Scalable Production of High-Purity 4-Cyano-2-Methoxybenzaldehyde via Continuous Photo-Induced Radical Bromination
Scalable Production of High-Purity 4-Cyano-2-Methoxybenzaldehyde via Continuous Photo-Induced Radical Bromination
The global pharmaceutical landscape is witnessing a surge in demand for advanced treatments targeting chronic kidney disease (CKD) and type 2 diabetes, driven largely by the success of non-steroidal mineralocorticoid receptor antagonists like Finerenone, commercially known as Kerendia®. As the market for this critical therapeutic expands, the pressure on the supply chain to deliver high-quality key intermediates has never been more intense.  Patent CN115991661A introduces a groundbreaking preparation method for 4-cyano-2-methoxybenzaldehyde, a pivotal building block in the synthesis of Finerenone. This innovation addresses long-standing industry pain points regarding toxicity, cost, and scalability by replacing hazardous cryogenic and heavy metal-dependent processes with a streamlined, photo-chemical approach. For R&D directors and procurement strategists, understanding this technological shift is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent purity specifications while optimizing manufacturing expenditures.
Patent CN115991661A introduces a groundbreaking preparation method for 4-cyano-2-methoxybenzaldehyde, a pivotal building block in the synthesis of Finerenone. This innovation addresses long-standing industry pain points regarding toxicity, cost, and scalability by replacing hazardous cryogenic and heavy metal-dependent processes with a streamlined, photo-chemical approach. For R&D directors and procurement strategists, understanding this technological shift is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent purity specifications while optimizing manufacturing expenditures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-cyano-2-methoxybenzaldehyde has been plagued by significant operational hazards and economic inefficiencies inherent to traditional organometallic and cyanation chemistries. One prevalent prior art route involves the use of n-butyllithium at cryogenic temperatures of -78°C to introduce aldehyde groups, followed by palladium-catalyzed cyanation using toxic zinc cyanide. 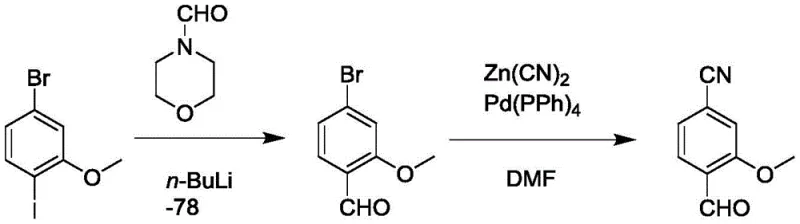 This methodology presents severe challenges for industrial scale-up, including the high energy costs associated with maintaining ultra-low temperatures and the substantial safety risks posed by pyrophoric reagents and cyanide sources. Furthermore, alternative routes utilizing potassium hexacyanoferrate and palladium acetate suffer from similar drawbacks, requiring expensive transition metal catalysts that necessitate complex downstream purification to meet residual metal limits. These conventional pathways not only inflate the cost reduction in pharmaceutical intermediates manufacturing but also generate significant hazardous waste, complicating environmental compliance and supply chain continuity.
This methodology presents severe challenges for industrial scale-up, including the high energy costs associated with maintaining ultra-low temperatures and the substantial safety risks posed by pyrophoric reagents and cyanide sources. Furthermore, alternative routes utilizing potassium hexacyanoferrate and palladium acetate suffer from similar drawbacks, requiring expensive transition metal catalysts that necessitate complex downstream purification to meet residual metal limits. These conventional pathways not only inflate the cost reduction in pharmaceutical intermediates manufacturing but also generate significant hazardous waste, complicating environmental compliance and supply chain continuity.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN115991661A leverages a continuous photo-free radical initiation strategy to achieve superior selectivity and safety profiles. This innovative route begins with the conversion of 4-methyl-3-methoxybenzoic acid into an amide, followed by dehydration to a nitrile, setting the stage for a highly controlled bromination step. 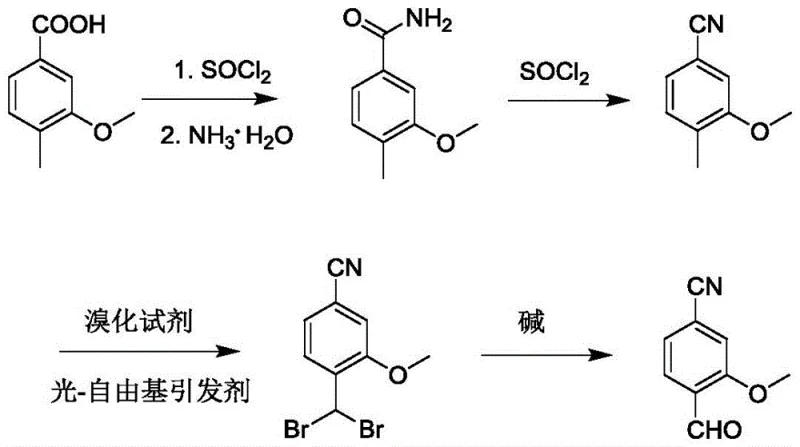 By employing an external LED light source in conjunction with radical initiators such as azobisisobutyronitrile (AIBN) or dibromohydantoin, the process effectively promotes the double bromination of the methyl group while suppressing the formation of monobrominated byproducts to levels below 0.1%. This mechanistic precision eliminates the need for precious metal catalysts and cryogenic conditions, resulting in a process that is not only economically superior but also environmentally sustainable. The final hydrolysis step proceeds under mild alkaline conditions, delivering the target aldehyde with yields exceeding 93% and purity reaching 99.8%, thereby establishing a new benchmark for the commercial scale-up of complex pharmaceutical intermediates.
