aldehyde groups are stabilized. This allows for the removal of methanol byproducts under mild vacuum conditions without triggering the thermal polymerization that typically degrades the product during concentration steps.
Q: What are the critical temperature controls required for this synthesis?
A: Strict temperature control is essential, particularly during the initial deprotection step which must be maintained at or below 5°C to prevent early polymerization. The final bromination step also benefits from cooling to 0°C prior to filtration to maximize crystal recovery and maintain the off-white appearance of the high-purity solid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromomalonaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the success of downstream pharmaceutical and electronic material applications. Our technical team has thoroughly analyzed the synthetic pathway described in CN115894192A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to implement this low-temperature stabilization technology effectively. We are committed to delivering 2-bromomalonaldehyde with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required for liquid crystal and API synthesis. Our facility is equipped to handle the precise temperature controls and specialized crystallization steps necessary to replicate the high yields and purity reported in the patent.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to our stabilized grade of 2-bromomalonaldehyde. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your specific volume requirements. Our goal is to function not just as a vendor, but as a strategic partner in your chemical development journey, providing the reliability and quality assurance needed to accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
