Advanced Resin Adsorption Technology for Commercial Scale Mupirocin Calcium Production
The pharmaceutical industry continuously seeks robust methodologies to enhance the purity and stability of active ingredients, particularly for topical antibiotics where consistency is paramount. Patent CN101511826B introduces a transformative approach to the preparation and purification of Mupirocin Calcium, addressing critical bottlenecks found in traditional fermentation downstream processing. This technology leverages hydrophobic adsorption resins to isolate pseudomonic acid directly from cell-depleted fermentation broths, followed by an innovative on-resin calcium exchange mechanism. By integrating purification and salt formation into a unified continuous process, this method drastically reduces the reliance on hazardous organic solvents and complex crystallization steps that have historically plagued the supply chain. For R&D Directors and Procurement Managers, this represents a significant opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates while ensuring rigorous compliance with environmental standards. The technical breakthrough lies in the selective binding capabilities of modified polystyrene and acrylic resins, which allow for the precise removal of fermentation impurities before the final elution of the active calcium salt.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of Mupirocin and its derivatives has relied heavily on liquid-liquid extraction techniques involving solvents such as methyl isobutyl ketone (MIBK) and isobutyl acetate. These conventional processes, as documented in prior art like US 4,222,942 and US 4,289,703, necessitate multiple phase separations and extensive solvent evaporation steps that introduce significant operational risks. The use of large volumes of volatile organic compounds not only escalates production costs due to solvent recovery requirements but also creates substantial environmental liabilities regarding waste disposal and emissions. Furthermore, traditional crystallization methods often struggle to achieve the desired thermal stability required for final dosage forms, frequently resulting in amorphous products that are hygroscopic and difficult to handle during tablet or ointment formulation. The reliance on barium salts in some older protocols also introduces the risk of heavy metal contamination, necessitating expensive and time-consuming purification steps to meet stringent pharmacopeial limits for residual metals. These inefficiencies collectively contribute to longer lead times and reduced overall process reliability for commercial scale-up of complex pharmaceutical additives.
The Novel Approach
The methodology described in CN101511826B circumvents these challenges by utilizing a solid-phase extraction strategy that combines purification and salt conversion in a single column operation. Instead of extracting the antibiotic into an organic phase, the process adsorbs the pseudomonate anion directly onto a hydrophobic resin matrix from an aqueous fermentation broth. This shift from liquid-liquid to solid-liquid interaction allows for the selective retention of the target molecule while washing away polar impurities and cellular debris with high efficiency. The subsequent exposure of the bound resin to a calcium-containing solution facilitates an immediate ion exchange, converting the adsorbed acid into the thermally stable calcium salt directly on the solid support. This eliminates the need for intermediate isolation of the free acid and avoids the use of toxic barium reagents entirely. The result is a streamlined workflow that significantly simplifies the downstream processing train, offering a more environmentally attractive and commercially viable pathway for producing high-purity Mupirocin Calcium suitable for sensitive topical applications.
Mechanistic Insights into Hydrophobic Adsorption and Ion Exchange
The core of this technological advancement relies on the specific interaction between the pseudomonic acid molecule and the hydrophobic surface of the adsorbent resin. Mupirocin, with its distinct lipophilic side chains and polar functional groups, exhibits strong affinity for modified polystyrene or acrylic matrices under controlled pH conditions. By adjusting the fermentation broth to a pH where the molecule is fully deprotonated, typically around pH 7.5 to 8.3, the pseudomonate anion becomes highly susceptible to hydrophobic adsorption. The addition of salting-out agents like ammonium sulfate further enhances this interaction by reducing the solubility of the organic molecule in the aqueous phase, thereby driving it onto the resin surface with high binding capacity. This mechanism ensures that even dilute fermentation broths can be processed effectively, concentrating the active ingredient while leaving behind hydrophilic fermentation byproducts in the flow-through. The reversibility of this binding is crucial, allowing for the subsequent elution of the purified product without degrading the sensitive epoxide and ester functionalities inherent to the Mupirocin structure.
Once the target molecule is secured on the solid carrier, the conversion to the calcium salt occurs through a precise stoichiometric exchange. When a solution containing calcium ions, such as calcium acetate or calcium chloride, is passed through the column, the divalent calcium cations displace the monovalent cations associated with the adsorbed pseudomonate. This on-resin reaction ensures that the molecular equivalence between the antibiotic and the calcium ion is maintained accurately, preventing the formation of mixed salts or incomplete conversion which could compromise stability. The crystalline nature of the resulting Mupirocin Calcium is superior to the free acid form, offering enhanced thermal stability that is critical for storage and formulation. This mechanistic control over the salt formation process minimizes the generation of amorphous impurities and ensures a consistent polymorphic form, which is a key quality attribute for regulatory approval and batch-to-batch reproducibility in API manufacturing.
