Advanced Divalent Copper Catalysts for Efficient Beta-Carbonyl Phosphine Oxide Manufacturing
Advanced Divalent Copper Catalysts for Efficient Beta-Carbonyl Phosphine Oxide Manufacturing
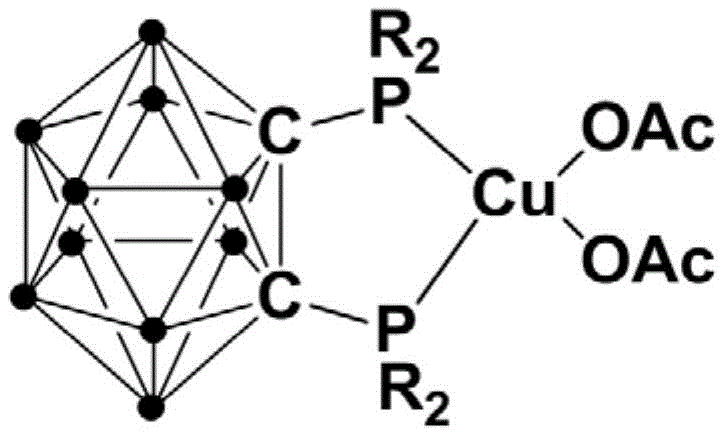
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the need for more sustainable and efficient synthetic methodologies. A pivotal development in this domain is detailed in patent CN111635435B, which introduces a novel class of divalent copper complexes featuring bisphosphine ortho-carborane ligands. These complexes represent a breakthrough in catalytic efficiency, specifically designed for the synthesis of beta-carbonyl phosphine oxide compounds. As a leading entity in the fine chemical sector, we recognize that the ability to produce high-purity pharmaceutical intermediates under mild conditions is paramount for modern supply chains. The structural integrity and electronic properties of these carborane-based ligands provide a unique platform for stabilizing the copper center, thereby enabling reactions that were previously plagued by harsh conditions or poor selectivity.
The strategic importance of beta-carbonyl phosphine oxides cannot be overstated, as they serve as critical building blocks in the synthesis of biologically active molecules, including potential anticancer agents and bone metabolism regulators. Furthermore, these compounds function as essential ligands in coordination chemistry, facilitating the development of new catalytic systems. The innovation described in the patent addresses the long-standing challenge of accessing these valuable motifs through a streamlined, one-pot process. By leveraging the robust nature of the ortho-carborane cage, the resulting copper complexes offer exceptional stability, allowing them to persist in air without rapid degradation. This characteristic is a game-changer for industrial applications, where the handling of air-sensitive reagents often necessitates costly inert atmosphere infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-carbonyl phosphine oxides has relied heavily on the Arbuzov reaction or acylation processes conducted under aggressive acidic or basic conditions. These traditional pathways are fraught with operational difficulties, including the requirement for stringent temperature control and the generation of substantial corrosive waste streams. Moreover, the selectivity of these methods is often compromised, leading to complex mixtures that require extensive and costly purification steps to isolate the desired product. In recent years, alternative strategies involving phosphorus-centered radical additions have emerged, yet these too present significant hurdles. Such methods typically mandate a multi-step sequence involving both addition and hydration phases, drastically increasing the processing time and resource consumption.
Furthermore, conventional radical approaches frequently depend on expensive and specialized reagents such as alkynes, multi-substituted alkenes, or allenes, which drives up the raw material costs significantly. The necessity for external oxidants in these radical pathways adds another layer of complexity and safety concern, particularly when scaling up to commercial production volumes. The cumulative effect of these limitations is a manufacturing process that is economically inefficient and environmentally burdensome. For procurement managers and supply chain directors, these factors translate into higher lead times, increased inventory costs for hazardous reagents, and greater regulatory compliance overhead. The industry urgently requires a paradigm shift towards methodologies that utilize commodity-grade starting materials while eliminating the need for hazardous auxiliaries.
