Scalable Production of High-Purity Oritavancin Intermediate A82846B via Novel Macroporous Resin Technology
Scalable Production of High-Purity Oritavancin Intermediate A82846B via Novel Macroporous Resin Technology
The pharmaceutical industry's demand for next-generation glycopeptide antibiotics continues to surge, driven by the critical need to combat resistant gram-positive bacterial infections. At the heart of this therapeutic class lies Oritavancin, a potent agent whose commercial viability depends heavily on the efficient supply of its key precursor, A82846B. Recent intellectual property developments, specifically patent CN106928323B, have unveiled a transformative preparation method that addresses long-standing bottlenecks in the isolation and purification of this complex intermediate. This technical insight report analyzes the breakthrough methodology, which leverages pH-controlled macroporous adsorption resin technology to replace inefficient cation exchange processes. For R&D Directors and Supply Chain Heads, understanding this shift is paramount, as it dictates the feasibility of scaling production from laboratory benchmarks to multi-ton commercial outputs without compromising the stringent purity profiles required for FDA-approved drug substances.
The significance of this patent extends beyond mere academic interest; it represents a tangible pathway to stabilizing the global supply chain for glycopeptide intermediates. Traditional methods have struggled with the structural similarities between A82846B and its co-fermentation byproducts, often resulting in suboptimal yields and excessive solvent waste. By integrating precise pH modulation with advanced resin chromatography, the disclosed method achieves a level of selectivity previously unattainable with standard ion-exchange techniques. This report will dissect the mechanistic advantages of this approach, providing procurement managers and technical leaders with the data necessary to evaluate potential partnerships with a reliable glycopeptide antibiotic intermediate supplier capable of executing these sophisticated purification protocols at an industrial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of A82846B from fermentation broths has been plagued by significant technical inefficiencies that hinder cost-effective manufacturing. Prior art, including European patent EP0280570A2 and US4845194, predominantly relies on cation exchange resins for the initial enrichment of the target molecule. However, extensive experimental data reveals that cationic resins exhibit notoriously low adsorption efficiency for this specific glycopeptide structure, leading to substantial product leakage during the loading and washing phases. This leakage not only depresses the overall yield but also necessitates complex downstream processing to recover lost material, thereby inflating operational costs. Furthermore, the structural homology between A82846B and its analogs, A82846A and A82846C, makes separation exceptionally difficult using non-selective ion-exchange mechanisms.
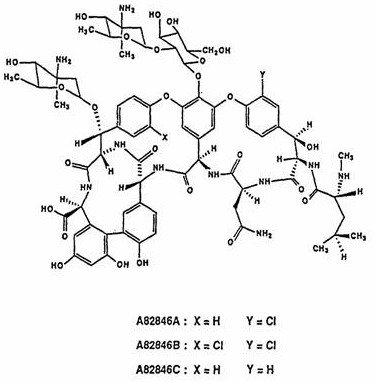
The challenge is visually evident when examining the molecular architecture, where minor variations in halogen and hydrogen substituents dictate biological activity yet complicate physical separation. Conventional methods often require multiple sequential resin columns and preparative high-performance liquid chromatography (HPLC) steps to achieve acceptable purity, a process that is both time-consuming and solvent-intensive. The reliance on large volumes of organic solvents for elution and regeneration creates a substantial environmental burden, generating significant waste liquid that requires costly treatment before disposal. Additionally, the multi-step nature of these traditional protocols increases the risk of product degradation and contamination, posing a threat to the consistent quality required for pharmaceutical grade intermediates. Consequently, the total yield of the latter purification steps in prior art has been reported as low as 29 percent, rendering the process economically unsustainable for large-scale commercial production.
