Advanced One-Pot Synthesis of Telmisartan Intermediates for Commercial Scale-Up
Advanced One-Pot Synthesis of Telmisartan Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally friendly pathways for the production of critical Active Pharmaceutical Ingredient (API) intermediates. A recent technological breakthrough documented in patent CN114436833A introduces a highly efficient preparation method for 4'-methylbiphenyl-2-carboxylate derivatives, which serve as pivotal building blocks in the synthesis of Telmisartan, a widely prescribed angiotensin II receptor antagonist. This novel approach fundamentally restructures the traditional synthetic landscape by integrating a one-pot hydrolysis-esterification strategy followed by a controlled catalytic bromination. For R&D directors and process chemists, this represents a significant leap forward in impurity control and operational simplicity, effectively addressing the longstanding challenges of low yields and difficult purification associated with legacy manufacturing routes. The technology promises to deliver high-purity intermediates with a total molar yield exceeding 90 percent, setting a new benchmark for quality and efficiency in cardiovascular drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key Telmisartan intermediates has been plagued by complex multi-step procedures that introduce significant inefficiencies and environmental burdens. Traditional methods, such as those cited in prior art patents, often rely on starting materials like 4'-bromomethyl-2-cyanobiphenyl, necessitating a cumbersome sequence of debromination and re-bromination that inflates production costs and waste generation. Furthermore, existing protocols frequently utilize low-boiling solvents like dichloromethane for the critical bromination step, typically employing N-bromosuccinimide (NBS) under harsh light irradiation to initiate free radical reactions. These conditions are notoriously difficult to control on a large scale, often leading to the formation of stubborn dibromo by-products that possess polarity profiles nearly identical to the target molecule. Consequently, manufacturers face substantial refining losses, often exceeding 10 percent, and struggle to achieve target purities above 98.5 percent, resulting in elevated operational expenditures and compromised supply chain reliability for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy constraints, the methodology outlined in CN114436833A offers a streamlined, two-step pathway that drastically simplifies the manufacturing workflow while enhancing product quality. The innovation begins with a one-pot hydrolysis and esterification of 2-cyano-4'-methylbiphenyl using high-boiling alcohol compounds under acidic conditions, which eliminates the need for intermediate isolation and reduces solvent consumption. The subsequent bromination step replaces hazardous light-dependent initiation with a sophisticated catalytic system utilizing hydrogen peroxide and a bromine source in the presence of a phase transfer catalyst. This shift not only improves safety by removing the need for intense UV irradiation but also utilizes higher-boiling, less polar organic solvents that facilitate better temperature control and reaction homogeneity. By optimizing these reaction parameters, the new process effectively suppresses the formation of dibromo impurities, allowing for the direct isolation of the target product with purity levels surpassing 99.3 percent without the need for extensive recrystallization, thereby offering a compelling solution for cost reduction in API manufacturing.
Mechanistic Insights into Catalytic Bromination and Impurity Control
The core chemical innovation of this process lies in the precise modulation of the free radical bromination mechanism through the strategic use of phase transfer catalysts and oxidant systems. Unlike traditional methods that rely on photolytic cleavage of NBS, this approach employs hydrogen peroxide in conjunction with a bromine source (such as hydrobromic acid or elemental bromine) and a catalyst like tetrabutylammonium bromide (TBAB) or azobisisobutyronitrile (AIBN). The phase transfer catalyst plays a critical role in shuttling reactive species between phases, ensuring a more uniform distribution of the brominating agent and preventing localized high concentrations that typically drive over-bromination. This controlled environment allows the reaction to proceed selectively at the benzylic position, minimizing the kinetic probability of a second bromination event which would lead to the undesired 4',4'-dibromo methyl biphenyl-2-carboxylate by-product. The use of solvents with higher boiling points, such as n-hexane or ethyl acetate, further stabilizes the reaction thermal profile, ensuring that the radical chain propagation is sustained efficiently without the volatility issues associated with dichloromethane.
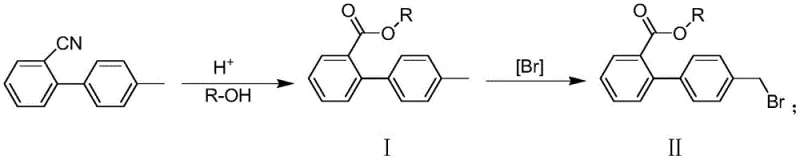
Furthermore, the impurity profile is meticulously managed through the specific stoichiometry of the oxidant and bromine source, which is tuned to match the molar requirements of the substrate precisely. The patent data indicates that by maintaining the molar ratio of hydrogen peroxide to the ester substrate within a specific range, the generation of oxidative side products is minimized while ensuring complete conversion of the starting material. The residual level of the precursor 4'-methylbiphenyl-2-carboxylate is tightly controlled to below 0.1 percent, and critically, the dibromo impurity is also suppressed to below 0.1 percent. This level of specificity is achieved because the catalytic cycle favors the abstraction of the benzylic hydrogen in a controlled manner, avoiding the chaotic radical generation seen in photochemical processes. For quality assurance teams, this mechanistic robustness translates directly into a cleaner crude product, reducing the burden on downstream purification units and ensuring that the final intermediate meets stringent pharmacopeial standards for heavy metals and organic volatiles, which is essential for reliable pharmaceutical intermediate supplier status.