The Novel Approach
The technology disclosed in patent CN111635435B offers a compelling solution by enabling the direct coupling of alpha-halogenated ketones with phosphite esters at room temperature. This novel approach bypasses the need for strong acids, bases, or oxidants, relying instead on the unique catalytic activity of the divalent copper-carborane complex. The reaction proceeds with excellent selectivity and high yields, transforming simple, readily available substrates into high-value intermediates in a single operational step. This simplification of the synthetic route is a critical advantage for cost reduction in pharmaceutical intermediate manufacturing, as it minimizes unit operations and reduces solvent consumption. The ability to run the reaction at ambient temperature further contributes to energy savings, aligning with global sustainability goals.
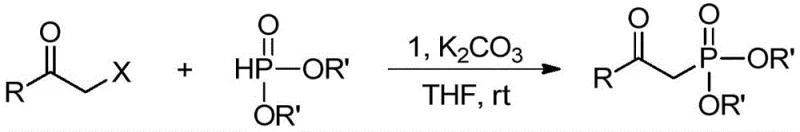
From a supply chain perspective, the use of alpha-halogenated ketones and dialkyl phosphites as feedstocks ensures a reliable and continuous supply of raw materials, as these are produced on a massive scale globally. The robustness of the catalyst allows for flexible batch scheduling without the fear of rapid deactivation, enhancing overall plant throughput. For R&D teams, the broad substrate scope demonstrated by this system means that a wide array of derivatives can be accessed using the same core protocol, accelerating the drug discovery pipeline. The elimination of harsh reagents also simplifies the workup procedure, often requiring only concentration and column chromatography, which streamlines the path from laboratory bench to pilot plant. This methodology exemplifies how advanced ligand design can unlock superior catalytic performance, turning a complex chemical transformation into a routine industrial process.
Mechanistic Insights into Cu-Carborane Catalyzed Coupling
The exceptional performance of this catalytic system is rooted in the sophisticated design of the bisphosphine ortho-carborane ligand. The ortho-carborane cage acts as a rigid, electron-withdrawing scaffold that imparts significant steric bulk and electronic stability to the phosphorus donor atoms. When coordinated to the divalent copper center, this ligand architecture creates a highly defined coordination environment that facilitates the activation of the carbon-halogen bond in the alpha-halogenated ketone. The rigidity of the carborane framework prevents unfavorable conformational changes that could lead to catalyst decomposition, thereby extending the catalytic lifetime. This stability is crucial for maintaining consistent reaction rates over extended periods, ensuring reproducible results across different batches.
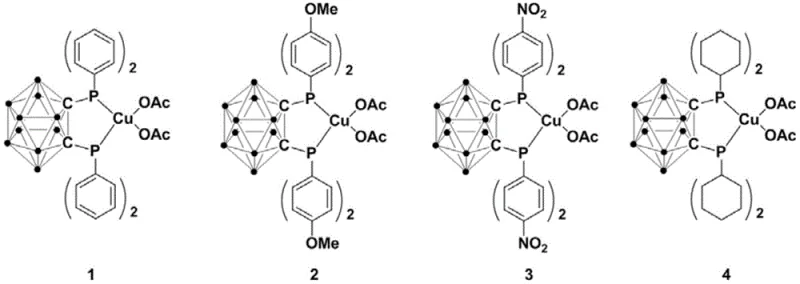
Impurity control is another critical aspect where this mechanism excels. The high selectivity of the copper complex minimizes side reactions such as over-alkylation or hydrolysis of the phosphite ester, which are common pitfalls in traditional methods. The specific electronic tuning provided by the substituents on the phosphorus atoms (such as phenyl, methoxy-phenyl, or nitro-phenyl groups) allows for fine-tuning the Lewis acidity of the metal center. This tunability ensures that the catalyst can accommodate various electronic environments on the ketone substrate without compromising yield. For quality control laboratories, this translates to cleaner crude reaction profiles and reduced levels of difficult-to-remove impurities. The mechanistic pathway likely involves the formation of a copper-phosphorus species that undergoes oxidative addition with the haloketone, followed by reductive elimination to forge the carbon-phosphorus bond, regenerating the active catalyst species in the process.
How to Synthesize Divalent Copper Bisphosphine Ortho-Carborane Complex Efficiently
The preparation of the catalyst itself is designed for operational simplicity, utilizing a sequential one-pot strategy that avoids the isolation of unstable intermediates. The process begins with the lithiation of ortho-carborane, followed immediately by phosphorylation and finally metallation with copper acetate. This telescoped approach reduces solvent usage and handling time, making it highly suitable for scale-up. The resulting complexes are air-stable solids that can be stored and weighed under ambient conditions, removing the need for glovebox techniques during the subsequent catalytic application. Detailed standardized synthesis steps for the catalyst preparation are provided in the guide below.
