Advanced Bisphosphite Ligands for High-Selectivity Hydroformylation and Commercial Scale-Up
The chemical manufacturing landscape is continuously evolving, driven by the relentless pursuit of efficiency and selectivity in catalytic processes. Patent CN101200476B introduces a groundbreaking class of bisphosphite ligands designed specifically for transition metal-catalyzed hydroformylation, addressing critical limitations found in earlier generations of catalysts. This technology represents a significant leap forward for producers of aldehydes and downstream derivatives, offering a pathway to higher purity products and more streamlined operations. By leveraging these advanced ligands, manufacturers can achieve superior control over reaction outcomes, particularly in the selective terminal hydroformylation of olefins. The implications for the global supply chain are profound, as improved catalyst performance directly translates to reduced waste and enhanced resource utilization. For industry leaders seeking to optimize their production capabilities, understanding the nuances of this patented technology is essential for maintaining a competitive edge in the specialty chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional hydroformylation processes often rely on ligand systems that suffer from significant drawbacks, particularly regarding selectivity and stability. Conventional phosphite ligands, such as those described in prior art like EP 0 472 071, frequently exhibit high isomerization activity, which leads to the unwanted conversion of terminal olefins into internal olefins before reaction. This side reaction not only reduces the yield of the desired linear aldehyde but also complicates downstream purification processes, increasing operational costs and energy consumption. Furthermore, many existing ligand systems are highly susceptible to hydrolysis, degrading rapidly in the presence of trace moisture which is often unavoidable in large-scale industrial settings. This instability necessitates stringent drying protocols and frequent catalyst replenishment, creating bottlenecks in production schedules and inflating the total cost of ownership. The inability to maintain high n/iso ratios consistently across different olefin substrates further limits the versatility of these older technologies, forcing manufacturers to utilize separate process lines for different feedstocks.
The Novel Approach
The innovative bisphosphite ligands disclosed in CN101200476B offer a robust solution to these persistent challenges by fundamentally altering the steric and electronic environment around the catalytic metal center. These novel structures are engineered to suppress isomerization activity effectively, ensuring that terminal double bonds remain intact and react preferentially to form the desired normal aldehydes with exceptional selectivity. This high degree of control allows for the processing of complex olefin mixtures without the need for extensive pre-purification, thereby simplifying the overall workflow. Additionally, the enhanced hydrolytic stability of these ligands means they can withstand harsher reaction conditions and tolerate higher levels of impurities without significant degradation. This durability extends the catalyst lifespan and reduces the frequency of replacement, leading to more consistent production runs. By minimizing side reactions and maximizing catalyst longevity, this approach provides a clear pathway to more sustainable and economically viable manufacturing processes for high-value chemical intermediates.
Mechanistic Insights into Bisphosphite-Catalyzed Hydroformylation
The superior performance of these ligands stems from their unique molecular architecture, which optimizes the coordination geometry around the rhodium or cobalt center during the catalytic cycle. The specific arrangement of the bisphosphite groups creates a steric environment that favors the formation of the linear alkyl-rhodium intermediate over the branched counterpart, directly influencing the n/iso product ratio. This geometric control is critical for applications requiring high-purity linear aldehydes, such as the production of plasticizers and detergents where branching can negatively impact material properties. The ligands facilitate the formation of active carbonylhydridophosphite complexes that are both highly reactive towards olefin insertion and resistant to decomposition pathways. This balance between activity and stability is achieved through the careful selection of substituents on the phosphite backbone, which tune the electron density at the metal center. Understanding these mechanistic details is vital for process chemists aiming to fine-tune reaction conditions for specific substrates.
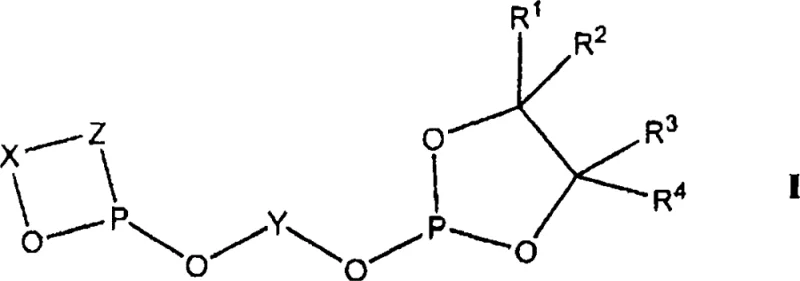
Furthermore, the mechanism involves a delicate interplay between the ligand and the synthesis gas components, where the ligand stability prevents the formation of inactive metal clusters that often plague less robust systems. The resistance to hydrolysis ensures that the phosphite groups remain coordinated to the metal throughout the reaction, preventing the release of free phosphoric acid which can corrode equipment and poison the catalyst. This mechanistic robustness allows for operation at elevated temperatures and pressures without sacrificing selectivity, enabling faster reaction rates and higher throughput. For R&D directors, this means the ability to push process boundaries safely, exploring more aggressive operating windows to maximize plant capacity. The detailed understanding of how substituents R1 through R4 influence the electronic properties provides a roadmap for future ligand customization, allowing for tailored solutions for specific niche applications within the fine chemical sector.
