Industrial Scale-Up of Levofosfomycin via Novel Configuration Inversion Technology
The pharmaceutical industry constantly seeks efficient pathways to produce high-value antibiotics, and the synthesis of levofosfomycin represents a critical area of innovation due to its broad-spectrum efficacy against urinary tract and respiratory infections. Patent CN101928301B introduces a groundbreaking method for synthesizing levofosfomycin dextrophenethylamine salt starting from dextrofosfomycin levophenethylamine salt, a material often considered a byproduct or waste in traditional chiral resolution processes. This technology addresses a significant inefficiency in the current manufacturing landscape where valuable chiral intermediates are discarded, leading to increased costs and environmental burdens. By employing a sophisticated configuration inversion strategy, this patent outlines a route that transforms the less desirable stereoisomer into the pharmacologically active form with high fidelity. The process is characterized by mild reaction conditions, the avoidance of highly toxic reagents such as dicyclohexylcarbodiimide (DCC), and a robust sequence of protection and deprotection steps that ensure the integrity of the sensitive epoxide ring. For R&D directors and process chemists, this represents a viable alternative to fermentation or direct synthesis, offering a pathway to maximize atom economy and reduce the overall carbon footprint of antibiotic production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of optically pure fosfomycin salts has relied heavily on fermentation followed by chiral resolution, a process that inherently generates a 50% waste stream of the unwanted enantiomer. In many existing protocols, the dextrofosfomycin levophenethylamine salt, which contains the incorrect stereochemistry for the final active pharmaceutical ingredient, is merely processed to recover the chiral amine resolver, while the fosfomycin acid component is discarded into wastewater. This practice not only represents a massive loss of raw material value but also exacerbates the difficulty of wastewater treatment due to the high organic load and phosphorus content. Furthermore, older chemical synthesis methods often resort to harsh conditions or expensive coupling agents like DCC, which pose severe toxicity risks and complicate purification processes on a commercial scale. The reliance on hydrogenation for deprotection in some prior art methods also introduces the risk of reductive ring-opening of the sensitive oxirane moiety, leading to lower yields and complex impurity profiles that are difficult to control under Good Manufacturing Practice (GMP) standards.
The Novel Approach
The novel approach detailed in this patent circumvents these issues by treating the "waste" isomer as a valuable starting material for configuration inversion. Instead of discarding the dextrofosfomycin derivative, the method subjects it to a series of controlled chemical transformations that effectively flip its stereochemistry to match the active levofosfomycin profile. This is achieved through a clever sequence involving the formation of a di-tert-alcohol ester, which protects the phosphonic acid group while allowing for selective manipulation of the epoxide ring. The process avoids the use of hazardous hydrogenation steps for deprotection, opting instead for acid-catalyzed hydrolysis and cyclization which are far more predictable and scalable. By integrating this inversion step into the production workflow, manufacturers can theoretically double the yield from their initial fermentation or resolution batches, turning a cost center (waste disposal) into a profit center (additional API production).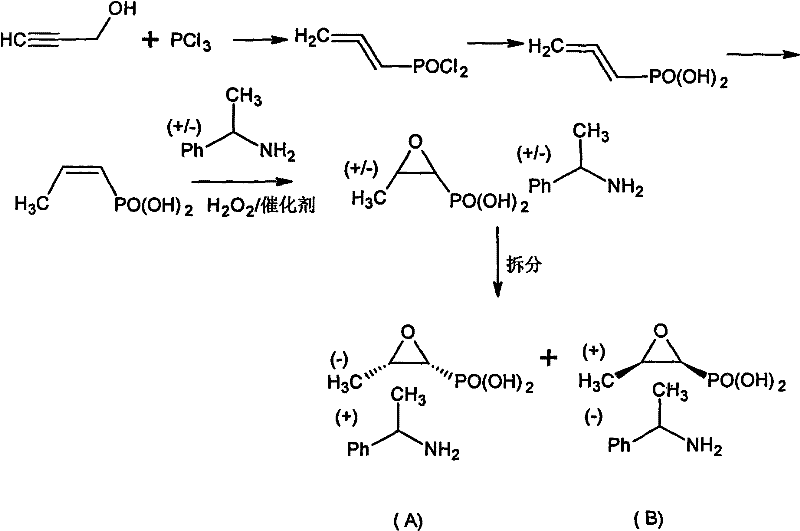
