Advanced Synthetic Route for 1-(3-Methoxypropyl)-4-Piperidylamine: Enhancing Purity and Scalability for Prucalopride Production
The pharmaceutical landscape for gastrointestinal motility disorders has been significantly advanced by the development of Prucalopride, a selective 5-HT4 receptor agonist. As detailed in patent CN103804281A, the efficient production of its critical precursor, 1-(3-methoxypropyl)-4-piperidylamine, remains a pivotal challenge for generic manufacturers and innovators alike. This specific intermediate serves as the structural cornerstone for the dihydrobenzofuran carboxylic acid derivative class of drugs, necessitating a synthesis route that balances high stereochemical integrity with economic viability. Traditional methods have often struggled to meet the rigorous purity standards required for final API registration, prompting the industry to seek alternative pathways that mitigate safety risks while maximizing yield. The technology disclosed in this patent represents a paradigm shift, moving away from hazardous high-pressure reductions toward a more controlled, step-wise functional group transformation strategy.
For R&D directors evaluating process robustness, the ability to source a reliable pharmaceutical intermediates supplier who understands these nuanced synthetic challenges is paramount. The transition from legacy manufacturing protocols to this novel three-step sequence offers a tangible solution to the bottleneck of intermediate availability. By leveraging a pathway that begins with readily available 4-piperidone derivatives, the process inherently reduces the complexity of the supply chain upstream. This report analyzes the technical merits of this approach, demonstrating how it aligns with modern green chemistry principles while delivering the high-purity prucalopride intermediate essential for downstream coupling reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-(3-methoxypropyl)-4-piperidylamine has relied heavily on direct reduction strategies that impose severe constraints on industrial feasibility. One common legacy approach involves the direct reduction of substituted piperidones under high-pressure hydrogenation conditions. While conceptually simple, this method demands specialized autoclave equipment capable of withstanding extreme pressures, introducing significant capital expenditure and safety liabilities for manufacturing sites. Furthermore, the selectivity of direct reduction can be problematic, often leading to over-reduction or the formation of secondary amine byproducts that are notoriously difficult to separate from the desired primary amine. Another prevalent method utilizes powerful reducing agents such as lithium aluminum hydride (LiAlH4). Although effective in a laboratory setting, LiAlH4 is pyrophoric, moisture-sensitive, and generates substantial amounts of aluminum waste, creating a heavy burden on environmental compliance and waste treatment protocols. These factors collectively drive up the cost of goods sold (COGS) and introduce volatility into the commercial scale-up of complex piperidine derivatives.
The Novel Approach
In stark contrast, the methodology outlined in CN103804281A introduces a refined, three-step sequence that circumvents these hazards through careful functional group manipulation. Instead of forcing a direct conversion, the new route first reduces the ketone to an alcohol using mild hydride sources, then activates the hydroxyl group as a tosylate, and finally employs a Gabriel synthesis to install the amine functionality. This strategic detour allows reactions to proceed at atmospheric pressure and near-ambient temperatures, drastically simplifying the engineering requirements for production. The use of sodium borohydride in the initial step provides a safer, more controllable reduction profile compared to aluminum-based reagents. Moreover, the Gabriel synthesis step is renowned for its specificity in generating primary amines without the risk of poly-alkylation, ensuring a cleaner impurity profile from the outset. This approach not only enhances operator safety but also streamlines the purification process, making it an ideal candidate for cost reduction in API manufacturing.
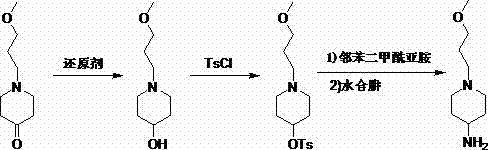
Mechanistic Insights into the Gabriel Synthesis Strategy
The core innovation of this process lies in the application of the Gabriel synthesis mechanism for the final amination step, a technique that offers superior control over nitrogen substitution patterns. In the third stage of the synthesis, the activated tosylate intermediate reacts with potassium phthalimide. Mechanistically, the phthalimide anion acts as a potent nucleophile, displacing the tosylate leaving group via an SN2 mechanism. This step is critical because the bulky phthalimide group sterically hinders further alkylation, effectively preventing the formation of secondary or tertiary amine impurities that plague direct amination routes. The result is a protected amine intermediate (the phthalimido-piperidine derivative) that is chemically stable and easy to isolate. This stability allows for rigorous quality control checks before the final deprotection step, ensuring that only the correct molecular scaffold proceeds to the final stage.
Following the substitution, the phthalimide protecting group is cleaved using hydrazine hydrate under reflux conditions. This hydrazinolysis reaction is highly specific, breaking the imide bonds to release the free primary amine and precipitating phthalhydrazide as a solid byproduct. The precipitation of phthalhydrazide is a distinct advantage for purification, as it can be removed via simple filtration, leaving the desired amine in the filtrate. This inherent "self-purifying" aspect of the mechanism significantly reduces the need for complex chromatographic separations, which are often cost-prohibitive at large scales. For procurement teams, this mechanistic elegance translates directly into higher overall yields and reduced solvent consumption, addressing the critical need for reducing lead time for high-purity pharmaceutical intermediates while maintaining strict adherence to regulatory impurity limits.