How to Synthesize Bisphosphite Ligands Efficiently
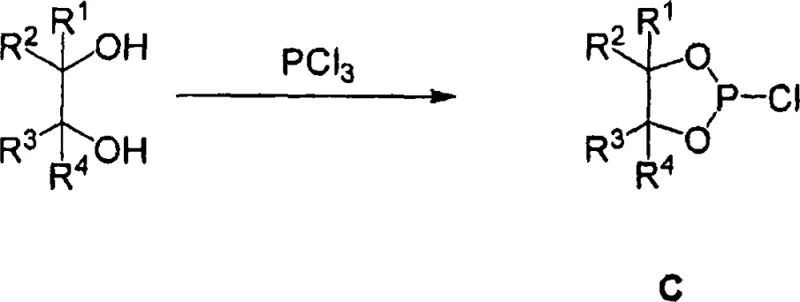
The preparation of these high-performance ligands follows a logical and scalable synthetic route that utilizes readily available starting materials and standard laboratory techniques. The process generally involves the stepwise reaction of specific diols or alcohol amines with phosphorus trihalides to generate reactive intermediates, which are then coupled to form the final bisphosphite structure. This modular approach allows for significant flexibility in tuning the ligand properties by varying the diol components, enabling the production of a diverse library of catalysts from a common set of precursors. The synthesis is typically conducted under inert atmosphere conditions to prevent premature oxidation, utilizing solvents such as tetrahydrofuran or toluene which are common in industrial chemical processing. The ability to isolate stable intermediates provides quality control checkpoints, ensuring that the final product meets stringent purity specifications before being deployed in sensitive catalytic applications. This straightforward synthetic accessibility is a key factor in the commercial viability of the technology.
- React diol or alcohol amine with phosphorus trihalide to generate the first intermediate.
- React tetraaryl glycol with phosphorus trihalide to form the monohalophosphite intermediate.
- Combine intermediates to form the final bisphosphite ligand structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this advanced ligand technology offers substantial strategic benefits that extend beyond simple chemical performance. The enhanced stability and selectivity of the catalyst system directly contribute to significant cost reduction in manufacturing by minimizing raw material waste and reducing the load on downstream purification units. Because the ligands suppress isomerization, the resulting product stream is cleaner, requiring less energy-intensive distillation or crystallization steps to achieve target purity levels. This efficiency gain translates into lower utility costs and a smaller carbon footprint, aligning with corporate sustainability goals. Moreover, the robustness of the ligands reduces the frequency of catalyst change-outs, leading to less downtime and more predictable production schedules. These operational improvements create a more resilient supply chain capable of meeting fluctuating market demands without the risk of unexpected process failures or quality deviations.
- Cost Reduction in Manufacturing: The implementation of these ligands eliminates the need for expensive metal scavenging steps often required with less stable catalyst systems, resulting in substantial cost savings. By maintaining high catalytic activity over extended periods, the total consumption of precious metals like rhodium is optimized, reducing the working capital tied up in catalyst inventory. The reduction in byproduct formation also means that raw olefin feedstocks are utilized more efficiently, lowering the cost per unit of the final aldehyde product. These cumulative savings enhance the overall profit margin for manufacturers operating in highly competitive markets where price sensitivity is a critical factor for success.
- Enhanced Supply Chain Reliability: The high hydrolytic stability of the ligands ensures consistent performance even when feedstock quality varies, reducing the risk of production delays caused by catalyst deactivation. This reliability allows supply chain planners to maintain leaner inventory levels of catalyst reserves, freeing up storage space and reducing holding costs. The ability to process a wider range of olefin feedstocks without re-optimizing the catalyst system provides flexibility in sourcing raw materials, mitigating the risk of supply disruptions from specific vendors. This adaptability is crucial for maintaining continuous operations in a global market subject to logistical challenges and geopolitical uncertainties.
- Scalability and Environmental Compliance: The synthetic route for these ligands is amenable to large-scale production, ensuring a stable supply of the catalyst itself for long-term commercial operations. The reduction in waste generation and energy consumption associated with the improved process efficiency supports compliance with increasingly stringent environmental regulations. By minimizing the release of volatile organic compounds and hazardous byproducts, manufacturers can reduce their environmental liability and improve their standing with regulatory bodies. This proactive approach to environmental stewardship not only avoids potential fines but also enhances the brand reputation of the company among eco-conscious customers and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bisphosphite ligand technology in industrial hydroformylation processes. These answers are derived directly from the patent data and provide clarity on performance expectations and operational requirements. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this catalyst system into existing production lines. The information covers aspects ranging from substrate compatibility to storage conditions, ensuring a comprehensive overview for decision-makers.
Q: How does this ligand improve hydroformylation selectivity?
A: The novel bisphosphite structure minimizes isomerization activity, ensuring terminal double bonds react preferentially to form linear aldehydes with high n/iso ratios.
Q: What are the stability advantages over prior art?
A: These ligands exhibit significantly higher hydrolytic stability compared to conventional phosphites, reducing decomposition during storage and reaction.
Q: Can this technology be scaled for industrial production?
A: Yes, the synthesis uses standard Schlenk techniques and common solvents, facilitating straightforward scale-up from laboratory to commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphosphite Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical innovation, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex catalytic systems. Our technical team possesses the expertise to adapt the synthesis of these advanced bisphosphite ligands to meet your specific volume and purity requirements, ensuring stringent purity specifications are met for every batch. We understand the critical nature of catalyst consistency in hydroformylation processes and employ rigorous QC labs to verify the structural integrity and performance of our products. By partnering with us, you gain access to a supply chain that prioritizes reliability and technical support, enabling you to focus on your core business objectives while we manage the complexities of catalyst manufacturing. Our commitment to quality ensures that your production processes run smoothly and efficiently.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your unique application needs. Our experts are ready to provide a Customized Cost-Saving Analysis that demonstrates the potential economic benefits of switching to our advanced ligand systems. Whether you are looking to optimize an existing process or develop a new product line, our solutions are designed to deliver measurable value and performance improvements. Let us help you unlock the full potential of your hydroformylation operations with our cutting-edge catalyst technology.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
