Advanced Synthesis of Omeprazole Intermediate for Commercial Scale Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical drug intermediates, and patent CN114044752A presents a significant breakthrough in the manufacturing of omeprazole precursors. This specific technology details a novel production method for 2-chloromethyl-3,5-dimethyl-4-methoxypyridine hydrochloride, a pivotal building block in the synthesis of the widely prescribed proton pump inhibitor, omeprazole. The innovation addresses long-standing challenges in heterocyclic chemistry by optimizing reaction conditions to enhance safety and efficiency. By leveraging a five-step sequence starting from readily available 2,3,5-trimethylpyridine, the process achieves exceptional conversion rates while minimizing hazardous waste. For R&D directors and procurement specialists, understanding this pathway is essential for securing a stable supply of high-quality gastrointestinal drug intermediates. The method not only improves the impurity profile but also aligns with modern green chemistry principles, making it a superior choice for sustainable pharmaceutical manufacturing operations globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyridine-based intermediates for omeprazole has been plagued by significant technical and safety hurdles that hinder efficient commercial production. Traditional routes often rely on extremely hazardous reducing agents such as lithium aluminum hydride (LiAlH4) and sodium hydride (NaH), which pose severe risks of fire and explosion during large-scale handling. Furthermore, conventional methods frequently utilize phosphorus trichloride (PCl3), a highly corrosive reagent that demands specialized, expensive equipment resistant to extreme chemical attack. These legacy processes often suffer from low conversion rates and generate complex byproduct mixtures, complicating downstream purification and reducing the overall yield of the final active pharmaceutical ingredient. The reliance on difficult-to-obtain starting materials further exacerbates supply chain vulnerabilities, leading to inconsistent availability and inflated costs for manufacturers. Consequently, the industry has urgently required a safer, more economically viable alternative to overcome these entrenched processing bottlenecks.
The Novel Approach
The innovative methodology disclosed in the patent data fundamentally reengineers the synthetic pathway to eliminate these critical vulnerabilities while enhancing overall process performance. By selecting 2,3,5-trimethylpyridine as the primary raw material, the new route ensures a stable and cost-effective supply chain foundation, as this feedstock is commercially abundant and easy to source. The process replaces dangerous reducing agents with a controlled oxidation system using hydrogen peroxide and acetic acid, drastically improving operational safety for plant personnel and reducing the need for specialized containment infrastructure. This strategic shift allows for milder reaction conditions that preserve equipment integrity and lower capital expenditure requirements for manufacturing facilities. Moreover, the optimized sequence minimizes byproduct formation through precise control of reaction parameters, resulting in a cleaner crude product that requires less intensive purification. This approach represents a paradigm shift towards safer, more sustainable, and economically superior pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pyridine N-Oxide Chemistry and Substitution
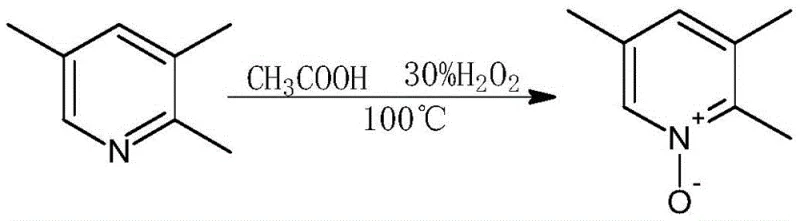
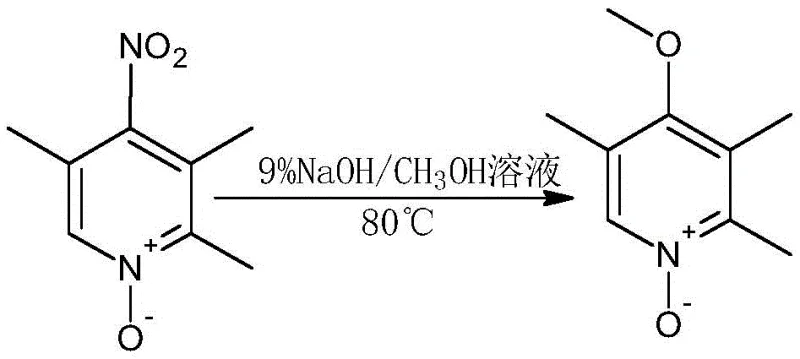
The core of this synthetic advancement lies in the strategic manipulation of the pyridine ring electronic properties through N-oxide formation and subsequent nucleophilic aromatic substitution. The initial oxidation step converts 2,3,5-trimethylpyridine into its N-oxide derivative using 30% hydrogen peroxide in a weak acid environment provided by acetic acid at 100°C. This transformation activates the pyridine ring towards electrophilic attack at the 4-position, facilitating the subsequent nitration step with mixed acid to introduce the nitro group with high regioselectivity. The mechanism leverages the electron-withdrawing nature of the N-oxide to direct substitution precisely, avoiding the formation of unwanted isomers that typically plague direct pyridine functionalization. Following nitration, the nitro group is displaced by a methoxy group via nucleophilic substitution using a sodium hydroxide and methanol solution. This alkoxylation step is critical for establishing the 4-methoxy motif essential for omeprazole activity, proceeding efficiently at 80°C to yield the methoxypyridine-N-oxide intermediate with minimal side reactions.
