Advanced Isoquinoline Ligand Complexes for High-Purity Asymmetric Hydrogenation and Commercial Scale-Up
The pharmaceutical and fine chemical industries continuously face the critical challenge of synthesizing optically pure compounds with high efficiency and minimal environmental impact. Patent CN102417523A introduces a groundbreaking advancement in this domain through the development of novel nitrogen heterocyclic ligand transition metal complexes. These complexes, specifically designed with isoquinoline derivatives combined with phosphine ligands, represent a significant leap forward in asymmetric catalytic hydrogenation and hydrogen transfer reactions. Unlike traditional catalysts that often struggle with substrate specificity, this innovation offers a versatile platform capable of transforming prochiral ketones and esters into chiral alcohols with remarkable precision. The technology addresses the persistent industrial pain point of processing sterically hindered substrates, which have historically resulted in low yields and poor stereoselectivity. By leveraging the unique electronic and steric properties of the isoquinoline backbone, this patent provides a robust solution for producing high-value intermediates essential for active pharmaceutical ingredients (APIs) and advanced agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of hindered ketones and esters has been plagued by significant technical barriers that limit commercial viability. Conventional catalytic systems often lack the necessary spatial configuration to effectively differentiate between enantiomers when bulky alkyl groups are present near the reaction center. This steric hindrance typically leads to sluggish reaction kinetics, requiring harsh conditions such as elevated temperatures and high pressures that can degrade sensitive functional groups. Furthermore, traditional catalysts frequently suffer from low turnover numbers and poor enantioselectivity, necessitating costly and time-consuming downstream purification steps to achieve the required optical purity. The reliance on expensive noble metals without efficient ligand support further exacerbates production costs, making the manufacturing of complex chiral intermediates economically challenging for many supply chains. These limitations create bottlenecks in the development of new drugs and specialty chemicals where specific stereochemistry is mandatory for biological activity.
The Novel Approach
The innovative approach detailed in the patent overcomes these obstacles by engineering a specialized ligand environment that optimizes both reactivity and selectivity. By integrating a nitrogen-containing heterocyclic ligand, specifically an isoquinoline derivative, with monodentate or bidentate phosphine ligands, the resulting transition metal complex achieves a superior geometric arrangement around the metal center. This configuration allows the catalyst to accommodate bulky substrates that would otherwise be rejected by standard systems, facilitating smooth conversion even in the presence of significant steric hindrance. The method operates under relatively mild conditions, ranging from 0°C to 120°C, which preserves the integrity of sensitive molecules while maintaining high catalytic activity. Additionally, the flexibility to use various transition metals such as Ruthenium, Rhodium, or Iridium allows chemists to fine-tune the system for specific substrate classes, ensuring broad applicability across different synthetic routes. This adaptability translates directly into more reliable manufacturing processes and reduced operational risks for chemical producers.
Mechanistic Insights into Isoquinoline-Phosphine Catalytic Systems
At the heart of this technology lies a sophisticated coordination chemistry mechanism where the interplay between the nitrogen heterocycle and the phosphine moiety dictates the stereochemical outcome. The transition metal, typically Ruthenium or Rhodium, is coordinated by the nitrogen atom of the isoquinoline ring and the phosphorus atom of the phosphine ligand, creating a rigid chiral pocket. This structural rigidity is crucial for inducing asymmetry during the hydrogenation process, as it forces the substrate to approach the metal center from a specific trajectory. The presence of substituents on the isoquinoline ring, such as alkyl or aryl groups, further modulates the electronic density and steric bulk around the active site, allowing for precise control over the reaction pathway. This level of molecular engineering ensures that the hydride transfer occurs with high fidelity, resulting in products with exceptional enantiomeric excess values. Understanding this mechanism is vital for R&D teams aiming to replicate or adapt the chemistry for novel target molecules.
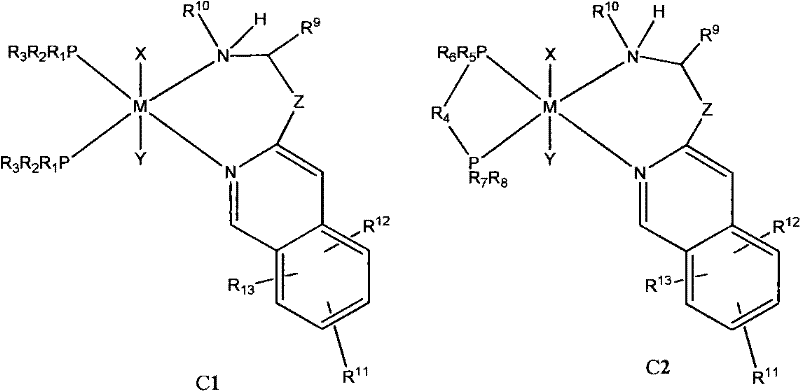
Beyond the primary coordination sphere, the patent highlights the importance of ancillary ligands and counterions in stabilizing the active catalytic species. The complex can exist in various configurations, including cis or trans isomers, depending on the specific ligands employed and the reaction conditions. The ability to form stable hydride species, often generated in situ using reducing agents like sodium borohydride, is a key feature that enhances the catalyst's longevity and turnover frequency. Moreover, the addition of additives such as triphenylphosphine can further boost stereoselectivity and reaction rates by modifying the coordination equilibrium. This dynamic behavior allows the system to self-optimize during the reaction, maintaining high performance even as substrate concentrations fluctuate. For process chemists, this means a more forgiving and robust system that can tolerate minor variations in feedstock quality without compromising the final product's optical purity.