How to Synthesize 1-(3-Methoxypropyl)-4-Piperidylamine Efficiently
The execution of this synthetic route requires precise control over reaction stoichiometry and temperature to maximize the efficiency of each transformation. The process begins with the reduction of the ketone, followed by activation and substitution, culminating in the hydrolysis step. Each stage has been optimized in the patent examples to demonstrate robustness across different solvent systems, providing flexibility for process engineers to adapt the method to their existing infrastructure. The following guide outlines the standardized operational framework derived from the patent data, serving as a blueprint for pilot and commercial implementation.
- Reduce 1-(3-methoxypropyl)-4-piperidone using sodium borohydride in ethanol to form the corresponding alcohol intermediate.
- Convert the alcohol to a leaving group by reacting with Tosyl chloride and triethylamine in dichloromethane to form the tosylate ester.
- Perform nucleophilic substitution with potassium phthalimide followed by hydrazine hydrate hydrolysis to yield the final primary amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for supply chain resilience and cost management. By shifting away from high-pressure and hazardous reagent-dependent processes, manufacturers can significantly lower their operational overheads. The elimination of specialized high-pressure reactors reduces both capital investment and maintenance costs, while the use of non-pyrophoric reagents lowers insurance premiums and safety training requirements. Furthermore, the reliance on commodity chemicals such as sodium borohydride, tosyl chloride, and hydrazine hydrate ensures that raw material sourcing remains stable and unaffected by the supply volatility often seen with exotic catalysts. This stability is crucial for long-term contract manufacturing agreements where price consistency is a key negotiation point.
- Cost Reduction in Manufacturing: The replacement of lithium aluminum hydride with sodium borohydride represents a direct material cost saving, as the former is significantly more expensive and requires stringent anhydrous conditions that increase utility costs. Additionally, the mild reaction temperatures (often ranging from 0°C to ambient) reduce the energy load required for heating or cryogenic cooling compared to extreme condition processes. The simplified workup procedures, particularly the filtration of solid byproducts in the final step, minimize solvent usage and waste disposal fees, contributing to a leaner manufacturing cost structure without compromising on yield.
- Enhanced Supply Chain Reliability: The raw materials utilized in this three-step sequence are bulk commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. Unlike processes that rely on proprietary catalysts or custom-synthesized reagents, the inputs for this route—such as ethanol, dichloromethane, and triethylamine—are standard inventory items for most chemical distributors. This ubiquity ensures that production schedules are less likely to be disrupted by raw material shortages, providing procurement managers with greater confidence in meeting delivery deadlines for downstream API synthesis.
- Scalability and Environmental Compliance: The absence of high-pressure steps and heavy metal catalysts simplifies the regulatory approval process for new manufacturing sites. The process generates waste streams that are easier to treat, primarily consisting of organic salts and solvents that can be recovered or incinerated according to standard protocols. This environmental compatibility facilitates faster technology transfer between sites and supports the industry's broader goals of sustainable manufacturing. The robustness of the chemistry allows for seamless scaling from kilogram-level R&D batches to multi-ton commercial production runs, ensuring a continuous supply of critical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this Prucalopride intermediate. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: Why is the Gabriel synthesis route preferred over direct high-pressure amination for this intermediate?
A: Direct high-pressure amination often suffers from harsh reaction conditions, lower yields, and difficulties in controlling selectivity. The Gabriel synthesis route described in patent CN103804281A utilizes mild temperatures and avoids expensive or dangerous catalysts like lithium aluminum hydride, resulting in higher product purity and safer industrial handling.
Q: What are the key cost drivers eliminated in this new synthetic method?
A: This method eliminates the need for specialized high-pressure equipment required for direct reduction and replaces hazardous, costly reducing agents like lithium aluminum hydride with standard sodium borohydride. Additionally, the use of commodity reagents such as Tosyl chloride and phthalimide ensures stable raw material pricing and supply chain reliability.
Q: How does this process ensure scalability for commercial API manufacturing?
A: The process operates under mild thermal conditions (often near room temperature or mild reflux) and uses common organic solvents like ethanol and dichloromethane. This eliminates the engineering complexities associated with high-pressure hydrogenation, allowing for straightforward scale-up from pilot plants to multi-ton commercial production facilities without significant capital expenditure on specialized reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(3-Methoxypropyl)-4-Piperidylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of gastrointestinal therapeutics depends on the uninterrupted supply of high-quality intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 1-(3-methoxypropyl)-4-piperidylamine meets the exacting standards required for GMP API synthesis. Our commitment to process excellence means we can consistently deliver the purity profiles necessary to streamline your downstream regulatory filings.
We invite you to collaborate with us to optimize your supply chain for Prucalopride production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this advanced synthetic route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey toward efficient and compliant drug manufacturing.
