Advanced Manufacturing of High-Purity Sodium Rabeprazole for Global Supply Chains
Advanced Manufacturing of High-Purity Sodium Rabeprazole for Global Supply Chains
The pharmaceutical landscape for Proton Pump Inhibitors (PPIs) is constantly evolving, with a critical demand for intermediates that offer superior stability and purity profiles. Patent CN102993179A introduces a groundbreaking preparation method for Sodium Rabeprazole that addresses long-standing challenges in moisture control and chemical stability. This innovation utilizes a novel solvent system comprising a mixture of aromatic hydrocarbons and alkyl alcohols, fundamentally shifting the paradigm from energy-intensive freeze-drying to efficient crystallization. For R&D Directors and Supply Chain Heads, this represents a significant opportunity to enhance the quality of the final drug product while simplifying the manufacturing workflow. The technology ensures that the resulting sodium salt exhibits low moisture content and exceptional stability, making it an ideal candidate for prolonged storage and pharmaceutical formulation without the risk of degradation commonly associated with earlier generation synthesis routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sodium Rabeprazole has been plagued by significant technical bottlenecks that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional methods often rely on aqueous sodium hydroxide solutions followed by freeze-drying, a process that demands substantial facility investment and incurs high power costs due to the extended production cycles involved. Alternatively, methods utilizing single solvents like water or alcohol require repeated concentration steps to induce crystallization, which poses a severe risk of product decomposition and peroxidation due to prolonged exposure to heat. Furthermore, the use of dichloromethane extraction systems often leads to emulsion formation, requiring excessive settling times of over ten hours to achieve clear layering, thereby drastically reducing throughput. These conventional approaches frequently result in products with high moisture content and variable color, creating substantial difficulties in meeting the rigorous quality specifications required by global regulatory bodies for high-purity OLED material and pharmaceutical standards alike.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach disclosed in the patent leverages a homogeneous reaction system within a mixed solvent of aromatic hydrocarbons and alkyl alcohols. This strategic solvent selection eliminates the need for distillation, extraction, or freeze-drying, allowing for direct crystallization upon the addition of alkane or ether anti-solvents. The process avoids the formation of thick, syrupy residues that typically adhere to reactor walls, ensuring smooth operation and consistent batch-to-batch reproducibility. By maintaining a homogeneous phase throughout the reaction, the method significantly reduces the potential for localized overheating and subsequent degradation of the sensitive benzimidazole structure. This streamlined workflow not only simplifies the operational complexity but also inherently enhances the safety profile of the manufacturing process by reducing the reliance on halogenated solvents and energy-intensive drying steps, offering a robust pathway for cost reduction in electronic chemical manufacturing and pharmaceutical production environments.
Mechanistic Insights into Salt Formation and Crystallization
The core of this technological advancement lies in the precise manipulation of solubility parameters during the salification of Rabeprazole. The reaction proceeds by dissolving Rabeprazole and a sodium-containing base, preferably sodium hydroxide, in a mixed system of C1 to C4 alkyl alcohols and aromatic hydrocarbons such as toluene or xylene. The molar ratio of Rabeprazole to the base is carefully controlled between 1:1.0 and 1:1.5 to ensure complete conversion while minimizing excess reagent carryover. The alkyl alcohol component serves a dual purpose: it facilitates the initial dissolution of reactants and subsequently acts as a dehydration agent during the crystallization phase, effectively adsorbing water molecules that could otherwise compromise the stability of the final salt. This mechanism is critical for achieving the reported moisture content of less than 1% (KF), a specification that is notoriously difficult to maintain in hygroscopic PPI salts. The subsequent addition of alkanes or ethers reduces the solubility of the formed sodium salt, triggering nucleation and crystal growth under controlled temperature conditions.
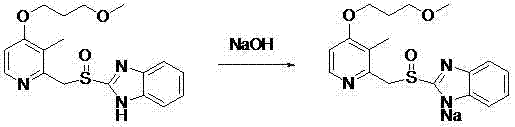
Following the initial reaction, the process incorporates a sophisticated purification strategy involving temperature-programmed crystallization and silica gel bed filtration. The solution is heated to 30-40°C and then gradually cooled to 0-10°C, a thermal profile designed to maximize crystal lattice integrity and exclude impurities from the growing crystal structure. Crucially, the filtration through a silica gel bed acts as a polishing step, specifically targeting the removal of peroxidation impurities that are common byproducts in the oxidation steps of benzimidazole synthesis. This step ensures that the final product achieves a chemical purity greater than 99.5% with single impurities controlled below 0.1%. The resulting crystals exhibit a high melting point and superior stability under high temperature, high humidity, and illumination conditions, validating the efficacy of the solvent system in protecting the labile sulfinyl group from hydrolysis and oxidation during the isolation phase.