How to Synthesize 4'-Bromomethyl-2-biphenylcarboxylate Efficiently
The practical implementation of this synthesis route involves a carefully orchestrated sequence of thermal and chemical operations designed to maximize throughput and safety. The process initiates with the charging of 2-cyano-4'-methylbiphenyl and a high-boiling alcohol, such as n-butanol or isobutanol, into a reactor equipped for heating and pressure control. Upon the addition of a mineral acid catalyst like sulfuric acid, the mixture is heated to temperatures between 100°C and 150°C to effect simultaneous hydrolysis of the nitrile and esterification of the resulting acid. Following the completion of this one-pot transformation, the reaction mass is concentrated to remove excess alcohol, and the crude ester is subjected to the bromination protocol in a separate or same vessel using a non-polar solvent system. The detailed standardized synthesis steps, including specific reagent grades, addition rates, and quenching procedures required for GMP compliance, are outlined in the guide below.
- Perform one-pot hydrolysis and esterification of 2-cyano-4'-methylbiphenyl using high-boiling alcohols and acid at 100-150°C.
- Conduct bromination of the resulting ester using hydrogen peroxide and a bromine source in a non-polar solvent with a phase transfer catalyst.
- Quench the reaction with sodium bisulfite, filter, and dry to obtain the high-purity bromo-ester intermediate without recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits that extend far beyond simple yield improvements, fundamentally altering the cost structure and risk profile of the supply chain. The elimination of low-boiling, hazardous solvents like dichloromethane in favor of higher-boiling alternatives such as n-hexane or ethyl acetate significantly simplifies solvent recovery and recycling operations, leading to substantial cost savings in raw material procurement and waste disposal. Moreover, the removal of the light-irradiation requirement eliminates the need for specialized photochemical reactors and the associated energy costs, allowing the reaction to be performed in standard stainless steel vessels that are ubiquitous in modern multipurpose chemical plants. This compatibility with existing infrastructure drastically reduces the capital expenditure required for technology transfer and scale-up, enabling faster time-to-market for generic Telmisartan formulations. Additionally, the high selectivity of the reaction minimizes the generation of difficult-to-remove impurities, which reduces the consumption of purification media and lowers the overall environmental footprint of the manufacturing process, aligning with increasingly strict global regulatory standards for green chemistry.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the consolidation of hydrolysis and esterification into a single unit operation, which inherently reduces labor hours, utility consumption, and equipment occupancy time. By avoiding the multi-step protection and deprotection strategies or the debromination-rebromination loops found in older patents, the material throughput is maximized, and the loss of valuable intermediates during isolation is virtually eliminated. The high total molar yield of over 90 percent means that less starting material is required to produce the same amount of finished goods, directly lowering the cost of goods sold (COGS). Furthermore, the ability to produce the intermediate with greater than 99.3 percent purity without recrystallization saves significant amounts of solvent and energy that would otherwise be spent on polishing steps, creating a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on commodity chemicals such as hydrogen peroxide, common mineral acids, and standard aliphatic alcohols ensures a stable and resilient supply base that is not subject to the volatility of specialized reagent markets. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry better than sensitive photochemical processes, ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. This consistency is crucial for maintaining continuous supply to downstream API manufacturers, preventing stockouts that could disrupt the production of life-saving cardiovascular medications. The simplified workflow also shortens the overall production cycle time, allowing for more frequent batch turns and improved responsiveness to fluctuating market demand for Telmisartan intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, utilizing reaction conditions that are easily managed in large-scale reactors without the heat transfer limitations often encountered in exothermic brominations. The substitution of chlorinated solvents with hydrocarbon or ester-based solvents significantly reduces the toxicity of the waste stream, simplifying wastewater treatment and lowering the costs associated with environmental compliance and hazardous waste disposal. The reduction in dibromo by-products not only improves yield but also decreases the load on effluent treatment plants, as fewer halogenated organic compounds need to be incinerated or treated. This alignment with green chemistry principles enhances the corporate social responsibility profile of the manufacturer and future-proofs the supply chain against tightening environmental regulations regarding volatile organic compound (VOC) emissions and halogenated waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the practical advantages over incumbent technologies. Understanding these nuances is essential for making informed decisions about process validation and vendor qualification.
Q: How does the new method control dibromo by-products?
A: By using a specific phase transfer catalyst and controlling the reaction temperature in a high-boiling non-polar solvent, the process selectively promotes mono-bromination, keeping dibromo impurities below 0.1%.
Q: What are the advantages of the one-pot esterification method?
A: The one-pot method combines hydrolysis and esterification, eliminating intermediate isolation steps, reducing solvent usage, and significantly lowering three-waste generation compared to traditional multi-step routes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process avoids hazardous conditions like strong light irradiation and low-boiling solvents (DCM), uses readily available raw materials, and achieves over 90% total molar yield, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4'-Methylbiphenyl-2-carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior manufacturing technologies requires a partner with both technical expertise and the capacity to execute at scale. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN114436833A can be seamlessly translated into reliable industrial output. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of this bromination process, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 4'-methylbiphenyl-2-carboxylate meets the highest international standards. We are committed to supporting your R&D and commercial teams by providing a stable supply of high-quality intermediates that enable the efficient production of Telmisartan.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this greener, higher-yielding process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring a smooth and successful partnership in the competitive cardiovascular therapeutics market.