The Novel Approach
In stark contrast to the limitations of cation exchange, the novel approach detailed in patent CN106928323B introduces a streamlined workflow centered on weakly polar or non-polar macroporous adsorption resins. This methodology fundamentally shifts the separation mechanism from ionic interaction to physical adsorption based on hydrophobicity and molecular size, which proves far more selective for the A82846B scaffold. A critical innovation in this process is the precise adjustment of the fermentation broth pH to an alkaline range of 10 to 11 prior to solid-liquid separation, followed by a secondary adjustment of the filtrate to pH 9.0 to 9.5 before resin loading. This dual pH control strategy optimizes the ionization state of the impurities and the target molecule, maximizing the adsorption capacity of resins such as LX18, XAD1600, or HP20 while minimizing the co-adsorption of unwanted pigments and proteins.
The implementation of this novel route drastically simplifies the operational workflow, eliminating the need for the cumbersome multi-column setups characteristic of older technologies. By utilizing macroporous resins with specific surface areas and pore sizes, the process achieves high enrichment factors in a single pass, significantly reducing the volume of eluent required. The desorption is efficiently carried out using a dilute acetic acid solution, which gently releases the target intermediate without causing structural degradation. This reduction in solvent consumption directly translates to cost reduction in pharmaceutical intermediate manufacturing, as the expenses associated with solvent procurement, recovery, and waste treatment are markedly lowered. Moreover, the simplified process flow enhances the robustness of the production line, making it more amenable to automation and continuous processing, which are key requirements for modern GMP facilities aiming for high throughput and consistency.
Mechanistic Insights into Macroporous Adsorption and Crystallization
To fully appreciate the technical superiority of this method, one must delve into the physicochemical interactions governing the purification cycle. The success of the macroporous resin step hinges on the delicate balance of hydrophobic interactions and hydrogen bonding between the resin matrix and the glycopeptide. At the optimized pH of 9.0 to 9.5, the A82846B molecule exists in a state that favors adsorption onto the non-polar surface of resins like HP20 or LX18, while many polar impurities remain in the aqueous phase. This selectivity is crucial for removing the closely related analogs A82846A and A82846C, which differ only by subtle substituent changes that affect their overall polarity. The subsequent washing step with purified water at a controlled pH further refines the profile by stripping away weakly bound contaminants without dislodging the target product. This mechanistic precision ensures that the desorption mixture obtained is already of high purity, reducing the load on the subsequent chromatographic steps and extending the lifespan of the more expensive reversed-phase media.
The final stage of the process involves a sophisticated crystallization protocol that serves as the ultimate polishing step for impurity control. After reversed-phase chromatography and concentration via nanofiltration, the addition of sodium chloride at a concentration of 20 to 30 percent (m/v) induces a salting-out effect that drastically reduces the solubility of A82846B in the aqueous medium. This is followed by low-temperature crystallization at 2 to 8 degrees Celsius, which promotes the formation of well-defined crystals while keeping residual impurities in the mother liquor. The inclusion of an ethanol washing step further enhances the purity by removing residual salts and organic solvents trapped within the crystal lattice. This rigorous control over the solid-state form ensures that the final dry powder meets the stringent purity specifications required for the subsequent reductive alkylation steps in Oritavancin synthesis. For R&D teams, this level of control over the impurity profile is essential for ensuring the safety and efficacy of the final drug product.
How to Synthesize A82846B Efficiently
Implementing this synthesis route requires a disciplined approach to process parameters to ensure reproducibility and high yield. The protocol begins with the fermentation broth, which must be carefully conditioned to the specified alkaline pH range before any separation occurs. Following solid-liquid separation, the filtrate is passed through the macroporous resin column at a controlled flow rate to ensure equilibrium adsorption. The detailed standardized synthesis steps involve specific elution gradients and concentration thresholds that are critical for maximizing recovery. For a comprehensive breakdown of the operational parameters, including flow rates, resin bed volumes, and specific solvent ratios, please refer to the technical guide below which outlines the exact procedure for industrial execution.
- Adjust fermentation broth pH to 10-11 for solid-liquid separation, then adjust filtrate pH to 9.0-9.5 for macroporous resin enrichment.
- Purify resin with water, desorb with 0.5-1.5% acetic acid, and concentrate the desorption mixture via nanofiltration.