Impurity control is meticulously managed throughout the reaction sequence through careful monitoring of stoichiometry and temperature profiles to prevent over-reaction or degradation. The acetylation and hydrolysis steps utilize acetic anhydride and hydrochloric acid to convert the N-oxide into the hydroxymethyl derivative, a transformation that proceeds via a Boekelheide-type rearrangement mechanism. This one-pot operation reduces the number of isolation steps, thereby minimizing material loss and exposure to potential contaminants during transfers. The final halogenation employs thionyl chloride under controlled cooling to convert the hydroxymethyl group into the target chloromethyl functionality without affecting the sensitive methoxy or pyridine nitrogen centers. By avoiding high-activity catalysts and maintaining strict pH control during workup, the process ensures that metal residues and acidic byproducts are kept to negligible levels. This rigorous attention to mechanistic detail guarantees a final product with a purity profile that meets the stringent specifications required for downstream API synthesis.
How to Synthesize 2-chloromethyl-3,5-dimethyl-4-methoxypyridine hydrochloride Efficiently
Implementing this synthesis route requires precise adherence to the defined operational parameters to maximize yield and ensure reproducibility across different production batches. The process begins with the oxidation of the collidine derivative, followed by sequential nitration, alkoxylation, and rearrangement steps that must be carefully monitored for completion. Each stage involves specific solvent recovery protocols, such as the recycling of acetic acid and methanol, which are integral to the economic viability of the method. Detailed standard operating procedures dictate the addition rates of reagents like mixed acid and thionyl chloride to manage exotherms and maintain safety margins. For technical teams looking to adopt this technology, understanding the critical process parameters regarding temperature and pH adjustment is vital for successful technology transfer. The standardized synthesis steps outlined in the technical documentation provide a clear roadmap for scaling this chemistry from laboratory verification to full commercial manufacturing.
- Oxidize 2,3,5-trimethylpyridine using hydrogen peroxide and acetic acid to form the N-oxide derivative with high conversion rates.
- Perform catalytic nitration using mixed acid followed by nucleophilic substitution with methanol to introduce the methoxy group at the 4-position.
- Execute acetylation and hydrolysis followed by halogenation with thionyl chloride to finalize the chloromethyl hydrochloride structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented production method offers substantial advantages that directly address the primary concerns of procurement managers and supply chain directors regarding cost and reliability. The elimination of expensive and hazardous reagents significantly lowers the raw material cost base, while the ability to recover and recycle solvents like acetic acid further reduces operational expenditures. The use of readily available starting materials mitigates the risk of supply disruptions that often occur with specialized or scarce chemical feedstocks, ensuring a more resilient supply chain for pharmaceutical manufacturers. Additionally, the milder reaction conditions reduce the wear and tear on production equipment, leading to lower maintenance costs and extended asset life cycles for manufacturing plants. These factors combine to create a highly competitive cost structure that allows for better pricing stability in the volatile pharmaceutical intermediate market. The process inherently supports a more sustainable operation, which is increasingly becoming a key criterion for supplier selection by major multinational corporations.
