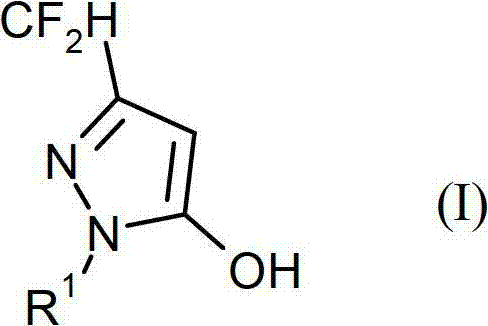
Advanced Synthesis of 1-Alkyl-3-Difluoromethyl-5-Hydroxypyrazoles for Commercial Fungicide Production
Advanced Synthesis of 1-Alkyl-3-Difluoromethyl-5-Hydroxypyrazoles for Commercial Fungicide Production
The global agrochemical industry is constantly seeking more efficient pathways to produce high-value fungicide intermediates, and the technology disclosed in patent CN102906073A represents a significant breakthrough in this domain. This patent details a novel process for preparing 1-alkyl-3-difluoromethyl-5-hydroxypyrazoles, which serve as critical building blocks for next-generation crop protection agents. Unlike traditional methods that struggle with poor regioselectivity and low conversion rates, this innovative approach utilizes an organic acid-catalyzed cyclization to achieve exceptional purity and yield. For R&D Directors and Procurement Managers, understanding the nuances of this synthesis is vital for optimizing supply chains and reducing the cost of goods sold (COGS) in agrochemical intermediate manufacturing. The ability to produce these complex heterocyclic structures with high precision directly impacts the viability of downstream fungicide formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazole derivatives has been plagued by significant technical hurdles that hinder commercial scalability and economic efficiency. Prior art, such as the method described by Takahashi in US2005/256004 A1, relies on reacting difluoroacetylacetic ester with monomethyl hydrazine in the presence of concentrated hydrochloric acid in ethanol. This conventional route suffers from a critically low yield of only 33.3%, which is commercially unsustainable for large-volume production. Furthermore, alternative methods described by Hamper utilize perfluoroalkyl ethynyl esters as starting materials; however, these raw materials are prohibitively expensive and often unavailable in the open market, creating severe supply chain bottlenecks. The formation of regioisomers is another persistent issue, where unwanted 2-alkyl isomers are generated alongside the desired product, necessitating complex and costly purification steps that erode profit margins.
The Novel Approach
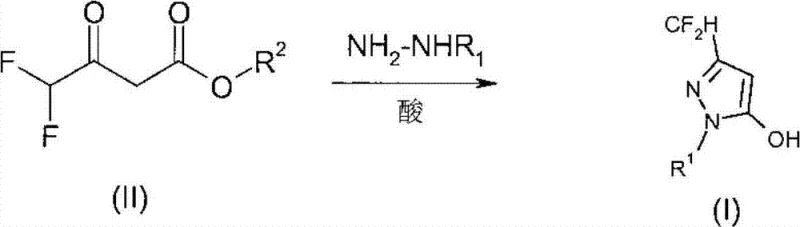
The methodology outlined in CN102906073A offers a transformative solution by shifting the reaction paradigm to an organic acid-mediated process in non-protic or moderately polar solvents. By reacting alkyl difluoroacetoacetates with alkylhydrazines in the presence of organic acids like formic acid or acetic acid, the process achieves a regioselectivity of nearly 100%, effectively eliminating the formation of undesirable isomers. This novel approach not only drastically simplifies the work-up procedure but also enables the reaction to proceed under mild conditions, typically between 20°C and 40°C, which reduces energy consumption and safety risks associated with high-temperature operations. For supply chain heads, this means a more reliable agrochemical intermediate supplier capability, as the process is robust enough to handle commercial scale-up of complex polymer additives and fine chemicals without the volatility associated with older, harsher chemistries.
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this technological advancement lies in the precise control of the cyclization mechanism through the use of specific organic acid catalysts. In the reaction between alkyl difluoroacetoacetates (Formula II) and alkylhydrazines (Formula III), the organic acid plays a dual role: it activates the carbonyl group of the beta-keto ester towards nucleophilic attack and simultaneously protonates the hydrazine to modulate its reactivity. This delicate balance prevents the over-reaction or polymerization that often occurs with strong mineral acids. The solvent choice, particularly methyl tert-butyl ether (MTBE) or tetrahydrofuran (THF), further stabilizes the transition state, ensuring that the nucleophilic attack occurs exclusively at the correct position to form the 1-alkyl substituted pyrazole ring. This mechanistic precision is what allows the process to maintain high regioselectivity, ensuring that the amount of undesirable regional isomer formed is lower than 1%, and often less than 0.1%.