- Perform reversed-phase chromatography, concentrate, add 20-30% NaCl, and crystallize at 2-8°C to obtain high-purity A82846B.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this purification technology offers profound advantages for procurement managers and supply chain leaders tasked with optimizing production costs and ensuring continuity. The primary benefit lies in the significant simplification of the manufacturing process, which directly correlates to reduced operational expenditure. By eliminating the need for multiple cation exchange columns and reducing the number of solvent-intensive HPLC steps, the facility can achieve higher throughput with the same infrastructure. This efficiency gain allows for a more competitive pricing structure for the high-purity oritavancin intermediate, making it an attractive option for generic drug manufacturers looking to reduce their cost of goods sold. Furthermore, the reduced reliance on hazardous organic solvents mitigates regulatory risks and lowers the costs associated with environmental compliance and waste disposal.
- Cost Reduction in Manufacturing: The transition to macroporous adsorption resin eliminates the need for expensive cation exchange resins that suffer from low capacity and frequent regeneration requirements. This switch reduces the consumption of eluents and acids, leading to substantial cost savings in raw material procurement. Additionally, the higher yield achieved through reduced product leakage means that less fermentation broth is required to produce the same amount of final API intermediate, effectively lowering the cost per kilogram of the active ingredient. The streamlined process also reduces labor hours and energy consumption associated with running complex multi-step purification trains.
- Enhanced Supply Chain Reliability: The robustness of the macroporous resin method ensures a more consistent supply of the key intermediate, minimizing the risk of batch failures that can disrupt downstream API production. The use of widely available and stable resin materials reduces the dependency on specialized supply chains for chromatography media. Moreover, the simplified process is easier to scale up from pilot plant to commercial production, ensuring that lead times for high-purity antibiotic intermediates can be met even during periods of high demand. This reliability is crucial for pharmaceutical companies managing tight launch schedules and regulatory filing deadlines.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with parameters that are easily transferable to large-scale columns and filtration units. The significant reduction in organic solvent usage aligns with global sustainability goals and stricter environmental regulations, reducing the carbon footprint of the manufacturing process. This environmental advantage not only lowers waste treatment costs but also enhances the corporate social responsibility profile of the supply chain. The ability to handle large volumes of fermentation broth efficiently makes this method ideal for meeting the growing global demand for glycopeptide antibiotics without compromising on ecological standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of A82846B using this novel method. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on the feasibility and advantages of the technology. Understanding these details is essential for stakeholders evaluating the technical transfer potential and the long-term viability of this supply route. The responses cover critical aspects ranging from resin selection to impurity management, ensuring that all technical risks are adequately addressed before commercial engagement.
Q: Why is macroporous resin preferred over cation exchange resin for A82846B?
A: Cation exchange resins suffer from low adsorption efficiency and significant product leakage, leading to reduced yields. Macroporous adsorption resins, when used with specific pH adjustments, overcome these leakage issues and improve total recovery rates significantly.
Q: How does the new method reduce environmental impact?
A: The novel process simplifies operation steps and drastically reduces the consumption of organic solvents compared to traditional multi-step resin separation methods. This reduction in solvent usage directly lowers waste liquid generation and treatment costs.
Q: What is the purity level achievable with this crystallization method?
A: By utilizing sodium chloride salting-out crystallization at low temperatures (2-8°C) followed by ethanol washing, the method achieves high purity levels suitable for subsequent glycopeptide synthesis, effectively removing pigments and salts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oritavancin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antibiotics. Our technical team has extensively analyzed the purification pathways described in patent CN106928323B and possesses the expertise to implement these advanced macroporous resin techniques at a commercial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of A82846B meets the exacting standards required for global pharmaceutical registration. We are committed to being a partner that not only supplies materials but also provides technical solutions to complex synthesis challenges.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency. By collaborating with us, you gain access to a reliable source of complex glycopeptide intermediates that combines technical excellence with commercial reliability. Contact us today to discuss how we can support your Oritavancin development goals with superior quality and consistent supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