- Cost Reduction in Manufacturing: The replacement of high-cost, dangerous reagents with common industrial chemicals like hydrogen peroxide and acetic acid drastically reduces the direct material costs associated with production. By avoiding the need for specialized corrosion-resistant equipment required by traditional methods, capital investment and maintenance expenses are significantly lowered for the manufacturing facility. The high conversion rates achieved in each step minimize waste generation, leading to reduced disposal costs and higher overall material efficiency. Furthermore, the solvent recovery systems integrated into the process allow for the reuse of valuable chemicals, compounding the savings over large production volumes. This economic efficiency translates into a more competitive pricing model for the final intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing 2,3,5-trimethylpyridine is far more straightforward than obtaining the specialized starting materials required by legacy synthesis routes, ensuring a consistent flow of raw materials. The robustness of the chemical process means that production schedules are less likely to be disrupted by technical failures or safety incidents, guaranteeing on-time delivery for downstream clients. The scalability of the method allows suppliers to rapidly ramp up production capacity in response to market demand fluctuations without requiring extensive process requalification. This reliability is crucial for pharmaceutical companies that need to maintain continuous API production to meet global medication needs. A stable supply of this key intermediate prevents bottlenecks in the broader drug manufacturing value chain.
- Scalability and Environmental Compliance: The process operates under atmospheric pressure and moderate temperatures, making it inherently easier to scale from pilot plants to multi-ton industrial reactors without complex engineering modifications. The reduction in hazardous waste and the avoidance of heavy metal catalysts simplify environmental compliance and waste treatment procedures, lowering the regulatory burden on the manufacturer. This alignment with green chemistry principles enhances the corporate sustainability profile of the supply chain, meeting the increasing environmental, social, and governance (ESG) requirements of modern businesses. The simplified workup procedures reduce the volume of aqueous waste generated, further minimizing the environmental footprint of the manufacturing operation. These attributes make the technology future-proof against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific omeprazole intermediate synthesis technology. These answers are derived directly from the patent specifications and technical data to provide accurate guidance for decision-makers. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing supply networks. The information covers safety profiles, yield expectations, and scalability factors that are critical for risk assessment. Clients are encouraged to review these points when considering supplier partnerships for long-term intermediate procurement strategies.
Q: What are the safety advantages of this new synthesis route compared to traditional methods?
A: This method avoids high-risk reagents like LiAlH4 and NaH, utilizing safer oxidants such as hydrogen peroxide and acetic acid, significantly reducing process hazards and equipment corrosion risks.
Q: How does this process impact the overall yield and purity of the omeprazole intermediate?
A: The optimized pathway achieves a total yield of approximately 76.3% with individual step yields exceeding 95% in early stages, ensuring high purity specifications suitable for stringent API manufacturing requirements.
Q: Is this synthesis method scalable for large-volume commercial production?
A: Yes, the process utilizes readily available raw materials like 2,3,5-trimethylpyridine and operates under mild conditions without high-pressure requirements, facilitating easy scale-up from pilot to industrial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-chloromethyl-3,5-dimethyl-4-methoxypyridine hydrochloride Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is fully equipped to implement this advanced synthesis route, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand the critical nature of omeprazole intermediates in the global pharmaceutical market and are committed to delivering consistent quality that supports your regulatory filings. Our facility is designed to handle complex heterocyclic chemistry safely and efficiently, leveraging the cost and safety benefits of this patented method. By partnering with us, you gain access to a supply chain that is both economically optimized and technically robust, ready to support your long-term production goals.
We invite you to initiate a dialogue with our technical procurement team to discuss how this synthesis method can optimize your specific supply chain requirements. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this superior production route for your operations. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your volume needs. Let us help you secure a reliable source of high-purity intermediates that drives value and stability for your pharmaceutical products. Contact us today to schedule a technical consultation and explore the possibilities of this advanced manufacturing technology.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