How to Synthesize Isoquinoline Ligand Complexes Efficiently
The synthesis of these high-performance catalysts involves a streamlined multi-step process that begins with the preparation of the specialized isoquinoline amine ligand. This precursor is typically generated through the reduction of isoquinoline nitriles or the reaction of isoquinoline carbonyl derivatives with Grignard reagents, followed by amination. Once the ligand is secured, it is reacted with a transition metal precursor, such as a ruthenium halide complex, in an organic solvent like DMF or toluene. The reaction is conducted under an inert atmosphere to prevent oxidation of the sensitive metal center, with temperatures carefully controlled to ensure complete complexation without decomposition. The resulting solid complex can be isolated and purified or used directly in situ for immediate catalytic applications, offering flexibility depending on the scale of operation.
- Prepare the nitrogen-containing heterocyclic ligand by reacting isoquinoline derivatives with appropriate amines or Grignard reagents under controlled temperatures.
- Synthesize the transition metal precursor using ruthenium, rhodium, or iridium halides in organic solvents such as DMF or toluene.
- Combine the ligand and metal precursor under inert atmosphere at 0-120°C for 0.5 to 20 hours to form the active catalytic complex.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this catalytic technology offers profound strategic benefits that extend beyond mere technical performance. The ability to process hindered ketones efficiently eliminates the need for multiple synthetic steps or protective group strategies that traditionally inflate material costs and extend lead times. By achieving high selectivity in a single step, manufacturers can significantly reduce the consumption of raw materials and solvents, leading to a leaner and more cost-effective production workflow. This efficiency is particularly valuable in the context of volatile raw material markets, where minimizing waste and maximizing yield per batch directly impacts the bottom line. Furthermore, the robustness of the catalyst system reduces the risk of batch failures, ensuring a more predictable and reliable supply of critical intermediates for downstream customers.
- Cost Reduction in Manufacturing: The implementation of this ligand system drives cost reduction by streamlining the purification process. Since the catalyst delivers high enantiomeric excess directly, the reliance on expensive chiral resolution techniques such as preparative HPLC or repeated recrystallization is drastically diminished. This reduction in downstream processing not only saves on solvent and energy costs but also shortens the overall cycle time for production. Additionally, the potential to use lower catalyst loadings due to high turnover numbers further decreases the expenditure on precious metals. These cumulative savings allow for a more competitive pricing structure for the final API intermediates, enhancing market positioning.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the versatility and stability of the catalyst precursors. The ligands and metal salts required for this synthesis are commercially available and do not rely on obscure or single-source suppliers, mitigating the risk of supply disruptions. The mild reaction conditions also mean that the process can be executed in a wider range of manufacturing facilities without requiring specialized high-pressure or cryogenic equipment. This flexibility allows for diversified manufacturing locations, reducing geopolitical and logistical risks associated with centralized production. Consequently, partners can expect consistent delivery schedules and greater resilience against external market shocks.
- Scalability and Environmental Compliance: Scaling this technology from laboratory to commercial production is facilitated by the use of standard organic solvents and manageable temperature ranges. The process aligns well with green chemistry principles by improving atom economy and reducing the generation of hazardous waste streams associated with inefficient reactions. The ability to operate effectively in protic or non-protic solvents provides additional options for optimizing environmental impact and solvent recovery rates. For supply chain heads, this translates to easier regulatory compliance and a smoother path to validation for large-scale manufacturing campaigns, ensuring long-term sustainability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isoquinoline-based catalytic technology. These insights are derived directly from the patent specifications and are intended to clarify the operational capabilities and strategic value of the system for potential partners. Understanding these details is essential for making informed decisions about integrating this chemistry into existing production pipelines.
Q: What represents the primary advantage of this isoquinoline ligand system over conventional catalysts?
A: The primary advantage lies in its exceptional ability to handle sterically hindered ketones and esters, which traditionally show poor conversion rates. The unique spatial arrangement of the nitrogen heterocyclic and phosphine ligands creates a highly specific chiral environment, resulting in significantly improved enantioselectivity and yield for difficult substrates.
Q: Can this catalytic system be adapted for large-scale industrial production?
A: Yes, the patent describes preparation methods using common organic solvents and moderate temperature ranges (0-120°C), which are compatible with standard industrial reactor setups. The robustness of the transition metal complex allows for scalable synthesis without requiring exotic or prohibitively expensive conditions, facilitating commercial scale-up.
Q: How does this technology impact impurity profiles in final API intermediates?
A: By achieving high enantiomeric excess (ee) directly during the hydrogenation step, this technology drastically reduces the burden on downstream purification processes. This leads to a cleaner impurity profile, minimizing the need for extensive recrystallization or chromatographic separation, thereby enhancing overall process efficiency and product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinoline Ligand Complex Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the isoquinoline ligand transition metal complex technology described in CN102417523A to meet your specific production requirements. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of catalyst or intermediate meets the highest international standards. Our commitment to quality assurance means that you can rely on us for consistent performance, whether you are in the early stages of process development or managing full-scale commercial manufacturing.
We invite you to collaborate with us to unlock the full potential of this asymmetric hydrogenation technology for your product portfolio. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule and volume needs. We encourage you to reach out for specific COA data and route feasibility assessments to verify how this innovative catalytic system can enhance your supply chain efficiency. Let us help you navigate the complexities of chiral synthesis with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