- Dropwise addition of n-BuLi solution to an ortho-carborane ether solution at low temperature (-5 to 5°C) followed by stirring for 20-40 minutes.
- Warm the reaction mixture to room temperature and stir for an additional 20-40 minutes before adding the halogenated phosphine reagent.
- Add copper acetate (Cu(OAc)2) to the system, react for 3-6 hours at room temperature, then isolate the product via solvent removal and ether washing.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this catalytic technology offers profound strategic benefits for organizations focused on optimizing their chemical supply chains and reducing manufacturing overheads. The shift from multi-step, reagent-intensive processes to a streamlined, room-temperature catalytic cycle fundamentally alters the cost structure of producing beta-carbonyl phosphine oxides. By eliminating the dependency on expensive alkynes and oxidants, companies can achieve substantial cost savings on raw material procurement. Furthermore, the mild reaction conditions reduce the energy load on production facilities, contributing to lower utility bills and a smaller carbon footprint. These efficiencies compound when viewed over the lifecycle of a product, making the technology highly attractive for long-term commercial partnerships.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic simplification of the synthetic route. Traditional methods often require stoichiometric amounts of hazardous reagents and multiple purification stages, each adding cost and time. In contrast, this catalytic method utilizes sub-stoichiometric amounts of a reusable, stable catalyst and inexpensive commodity chemicals. The removal of strong acid or base requirements also mitigates the costs associated with corrosion-resistant equipment and neutralization waste treatment. Consequently, the overall cost of goods sold (COGS) for the final intermediate is significantly lowered, improving margin potential for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Alpha-halogenated ketones and phosphite esters are produced by numerous global suppliers, reducing the risk of single-source bottlenecks. Additionally, the air stability of the copper catalyst eliminates the logistical complexities and costs associated with shipping and storing pyrophoric or moisture-sensitive reagents. This reliability ensures consistent production schedules and reduces the likelihood of delays caused by reagent degradation or availability issues. For supply chain heads, this means a more predictable and robust procurement strategy that can withstand market fluctuations.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its one-pot nature and tolerance to ambient conditions, which simplifies the transition from laboratory to commercial scale. The absence of toxic oxidants and the generation of minimal waste streams align with increasingly stringent environmental regulations. This compliance advantage reduces the regulatory burden and potential fines associated with hazardous waste disposal. Moreover, the high atom economy of the reaction ensures that a greater proportion of input materials end up in the final product, minimizing waste generation at the source. This makes the technology not only commercially viable but also environmentally sustainable, a key factor for modern corporate responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic system. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this chemistry into their existing workflows. The answers highlight the versatility and robustness of the copper-carborane complexes in real-world applications.
Q: What are the stability characteristics of the bisphosphine ortho-carborane copper complex?
A: The complex exhibits high stability and can exist stably in air, which simplifies storage and handling compared to air-sensitive organometallic catalysts.
Q: Does this catalytic system require harsh reaction conditions or oxidants?
A: No, the system operates efficiently at room temperature without the need for strong acids, bases, or external oxidants, utilizing inexpensive alpha-halogenated ketones and phosphites.
Q: What is the substrate scope for this catalytic application?
A: The catalyst demonstrates a broad substrate scope, effectively converting various alpha-halogenated ketones including those with electron-donating and electron-withdrawing groups into beta-carbonyl phosphine oxides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Carbonyl Phosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the divalent copper-carborane catalytic system can be seamlessly translated into industrial reality. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of intermediate meets the exacting standards required by the global pharmaceutical industry. We understand that the successful commercialization of complex fine chemicals requires not just a recipe, but a partner who can navigate the intricacies of process safety, waste management, and regulatory compliance with expertise.
We invite you to engage with our technical procurement team to discuss how this advanced catalytic route can optimize your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your production volume. We encourage potential partners to reach out for specific COA data and route feasibility assessments to validate the performance of these high-purity intermediates in your downstream processes. Let us collaborate to drive efficiency and innovation in your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