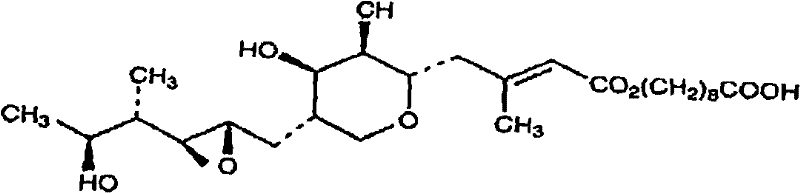
How to Synthesize Mupirocin Calcium Efficiently
Implementing this resin-based purification protocol requires careful attention to the preparation of the fermentation feed and the selection of the appropriate adsorbent material. The process begins with the clarification of the fermentation broth to remove biomass, followed by pH adjustment and the addition of ammonium sulfate to optimize loading conditions. The solution is then passed through a column packed with a hydrophobic resin such as XAD7HP or XAD1600, where the mupirocin is captured. Following a wash step to remove impurities, a calcium solution is introduced to effect the salt conversion, and the final product is eluted using a methanol-water mixture. Detailed standard operating procedures regarding flow rates, resin bed volumes, and elution profiles are critical for maximizing yield and purity. For a comprehensive breakdown of the specific experimental parameters and step-by-step instructions, please refer to the technical guide below.
- Ferment Pseudomonas fluorescens and clarify the broth to obtain a cell-depleted mupirocin preparation.
- Adsorb the pseudomonate salt onto a hydrophobic solid carrier resin such as XAD7HP or XAD1600.
- Treat the bound resin with a calcium-containing solution, wash impurities, and elute purified Mupirocin Calcium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resin adsorption technology offers substantial strategic benefits beyond mere technical performance. The elimination of hazardous solvents like MIBK and the removal of heavy metal reagents such as barium salts drastically simplify the waste management infrastructure required at the manufacturing site. This reduction in chemical complexity translates directly into lower operational expenditures related to solvent recovery, hazardous waste disposal, and environmental compliance monitoring. Furthermore, the ability to process dilute fermentation broths directly without extensive concentration steps reduces energy consumption and shortens the overall production cycle time. These efficiencies contribute to a more resilient supply chain capable of responding to market demands with greater agility and reduced risk of production stoppages due to regulatory or environmental constraints.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers manufacturing costs by removing the need for expensive solvent extraction and recovery systems. By consolidating purification and salt formation into a single unit operation, the facility reduces labor hours, equipment footprint, and utility consumption. The avoidance of toxic reagents also minimizes the costs associated with safety protocols and specialized handling, leading to substantial cost savings in the overall production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing widely available hydrophobic resins and common calcium salts ensures that the raw material supply chain is robust and less susceptible to geopolitical or market fluctuations. The simplified process flow reduces the number of critical process parameters that can fail, thereby increasing the reliability of batch outcomes. This stability is crucial for maintaining continuous supply to downstream formulators and ensuring that lead times for high-purity APIs remain consistent and predictable throughout the year.
- Scalability and Environmental Compliance: The column-based nature of this technology is inherently scalable, allowing for seamless transition from laboratory pilot studies to multi-ton commercial production without significant process redesign. The reduction in volatile organic compound emissions and hazardous waste generation aligns perfectly with modern green chemistry initiatives and strict environmental regulations. This compliance advantage mitigates regulatory risk and enhances the corporate sustainability profile, making the manufacturing process more attractive to environmentally conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the resin-based purification of Mupirocin Calcium. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the feasibility and benefits of this approach. Understanding these details is essential for technical teams evaluating the integration of this method into existing production lines or for procurement specialists assessing the quality implications of the final product.
Q: What are the advantages of resin adsorption over solvent extraction for Mupirocin?
A: Resin adsorption eliminates the need for large volumes of hazardous organic solvents like MIBK, significantly reducing environmental waste and improving operator safety while maintaining high recovery yields.
Q: How does this method ensure the correct stoichiometry of the calcium salt?
A: By exposing the adsorbed pseudomonate directly to a calcium-containing solution on the solid support, the method ensures complete salt exchange and correct molecular equivalence between mupirocin and calcium ions.
Q: Is the resulting Mupirocin Calcium suitable for topical pharmaceutical formulations?
A: Yes, the process yields a thermally stable crystalline form with high specific activity, making it ideal for stable ointments and creams that require robust shelf-life performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mupirocin Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced purification technologies to meet the evolving demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the resin adsorption method for Mupirocin Calcium are implemented with precision and reliability. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of API or intermediate meets the highest international standards. Our commitment to technical excellence allows us to deliver products that not only satisfy regulatory requirements but also enhance the performance of your final formulations.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of these superior processing techniques. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our advanced capabilities can support your long-term strategic goals. By partnering with us, you gain access to a reliable source of high-quality chemicals backed by deep technical expertise and a commitment to sustainable manufacturing practices.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