By employing an external LED light source in conjunction with radical initiators such as azobisisobutyronitrile (AIBN) or dibromohydantoin, the process effectively promotes the double bromination of the methyl group while suppressing the formation of monobrominated byproducts to levels below 0.1%. This mechanistic precision eliminates the need for precious metal catalysts and cryogenic conditions, resulting in a process that is not only economically superior but also environmentally sustainable. The final hydrolysis step proceeds under mild alkaline conditions, delivering the target aldehyde with yields exceeding 93% and purity reaching 99.8%, thereby establishing a new benchmark for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Photo-Induced Radical Bromination
The core technological breakthrough of this synthesis lies in the third step, where a continuous photo-free radical mechanism drives the transformation of the methyl group into a dibromomethyl moiety with exceptional fidelity. Upon irradiation with an LED light source, the radical initiator undergoes homolytic cleavage to generate free radicals that abstract hydrogen atoms from the benzylic methyl position. This generates a stable benzylic radical which subsequently reacts with the brominating agent, such as N-bromosuccinimide (NBS) or dibromohydantoin, to form the carbon-bromine bond. The continuous nature of the photo-excitation ensures a steady state of radical concentration, which is critical for driving the reaction to the dibromo stage without stalling at the monobromo intermediate. This kinetic control is what allows the process to maintain monobrominated impurity levels at less than 0.1%, a feat difficult to achieve with thermal radical initiators alone. The absence of transition metals in this step further simplifies the reaction matrix, preventing the formation of metal-complexed side products that often plague palladium-catalyzed cross-couplings.
From an impurity control perspective, the mechanistic elegance of this route provides a robust defense against the formation of hard-to-remove contaminants that typically compromise API quality. In traditional cyanation routes, side reactions involving the cyano group or incomplete conversion of halides can lead to structurally similar impurities that require costly chromatographic purification. However, the photo-bromination pathway followed by straightforward alkaline hydrolysis creates a clean reaction profile where the primary byproduct is simply the unreacted starting material or the easily separable monobromo species. The hydrolysis of the gem-dibromide to the aldehyde is a well-understood nucleophilic substitution that proceeds cleanly in aqueous media, avoiding the use of aggressive oxidants like sodium periodate found in other prior art. This results in a final product with a single impurity profile of less than 0.1%, ensuring that the material meets the rigorous quality standards required for GMP manufacturing of active pharmaceutical ingredients without the need for extensive recrystallization cycles.
How to Synthesize 4-Cyano-2-Methoxybenzaldehyde Efficiently
The execution of this synthesis protocol requires careful attention to the photo-reactor setup and the stoichiometry of the brominating agents to maximize the efficiency of the radical chain reaction. The process is designed to be operationally simple, utilizing common solvents like chloroform or benzotrifluoride and standard heating equipment, making it accessible for facilities looking to reduce lead time for high-purity pharmaceutical intermediates. Operators must ensure that the light source intensity and wavelength are optimized to match the absorption characteristics of the radical initiator, typically using 35w LED sources as demonstrated in the patent examples. The following guide outlines the standardized operational sequence derived from the patent data, providing a clear roadmap for technical teams to implement this superior manufacturing strategy.
- Convert 4-methyl-3-methoxybenzoic acid to its corresponding amide using thionyl chloride and ammonia water.