How to Synthesize Sodium Rabeprazole Efficiently
The implementation of this synthesis route requires precise adherence to the solvent ratios and temperature profiles outlined in the patent data to ensure optimal yield and quality. The process begins with the dissolution of starting materials in the specific aromatic-alcohol mixture, followed by the controlled addition of anti-solvents to induce precipitation. Detailed standardized synthetic steps are essential for maintaining the homogeneity of the reaction and preventing the formation of amorphous solids that could trap impurities. Operators must monitor the cooling rates closely to facilitate the formation of well-defined crystals that filter easily and wash cleanly. The following guide outlines the critical operational parameters required to replicate the high-purity results demonstrated in the patent examples, ensuring that the commercial scale-up of complex polymer additives or pharmaceutical intermediates can proceed with minimal technical risk.
- Dissolve Rabeprazole and a sodium-containing base in a mixed solvent system comprising C1-C4 alkyl alcohol and aromatic hydrocarbon.
- Add alkane and/or ether solvents to the reaction solution to induce crystallization without concentration.
- Perform solid-liquid separation followed by temperature-programmed cooling to obtain high-purity Sodium Rabeprazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis method translates into tangible operational efficiencies and risk mitigation strategies. By eliminating the need for freeze-drying and repeated concentration steps, the process significantly reduces energy consumption and equipment wear, leading to substantial cost savings in the overall manufacturing budget. The simplified workflow also shortens the production cycle time, enhancing the responsiveness of the supply chain to market demands without compromising on quality standards. Furthermore, the use of readily available solvents like toluene and ethanol reduces dependency on specialized or hazardous reagents, thereby improving supply chain reliability and reducing the logistical complexities associated with raw material sourcing. This robustness ensures a continuous supply of high-quality intermediates, critical for maintaining the production schedules of downstream pharmaceutical manufacturers.
- Cost Reduction in Manufacturing: The elimination of energy-intensive unit operations such as freeze-drying and vacuum distillation directly lowers the utility costs associated with production. Additionally, the homogeneous reaction system minimizes product loss due to adhesion to reactor walls, improving the overall mass balance and yield efficiency. The removal of expensive transition metal catalysts or complex purification resins further contributes to a leaner cost structure, allowing for competitive pricing without sacrificing margin. These cumulative efficiencies create a compelling economic case for switching to this technology, driving significant value for stakeholders focused on cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The use of common industrial solvents ensures that raw material availability is not a bottleneck, safeguarding against supply disruptions that can plague specialty chemical supply chains. The robustness of the process against minor variations in operating conditions means that batch failures are rare, ensuring a consistent output of material that meets specification every time. This reliability is paramount for long-term supply agreements, where consistency is valued as highly as price. By stabilizing the production process, manufacturers can offer more predictable lead times, reducing the need for safety stock and optimizing inventory management for their clients.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial production without the need for re-engineering the core chemistry. The reduction in solvent usage and the avoidance of halogenated solvents like dichloromethane align with increasingly stringent environmental regulations and green chemistry principles. This compliance reduces the burden on waste treatment facilities and lowers the environmental footprint of the manufacturing site. The ability to scale up complex synthesis routes while maintaining environmental standards is a key differentiator in the modern chemical industry, appealing to partners who prioritize sustainability in their supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific Sodium Rabeprazole synthesis technology. These insights are derived directly from the patent data and are intended to clarify the operational advantages and quality improvements offered by this method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the rigorous testing and validation performed during the patent development phase, ensuring accuracy and reliability for decision-makers.
Q: How does the new solvent system improve product stability compared to traditional methods?
A: The novel mixed solvent system of aromatic hydrocarbons and alkyl alcohols eliminates the need for concentration and freeze-drying, which are known to cause decomposition and peroxidation. This results in a product with significantly lower moisture content (KF < 1%) and superior chemical stability under high temperature and humidity conditions.
Q: What are the primary impurity control mechanisms in this synthesis route?
A: The process incorporates a silica gel bed filtration step specifically designed to purify the reaction liquid. This effectively removes peroxidation impurities that are difficult to eliminate in conventional salification processes, ensuring single impurity levels remain below 0.1% and overall purity exceeds 99.5%.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method avoids complex operations like repeated concentration, extraction, or freeze-drying. The homogeneous reaction system prevents the formation of thick syrups that adhere to reactor walls, making filtration and scale-up significantly easier and more reliable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium Rabeprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of stable and effective pharmaceutical formulations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering Sodium Rabeprazole that meets stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our capability to implement advanced solvent systems and crystallization technologies allows us to offer products with superior stability profiles, directly addressing the moisture and degradation challenges highlighted in recent patent literature.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this advanced synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial viability. Let us be your partner in delivering excellence in fine chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