Mechanistic Insights into Stereochemical Inversion and Epoxide Manipulation
The core of this synthetic strategy lies in the precise manipulation of the epoxide ring and the phosphonic acid functionality to achieve stereochemical inversion without degrading the molecule. The process begins with the conversion of the starting salt into a dialkali metal salt, which is then transformed into an acyl chloride using reagents like thionyl chloride or triphosgene. This activation step is crucial as it allows for the subsequent esterification with bulky tertiary alcohols, such as tert-butanol, forming a stable di-tert-alcohol ester. This steric bulk provides essential protection for the phosphonate group during the subsequent acidic and basic treatments. The pivotal moment in the mechanism occurs during the hydrolysis of the epoxy ring under weak acid catalysis, which opens the ring to form a dihydroxy intermediate. This open-chain structure allows for the introduction of a leaving group, specifically an alkyl sulfonyloxy group, via reaction with sulfonyl chlorides like tosyl chloride.
Once the sulfonate ester is formed, the stage is set for the actual inversion of configuration. Under strong acid catalysis, the tert-alcohol protecting groups are removed, and subsequent treatment with a base induces an intramolecular nucleophilic substitution. The hydroxyl group attacks the carbon bearing the sulfonate leaving group, forcing the closure of the epoxide ring with inverted stereochemistry. This SN2-like displacement ensures that the spatial arrangement of the atoms is flipped, converting the (1R, 2S) configuration of the starting material into the desired (1S, 2S) configuration of levofosfomycin. The final step involves salt formation with dextro-phenylethylamine, which not only isolates the product but also serves as a final purification step through recrystallization. This mechanistic pathway is highly advantageous because it avoids the use of transition metal catalysts that could leave toxic residues, ensuring the final API intermediate meets stringent purity specifications required for parenteral administration.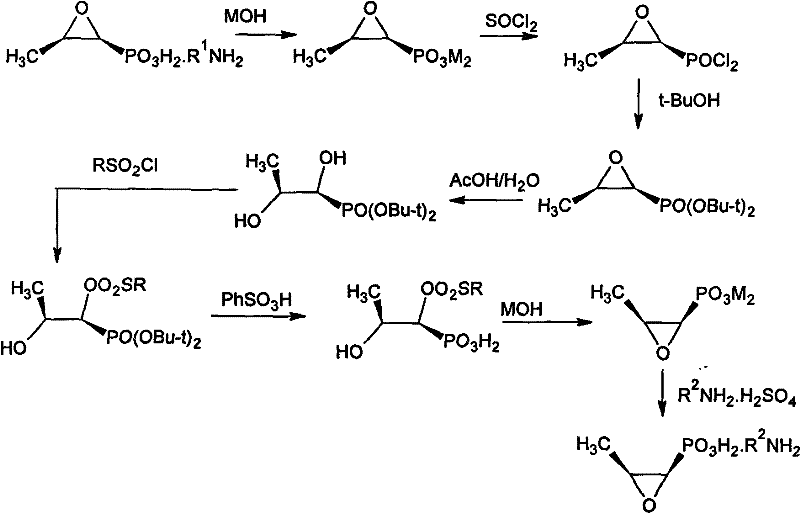
How to Synthesize Levofosfomycin Dextrophenethylamine Salt Efficiently
The synthesis of this critical antibiotic intermediate requires careful control of reaction parameters to maintain high optical purity and yield throughout the multi-step sequence. The patented procedure outlines a robust workflow that begins with the preparation of the anhydrous dialkali metal salt, followed by acyl chlorination and esterification to protect the sensitive functional groups. The subsequent ring-opening, sulfonylation, and cyclization steps must be performed under strictly controlled temperatures and pH conditions to prevent side reactions such as polymerization or over-hydrolysis. For process chemists looking to implement this technology, the detailed standardized synthesis steps provided in the patent serve as a foundational guide for optimizing reaction times and reagent stoichiometry.