- Dehydrate the amide to 4-methyl-3-methoxybenzonitrile using thionyl chloride under reflux conditions.
- Perform continuous photo-induced radical bromination on the methyl group using an LED light source and radical initiator to form the dibromo intermediate.
- Hydrolyze the dibromo intermediate using an aqueous base to yield the final 4-cyano-2-methoxybenzaldehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photo-chemical synthesis route represents a strategic opportunity to de-risk the supply of critical Finerenone intermediates while achieving substantial cost savings. The elimination of palladium catalysts and toxic cyanide salts removes two of the most volatile cost drivers in fine chemical manufacturing, as the prices of precious metals and hazardous waste disposal can fluctuate wildly. By shifting to a process based on abundant organic acids and benign brominating reagents, manufacturers can stabilize their raw material costs and insulate themselves from supply shocks associated with specialty metal catalysts. Furthermore, the mild reaction conditions eliminate the need for specialized cryogenic infrastructure, allowing production to occur in standard glass-lined or stainless steel reactors, which significantly lowers capital expenditure requirements for scale-up.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of expensive catalytic systems and the simplification of downstream processing. Traditional routes relying on palladium require extensive scavenging steps to reduce metal residues to ppm levels, adding both time and material costs to the production cycle. In contrast, this metal-free approach yields a crude product of such high purity that purification becomes a trivial matter of filtration and concentration, drastically reducing solvent consumption and processing time. Additionally, the high atom economy of the bromination step and the near-quantitative yields observed in the dehydration and hydrolysis steps ensure that raw material utilization is maximized, directly contributing to a lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity chemicals rather than specialized, hazard-classified reagents. Reagents like thionyl chloride and sodium bicarbonate are globally available in bulk quantities, reducing the risk of bottlenecks that often occur with niche catalysts or cryogenic gases. The safety profile of the process also plays a crucial role in reliability; by avoiding pyrophoric n-butyllithium and highly toxic zinc cyanide, facilities can operate with fewer regulatory hurdles and lower insurance premiums. This operational safety translates to fewer unplanned shutdowns and a more consistent ability to meet delivery schedules, ensuring that downstream API manufacturers receive their materials on time without interruption.
- Scalability and Environmental Compliance: The scalability of this technology is evidenced by its reliance on continuous photo-irradiation, a technique that scales linearly with reactor surface area or through the use of flow chemistry modules. Unlike batch processes limited by heat transfer issues at low temperatures, this exothermic radical reaction can be managed effectively even at multi-ton scales. From an environmental standpoint, the process aligns with green chemistry principles by reducing the emission of three wastes, particularly heavy metal effluents and cyanide-containing wastewater. This compliance advantage facilitates smoother permitting processes and enhances the sustainability credentials of the supply chain, a factor increasingly weighted by multinational pharmaceutical buyers in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational feasibility and quality advantages of the photo-bromination method for stakeholders evaluating this technology for potential adoption or outsourcing.
Q: How does the new photo-bromination method improve impurity control compared to traditional routes?
A: The patented method utilizes a continuous photo-free radical initiator system which achieves high selectivity for the dibromo product, controlling the monobrominated byproduct content to less than 0.1%, significantly higher purity than conventional thermal bromination.
Q: What are the safety advantages of this synthesis route over prior art involving zinc cyanide?
A: Unlike prior art routes that require toxic zinc cyanide or potassium hexacyanoferrate and expensive palladium catalysts, this method avoids highly toxic cyanide sources entirely, utilizing safer brominating reagents and mild hydrolysis conditions.
Q: Can this process be scaled for industrial production of Finerenone intermediates?
A: Yes, the process operates under mild conditions (55-95°C) without the need for cryogenic temperatures (-78°C) required by lithiation routes, making it highly suitable for large-scale commercial manufacturing with reduced energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Cyano-2-Methoxybenzaldehyde Supplier
As the demand for non-steroidal mineralocorticoid receptor antagonists continues to grow, securing a partner with the technical capability to execute complex, high-purity syntheses is paramount for maintaining competitive advantage. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped to handle photo-chemical reactions and stringent purity specifications, supported by rigorous QC labs that guarantee every batch of 4-cyano-2-methoxybenzaldehyde meets the highest international standards for pharmaceutical intermediates.
We invite you to engage with our technical team to explore how this innovative patent-based route can be integrated into your supply chain to drive efficiency and quality. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this metal-free process for your specific volume requirements. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most robust and cost-effective manufacturing strategy available.
