Advanced Purification Technology for High-Purity Ornithine Aspartate Commercial Production
Advanced Purification Technology for High-Purity Ornithine Aspartate Commercial Production
The pharmaceutical landscape for hepatopathy therapeutics demands increasingly stringent purity standards to ensure patient safety and therapeutic efficacy. Patent CN102093236A introduces a groundbreaking purification methodology for Ornithine Aspartate, a critical compound used in the treatment of liver diseases and hyperammonemia. This technology addresses the longstanding challenges of impurity removal and yield optimization that have plagued conventional manufacturing routes. By integrating activated carbon adsorption with a novel neutral alumina chromatographic system, the process achieves exceptional purity levels while maintaining robust operational feasibility. For global procurement teams and R&D directors, this represents a significant opportunity to secure a reliable active pharmaceutical ingredient supplier capable of delivering consistent quality. The technical breakthroughs detailed in this patent not only enhance the chemical profile of the compound but also streamline the production workflow, offering substantial advantages in cost reduction in pharmaceutical manufacturing. As the demand for high-quality liver health interventions grows, adopting this advanced purification protocol becomes a strategic imperative for maintaining competitive supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis and purification of Ornithine Aspartate have been hindered by complex, multi-step processes that introduce significant operational risks and cost inefficiencies. Traditional methods often rely on enzymatic conversion using arginase, which is prohibitively expensive and creates supply bottlenecks due to the high cost of the enzyme itself. Alternative chemical routes frequently employ ion-exchange resins or toxic heavy metal salts, such as barium hydroxide, to facilitate salt formation and purification. These approaches suffer from critical drawbacks, including the generation of hazardous three-waste streams that require expensive disposal protocols and regulatory compliance measures. Furthermore, the use of strong acid ion-exchange resins can lead to the introduction of unwanted sodium ions and incomplete removal of impurities, resulting in final product purity that often fails to meet modern pharmacopoeia standards. The complexity of these legacy processes also limits scalability, making it difficult to transition from laboratory synthesis to commercial scale-up of complex amino acid derivatives without significant yield losses.
The Novel Approach
The innovative method disclosed in the patent data circumvents these historical limitations by employing a streamlined, non-toxic purification sequence that prioritizes both efficiency and safety. Instead of relying on expensive enzymes or hazardous heavy metals, this approach utilizes a combination of activated carbon decolorization and precision chromatography using neutral alumina. This shift eliminates the need for toxic barium ions and complex resin regeneration cycles, thereby drastically simplifying the operational workflow. The process is designed to be robust, utilizing common organic solvents like acetone and acetonitrile in specific ratios to precipitate and crystallize the product effectively. By optimizing parameters such as temperature, solvent volume ratios, and flow rates, the method ensures high recovery rates without compromising on purity. This novel approach not only enhances the quality of the preparation product but also aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing process and ensuring long-term supply continuity for global markets.
Mechanistic Insights into Alumina-Based Chromatographic Purification
The core of this technological advancement lies in the specific interaction between the Ornithine Aspartate compound and the neutral alumina stationary phase during chromatographic separation. Unlike silica gel, which can exhibit acidic properties that might degrade sensitive amino acid salts, neutral alumina provides a chemically inert environment that preserves the structural integrity of the molecule. The mechanism involves the selective adsorption of polar impurities and colored by-products onto the alumina surface, allowing the target compound to elute with high fidelity. The optimization of the mobile phase, specifically a 1:2 volume ratio of acetonitrile to water, is critical for controlling the elution profile and ensuring that impurities are washed out prematurely while the product is retained and then released in high-concentration fractions. This precise control over the separation dynamics is what enables the achievement of purity levels greater than 99.5%, a benchmark that is difficult to reach with standard crystallization techniques alone.
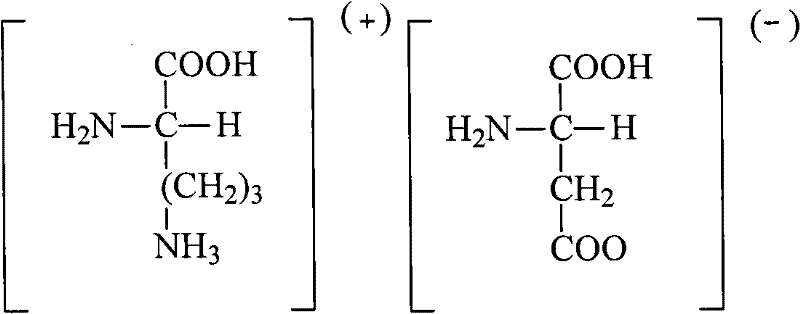
Furthermore, the purification mechanism includes a preliminary activated carbon treatment step that plays a vital role in impurity control. By heating the solution to 60-70°C in the presence of activated carbon, high molecular weight impurities and colored substances are effectively adsorbed before the chromatographic step. This pre-purification reduces the load on the chromatographic column, extending its lifespan and maintaining consistent separation efficiency over multiple batches. The subsequent precipitation step using organic solvents further refines the crude material by removing soluble impurities that do not co-precipitate with the target salt. This multi-stage filtration of impurities ensures that the final crystallization yields a product with a significantly reduced toxic side effect profile. For R&D directors, understanding this mechanistic depth is crucial for validating the process's robustness and ensuring that the impurity profile remains within strict regulatory limits for pharmaceutical intermediates.