- Preparation of dextrorotation phosphonomycin dialkali metal salt from the starting levophenethylamine salt using alkali metal hydroxide.
- Conversion to acyl chloride followed by esterification with tertiary alcohol to form the di-tert-alcohol ester.
- Hydrolytic ring opening, sulfonylation, acidolysis, and final cyclization to invert configuration and form the target salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers transformative potential for reducing the cost of goods sold (COGS) for fosfomycin-based antibiotics. By enabling the conversion of low-value waste streams into high-value active ingredients, the process fundamentally alters the economics of production, allowing manufacturers to extract significantly more value from the same amount of initial raw materials. This efficiency gain translates directly into improved margins and a more competitive pricing structure in the global generic pharmaceutical market. Furthermore, the reliance on commodity chemicals like thionyl chloride, tertiary alcohols, and common sulfonic acids ensures that the supply chain is not dependent on exotic or single-source catalysts that could introduce bottlenecks. The elimination of toxic reagents like DCC also simplifies the regulatory compliance landscape, reducing the costs associated with environmental health and safety (EHS) management and waste disposal.
- Cost Reduction in Manufacturing: The primary economic driver of this process is the valorization of waste isomers, which effectively lowers the raw material cost per kilogram of the final active ingredient. By avoiding the need for expensive chiral catalysts or complex enzymatic resolutions for every batch, the overall operational expenditure is drastically simplified. The use of standard industrial solvents and reagents further contributes to cost stability, shielding the production process from the volatility associated with specialty chemical markets. Additionally, the high recovery rates reported in the embodiments suggest that material losses are minimized, ensuring that the theoretical cost savings are realized in practical commercial operations.
- Enhanced Supply Chain Reliability: The robustness of this chemical synthesis route enhances supply chain security by reducing dependency on biological fermentation cycles, which can be susceptible to contamination and variability. The ability to synthesize the active isomer from the inactive one provides a strategic buffer, allowing manufacturers to balance inventory levels more effectively and respond to sudden spikes in demand for urinary tract infection treatments. Since the reagents involved are widely available in the global chemical market, the risk of supply disruption due to geopolitical or logistical issues is significantly mitigated compared to processes relying on niche biocatalysts.
- Scalability and Environmental Compliance: This method is inherently designed for industrial scale-up, utilizing unit operations such as filtration, distillation, and crystallization that are standard in multipurpose pharmaceutical plants. The avoidance of heavy metal catalysts and the reduction of hazardous waste streams align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. This compliance advantage reduces the administrative burden on supply chain teams regarding waste permitting and disposal, facilitating smoother audits and faster time-to-market for new generic filings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this configuration inversion technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What is the primary advantage of this synthesis method over traditional fermentation?
A: This chemical synthesis method allows for the utilization of waste isomers from chiral resolution processes, significantly reducing raw material costs and environmental waste compared to discarding unwanted enantiomers.
Q: How is high optical purity achieved in this process?
A: High optical purity (>99%) is achieved through a rigorous stereochemical inversion mechanism involving epoxide ring opening and reclosure, combined with final recrystallization steps that remove impurities.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions, common reagents like thionyl chloride and tertiary alcohols, and avoids toxic catalysts like DCC, making it highly scalable and compliant with industrial safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levofosfomycin Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient antibiotic intermediate synthesis in the current global healthcare landscape. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex stereochemical inversions like the one described in CN101928301B can be translated from the laboratory bench to the manufacturing floor with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of levofosfomycin meets the highest international pharmacopoeia standards. Our commitment to quality ensures that our partners receive materials that are ready for immediate formulation, minimizing downstream processing risks.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for fosfomycin derivatives. By leveraging our technical expertise, you can access a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive both profitability and product excellence. Let us be your partner in delivering life-saving antibiotics to the market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