From an impurity control perspective, this mechanism is superior because it avoids the generation of side products that are structurally similar to the target molecule, which are notoriously difficult to separate via crystallization or distillation. The absence of heavy metal catalysts or exotic reagents means that the impurity profile is clean, consisting primarily of unreacted starting materials that are easily removed during the aqueous work-up phase. For R&D teams focused on high-purity agrochemical intermediate specifications, this translates to a product that meets stringent regulatory requirements for residue limits without the need for additional, yield-losing purification steps. The reaction kinetics are also favorable, with conversion typically completing within 8 to 20 hours, allowing for high throughput in batch reactors.
How to Synthesize 1-Alkyl-3-Difluoromethyl-5-Hydroxypyrazoles Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize the benefits of the acid-catalyzed mechanism. The process begins with the dissolution of the difluoroacetoacetate in a suitable solvent, followed by the controlled addition of the organic acid and the alkylhydrazine. Maintaining the temperature within the 0-60°C range is critical; while the reaction can proceed at room temperature, slight cooling during the exothermic addition of hydrazine helps maintain selectivity. The detailed standardized synthesis steps, including specific molar ratios and work-up procedures, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by dissolving alkyl difluoroacetoacetates in a suitable solvent such as methyl tert-butyl ether.
- Add an organic acid catalyst, preferably formic acid or acetic acid, and cool the mixture to a temperature range of 0-60°C.
- Introduce alkylhydrazines slowly to the mixture, maintain stirring for 8-20 hours, and isolate the product via phase separation and solvent removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers substantial strategic advantages that go beyond simple chemical yield. The elimination of expensive and hard-to-source raw materials, such as perfluoroalkyl ethynyl esters, directly translates to a more stable and predictable cost structure. By utilizing commercially available alkyl difluoroacetoacetates and alkylhydrazines, manufacturers can secure raw material supply from multiple vendors, reducing the risk of single-source dependency. This flexibility is crucial for maintaining supply chain reliability in the volatile agrochemical market, where demand fluctuations can be sharp. Furthermore, the mild reaction conditions reduce the burden on facility infrastructure, allowing for production in standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment.
- Cost Reduction in Manufacturing: The process achieves a dramatic reduction in manufacturing costs by significantly improving atom economy and reducing waste generation. With yields consistently reaching 90-95%, the amount of raw material required per kilogram of final product is minimized, leading to substantial savings on input costs. Additionally, the high regioselectivity eliminates the need for costly chromatographic purification or multiple recrystallizations to remove isomers, which are often the most expensive steps in fine chemical production. The use of recoverable solvents like MTBE further enhances the economic profile by allowing for solvent recycling, thereby lowering the overall environmental and financial footprint of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for this synthesis ensures that lead times are significantly reduced compared to processes requiring custom-synthesized precursors. Since alkyl hydrazines and difluoroacetoacetates are standard industrial chemicals, they can be sourced with short notice, enabling manufacturers to respond quickly to market demand spikes. This availability also mitigates the risk of production stoppages due to raw material shortages, a common pain point in the commercial scale-up of complex agrochemical intermediates. The robustness of the reaction also means that batch-to-batch variability is low, ensuring consistent quality for downstream customers.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this route is inherently greener, as it avoids the use of toxic heavy metals and generates less hazardous waste. The organic acid catalysts are less corrosive than mineral acids, reducing equipment maintenance costs and safety risks for operators. The process is easily scalable from laboratory benchtop to multi-ton production, as demonstrated by the patent examples which show consistent performance at the 1-mole scale and above. This scalability ensures that the technology can meet the growing global demand for fungicides without compromising on environmental compliance or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of the acid-catalyzed method over conventional synthesis?
A: The primary advantage is the exceptional regioselectivity, achieving over 99% preference for the 1-alkyl isomer while suppressing the undesirable 2-alkyl isomer, which significantly simplifies downstream purification.
Q: Can this process be scaled for industrial fungicide manufacturing?
A: Yes, the process operates under mild conditions (20-40°C) using commercially available solvents and reagents, making it highly suitable for large-scale commercial production without specialized high-pressure equipment.
Q: What are the typical yield and purity specifications for this intermediate?
A: According to patent data, the process consistently delivers yields between 90% and 95% with product purity reaching up to 96%, ensuring high efficiency for agrochemical supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Alkyl-3-Difluoromethyl-5-Hydroxypyrazoles Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the development of next-generation agrochemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1-alkyl-3-difluoromethyl-5-hydroxypyrazoles meets the highest industry standards. We are committed to supporting your R&D and supply chain goals with reliable, high-quality chemical solutions.
We invite you to collaborate with us to optimize your production costs and secure your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your competitive advantage in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
