Advanced Synthesis of Blue Fluorescent Pyridine Diimine Cadmium Complexes for High-Performance OLED Displays
Introduction to High-Performance Blue OLED Emitters
The rapid evolution of organic light-emitting diode (OLED) technology has created an urgent demand for stable and efficient blue light-emitting materials, a segment that has historically lagged behind red and green counterparts in terms of performance and longevity. Patent CN101402646B introduces a groundbreaking class of blue fluorescent organic luminescent materials based on pyridine diimine cadmium complexes, specifically designed to overcome the low luminous intensity plaguing existing solutions. This proprietary technology leverages the tunability of metal ions and nitrogen-containing ligands to create robust molecular architectures that emit intense blue fluorescence with excellent monochromatic properties. The disclosed compounds, with molecular formulas such as C23H23N3CdCl2, C29H35N3CdCl2, and C27H31N3CdCl2, represent a significant leap forward in electroluminescent material science. By utilizing a straightforward two-step synthesis involving ligand preparation followed by metal coordination under inert atmosphere, the patent achieves isolated yields exceeding 84%, ensuring that these advanced materials are not only scientifically superior but also commercially viable for large-scale display manufacturing applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing blue organic light-emitting materials have frequently encountered substantial hurdles regarding stability and emission efficiency. Many existing blue emitters suffer from rapid degradation under operational conditions or exhibit insufficient luminous intensity, necessitating higher driving voltages that compromise device lifespan and energy efficiency. Furthermore, the synthesis of complex conjugated systems often involves multi-step pathways with harsh reaction conditions, leading to poor atom economy and difficult purification processes that drive up production costs. The lack of effective steric hindrance in many conventional ligands can also lead to undesirable intermolecular interactions, such as excimer formation, which quenches fluorescence and shifts emission colors away from the desired pure blue spectrum. These technical bottlenecks have slowed the widespread adoption of full-color OLED displays, creating a critical supply gap for high-performance blue emissive layers that can match the longevity and brightness of their red and green counterparts in commercial devices.
The Novel Approach
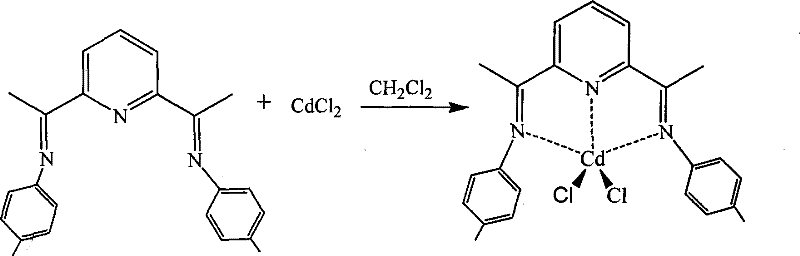
The innovative methodology described in the patent addresses these challenges through the strategic design of sterically hindered pyridine diimine ligands coordinated with cadmium centers. By introducing specific substituents such as methyl, ethyl, or trimethyl groups at the ortho-positions of the N-aryl rings, the synthesis effectively prevents close packing of the molecules, thereby minimizing fluorescence quenching and enhancing solid-state emission. This structural modification allows for the formation of well-defined single crystals with intact crystal forms, which is crucial for consistent film formation in device fabrication. The process utilizes readily available starting materials like 2,6-diacetylpyridine and various substituted anilines, reacting them under mild acidic conditions to form the ligand precursors with high purity.
![Synthesis reaction scheme of 2,6-bis[1-(4-methylphenylimine)ethyl]pyridine ligand via condensation](/insights/img/pyridine-diimine-cadmium-complex-oled-supplier-20260314200327-05.png)
Subsequent coordination with anhydrous cadmium chloride in solvents like dichloromethane or acetonitrile yields the final complexes with remarkable efficiency. The versatility of this approach is evident in its ability to accommodate different substituents, allowing manufacturers to fine-tune the electronic properties and emission wavelengths to meet specific display standards without overhauling the entire synthetic route.
Mechanistic Insights into Cadmium Coordination and Fluorescence
The core of this technology lies in the precise coordination chemistry between the tridentate pyridine diimine ligand and the cadmium ion, which creates a rigid molecular framework conducive to efficient radiative decay. The nitrogen atoms from the pyridine ring and the two imine groups act as electron donors, forming stable chelate rings with the Cd(II) center that restrict intramolecular rotation and vibration, non-radiative pathways that typically dissipate energy as heat. This rigidity is further enhanced by the bulky substituents on the phenyl rings, which enforce a specific geometry that promotes the desired pi-star to pi electronic transitions responsible for the blue fluorescence. Spectroscopic analysis confirms that these complexes exhibit strong emission in the 400-430nm range, a critical window for true blue color reproduction in high-definition displays. The presence of chlorine atoms in the coordination sphere also plays a role in modulating the electronic environment of the metal center, contributing to the overall stability and photophysical characteristics of the material.