How to Synthesize Ornithine Aspartate Efficiently
Implementing this purification protocol requires precise adherence to the optimized parameters regarding solvent ratios, temperature controls, and flow rates to ensure reproducibility at scale. The process begins with the dissolution of the crude compound followed by the critical decolorization step, which sets the foundation for high-purity output. Detailed standard operating procedures regarding the specific mesh size of the alumina and the exact flow velocity are essential for maintaining the separation resolution described in the patent. The following guide outlines the standardized synthesis steps derived from the experimental data, providing a clear roadmap for technical teams to replicate the high-yield results in a production environment.
- Dissolve crude Ornithine Aspartate in purified water and treat with 0.1-0.3% activated carbon at 60-70°C to remove colored impurities.
- Precipitate the compound by adding a mixed organic solvent (acetone and acetonitrile) to the filtrate under stirring conditions.
- Perform chromatographic separation using neutral alumina as the stationary phase and a 1: 2 acetonitrile-water mixture as the mobile phase.
- Crystallize the purified eluent by adding organic solvent and cooling to 2-8°C, followed by filtration and vacuum drying at 50°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this purification technology offers transformative benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for hepatopathy therapeutics. The elimination of expensive enzymatic catalysts and toxic heavy metal reagents translates directly into a more cost-effective manufacturing process, allowing for significant cost savings without sacrificing quality. The simplified workflow reduces the number of unit operations required, which in turn minimizes the potential for operational errors and batch failures. This reliability is paramount for maintaining a consistent supply of high-purity Ornithine Aspartate, ensuring that downstream formulation partners receive materials that meet stringent specifications every time. Additionally, the reduced generation of hazardous waste lowers the environmental compliance burden, making the supply chain more sustainable and resilient against regulatory changes.
- Cost Reduction in Manufacturing: The removal of costly arginase enzymes and toxic barium salts from the process flow significantly lowers the raw material expenditure and waste disposal costs associated with production. By utilizing readily available solvents and neutral alumina, the overall cost of goods sold is optimized, providing a competitive pricing structure for bulk purchasers. This economic efficiency is achieved without compromising the high purity standards required for pharmaceutical applications, ensuring value retention throughout the supply chain.
- Enhanced Supply Chain Reliability: The reliance on stable, non-biological reagents eliminates the supply bottlenecks often associated with enzymatic processes, ensuring a more predictable and continuous production schedule. The robustness of the chromatographic method allows for consistent batch-to-batch quality, reducing the risk of supply disruptions due to out-of-specification results. This stability is crucial for long-term procurement contracts and ensures that manufacturing partners can rely on a steady flow of materials for their own production lines.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing standard equipment that can be easily expanded from pilot to commercial volumes without complex re-engineering. The reduction in toxic waste generation aligns with increasingly strict environmental regulations, reducing the risk of compliance-related shutdowns. This scalability ensures that the supply can grow in tandem with market demand, supporting the commercial expansion of liver health treatments globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of Ornithine Aspartate, based on the specific advantages and mechanisms detailed in the patent literature. These insights are intended to clarify the operational benefits and quality assurances provided by this advanced manufacturing route. Understanding these details helps stakeholders make informed decisions regarding the integration of this high-purity compound into their product portfolios.
Q: Why is neutral alumina preferred over silica gel for Ornithine Aspartate purification?
A: Neutral alumina offers superior adsorption selectivity for specific impurities without the acidity issues associated with silica gel, resulting in higher product stability and purity levels exceeding 99.5%.
Q: How does this purification method impact the safety profile of the final pharmaceutical product?
A:
Q: Is this purification process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes scalable unit operations such as standard chromatography and crystallization, avoiding expensive enzymatic steps or hazardous reagents, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ornithine Aspartate Supplier
The technical potential of this purification route underscores the importance of partnering with a CDMO expert who understands the nuances of scaling complex amino acid derivatives. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to market is seamless and efficient. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of Ornithine Aspartate meets the highest international standards for safety and efficacy. We are dedicated to supporting your R&D and commercial goals with a supply chain that is both robust and responsive to your specific needs.
We invite you to initiate a conversation about optimizing your supply chain for liver health therapeutics with our technical procurement team. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how this purification technology can benefit your specific production requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