Furthermore, the patent highlights that the resulting materials possess excellent monochromatic performance, meaning the emission spectrum is narrow and centered precisely in the blue region, which reduces the need for expensive color filters in display panels. The mechanism also benefits from the absence of heavy metal quenching effects often seen in other transition metal complexes, as the specific electronic configuration of the cadmium-ligand system favors singlet emission. Understanding these mechanistic details is vital for R&D teams aiming to replicate or further optimize the synthesis, as slight deviations in stoichiometry or solvent choice can impact the crystal quality and, consequently, the luminescent efficiency of the final product.
How to Synthesize Pyridine Diimine Cadmium Complex Efficiently
The synthesis protocol outlined in the patent provides a robust and scalable pathway for producing these high-value electronic chemicals, emphasizing reproducibility and ease of operation. The process begins with the condensation of 2,6-diacetylpyridine with the appropriate aniline derivative in absolute methanol, catalyzed by formic acid or glacial acetic acid, followed by refluxing to drive the reaction to completion. After cooling and filtration, the crude ligand is purified through recrystallization, ensuring that only the highest quality precursor enters the complexation stage. The second step involves dissolving the purified ligand and anhydrous CdCl2 in a suitable organic solvent under a nitrogen atmosphere to prevent oxidation or moisture interference, which could degrade the sensitive metal-ligand bonds. Detailed standardized synthesis steps for scaling this process from gram to kilogram quantities are provided below to ensure consistent quality control.
- Condense 2,6-diacetylpyridine with substituted anilines in methanol with acid catalysis to form the diimine ligand.
- Dissolve the purified ligand and anhydrous CdCl2 in organic solvents like dichloromethane or acetonitrile under nitrogen.
- Reflux the mixture at controlled temperatures (38-82°C) and recrystallize to obtain the final blue fluorescent single crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling economic and logistical benefits that extend beyond mere technical performance. The reliance on commodity chemicals such as substituted anilines and simple ketones means that raw material sourcing is straightforward and less susceptible to the volatility associated with exotic or highly specialized reagents. This accessibility translates directly into cost reduction in electronic chemical manufacturing, as the supply chain becomes more resilient and less dependent on single-source suppliers for critical intermediates. Moreover, the high yields reported in the patent, consistently reaching above 84%, imply a significant reduction in waste generation and solvent usage per unit of product, aligning with modern sustainability goals and reducing disposal costs.
- Cost Reduction in Manufacturing: The streamlined two-step process eliminates the need for expensive transition metal catalysts or complex purification columns often required in alternative synthetic routes, leading to substantial cost savings in both capital expenditure and operational overhead. By avoiding the use of precious metals like iridium or platinum, which are common in phosphorescent OLED materials, this cadmium-based approach offers a more budget-friendly alternative without sacrificing luminous intensity. The ability to recycle solvents like dichloromethane and acetonitrile further enhances the economic viability, making the overall cost of goods sold highly competitive for mass-market display applications.
- Enhanced Supply Chain Reliability: The use of stable, shelf-stable reagents ensures that production schedules are not disrupted by the degradation of sensitive starting materials, a common issue in fine chemical synthesis. The robustness of the reaction conditions, which tolerate a range of temperatures and reflux times, provides manufacturing flexibility that allows facilities to optimize throughput based on demand without compromising product quality. This reliability is crucial for maintaining continuous supply lines to panel manufacturers who operate on tight just-in-time delivery schedules, ensuring that downstream production is never halted due to material shortages.
- Scalability and Environmental Compliance: The synthesis is inherently scalable, moving seamlessly from laboratory benchtop experiments to pilot plant and full commercial production without requiring fundamental changes to the reaction engineering. The solvents employed are standard industrial chemicals with well-established recovery and recycling protocols, facilitating compliance with environmental regulations regarding volatile organic compound (VOC) emissions. Additionally, the formation of well-defined crystals simplifies the isolation process, reducing the energy consumption associated with drying and milling, which contributes to a lower carbon footprint for the final OLED material.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridine diimine cadmium complex technology in industrial settings. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details helps stakeholders assess the feasibility of integrating this material into their existing product lines or R&D pipelines.
Q: What is the emission wavelength range of these cadmium complexes?
A: The synthesized pyridine diimine cadmium complexes exhibit strong blue fluorescence with a maximum emission peak wavelength ranging from 400nm to 430nm, specifically tuned by the substituents on the phenyl rings.
Q: How does the luminescence intensity compare to existing materials?
A: According to patent data, these novel complexes demonstrate a luminescence intensity increase of 2 to 10 times compared to conventional blue light-emitting organic materials, addressing a key bottleneck in OLED development.
Q: What solvents are suitable for the complexation reaction?
A: The process utilizes common organic solvents such as dichloromethane, acetonitrile, or tetrahydrofuran (THF), allowing for flexible process optimization based on solubility and recrystallization requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine Diimine Cadmium Complex Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure required to bring this sophisticated blue light-emitting material from patent to production scale. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. We understand the stringent purity specifications required for electronic grade materials and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to excellence means that we do not just supply chemicals; we deliver validated solutions that enhance the performance and reliability of your final OLED devices.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing this supply chain can improve your bottom line. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to be your long-term strategic partner in the electronic materials sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
