Advanced Cobamamide Extraction Technology for Commercial Scale Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to enhance the purity and yield of critical vitamin derivatives, particularly complex molecules like Cobamamide, also known as Vitamin B12 coenzyme. A pivotal advancement in this domain is detailed in the Chinese patent CN101948494B, which outlines a sophisticated method for extracting Cobamamide from fermentation broths with unprecedented efficiency. This technology addresses the longstanding bottlenecks of traditional extraction processes by integrating macroporous resin adsorption, chemical flocculation, and dynamic crystallization techniques. For R&D directors and procurement specialists, understanding the nuances of this patent is essential, as it represents a shift from labor-intensive, low-yield protocols to a streamlined, high-throughput manufacturing paradigm. The method achieves a product purity exceeding 98.0% with impurity levels maintained below 1.0%, setting a new benchmark for quality in the supply chain of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial extraction of Vitamin B12 coenzyme has been plagued by significant inefficiencies inherent to reliance on multi-step alumina column chromatography without adequate pre-purification. Traditional workflows often suffer from poor separation capabilities when dealing with the complex matrix of fermentation broths, leading to low batch processing capacities and sluggish production rates. A critical failure point in conventional methods is the crystallization stage; due to the presence of insoluble materials and low purity in the crystallization stock solution, manufacturers are forced to employ extremely slow, static crystallization processes to avoid turbidity. This necessity restricts stirring speeds and requires staged addition of acetone over intervals exceeding 12 hours, extending the total crystallization time to approximately 72 hours. Furthermore, the heavy reliance on alumina creates substantial environmental burdens due to difficult regeneration processes and high solid waste generation, driving up both operational costs and environmental compliance expenditures for manufacturers.
The Novel Approach
The innovative methodology disclosed in the patent fundamentally restructures the purification train by introducing a strategic flocculation step and replacing excessive alumina usage with selective chromatographic resins. By incorporating divalent zinc or trivalent aluminum salts as flocculating agents prior to final chromatography, the process effectively precipitates macromolecular impurities such as proteins and pigments that typically interfere with downstream separation. This pre-treatment allows for the utilization of styrenic modified chromatographic resins which possess superior selectivity for separating different types of Vitamin B12 analogues compared to standard alumina. Consequently, the resulting crystallization stock solution is of such high purity that it permits rapid dynamic crystallization under vigorous stirring conditions. This transformation reduces the crystallization cycle dramatically to between 4 and 10 hours, while simultaneously increasing the batch processing capacity by more than threefold, thereby offering a scalable solution for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Resin Adsorption and Flocculation Purification
The core technical breakthrough of this extraction method lies in the synergistic application of macroporous resin adsorption and chemical flocculation to manage the complex molecular architecture of Cobamamide. As illustrated in the structural complexity of the target molecule, Cobamamide contains multiple functional groups including amide side chains, a corrin ring system, and a nucleotide loop, making it susceptible to degradation and difficult to separate from structurally similar impurities.
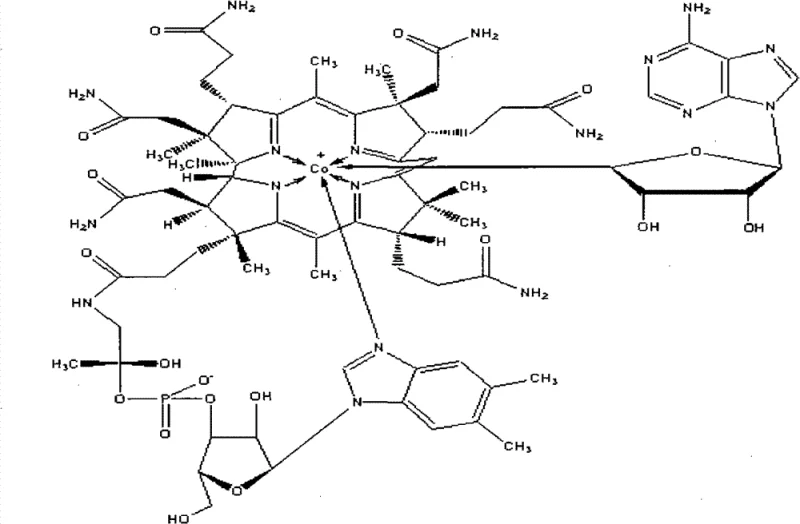
The mechanism begins with the hydrolysis of the fermentation broth under light-resistant conditions to preserve the photosensitive corrin ring, followed by adsorption onto macroporous resin which captures the target molecule while allowing highly polar waste to pass through. The subsequent addition of flocculating agents operates on the principle of charge neutralization and bridging, where metal cations interact with negatively charged protein residues and pigment molecules to form large aggregates that can be easily filtered out. This step is crucial for protecting the downstream chromatographic resin from fouling and ensuring that the final alumina polish is only tasked with removing trace related substances rather than bulk impurities. The final dynamic crystallization relies on the supersaturation of this highly purified solution, where the absence of particulate contaminants allows for uniform crystal growth even under mechanical agitation, preventing the occlusion of mother liquor and ensuring high chemical purity.
How to Synthesize Cobamamide Efficiently
The synthesis and extraction of Cobamamide via this patented route require precise control over pH, solvent ratios, and stirring dynamics to maximize yield and purity. The process moves away from static batch operations towards a more continuous and controlled flow, leveraging the high capacity of modern resin technologies. For technical teams looking to implement this, the following standardized synthesis steps outline the critical operational parameters derived from the patent examples.
- Hydrolyze Vitamin B12 fermentation solution under light-resistant conditions and filter to obtain the initial filtrate.
- Adsorb the filtrate onto a macroporous resin column, wash, and desorb to obtain the primary desorption solution.
- Concentrate the solution, add a flocculating agent (zinc or aluminum salt) to remove impurities, and filter to obtain a purified solution.
- Pass the purified solution through a chromatographic resin column to separate other Vitamin B12 types, followed by an alumina column for final polishing.
- Add acetone to the final stock solution under stirring to induce dynamic crystallization, then filter and dry to obtain the finished Cobamamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this advanced extraction technology translates directly into enhanced supply security and significant cost optimization opportunities without compromising on quality standards. The shift from a low-yield, alumina-heavy process to a resin-based flocculation system eliminates several costly unit operations and drastically reduces the consumption of consumables that require frequent replacement. By removing the bottleneck of slow static crystallization, manufacturers can achieve a much faster turnover rate for production batches, effectively increasing the available inventory velocity and reducing the risk of supply disruptions during peak demand periods. Furthermore, the reduction in solid waste generation from alumina disposal aligns with increasingly stringent environmental regulations, mitigating the risk of regulatory fines and shutdowns that can jeopardize supply continuity.
- Cost Reduction in Manufacturing: The implementation of flocculation and chromatographic resin significantly lowers the cost of goods sold by improving the overall recovery rate of Vitamin B12 from the fermentation broth. By achieving a total yield of over 80% compared to the traditional 60%, the process maximizes the value extracted from raw fermentation materials, effectively reducing the raw material cost per kilogram of finished product. Additionally, the elimination of tedious alumina regeneration steps reduces labor costs and energy consumption associated with washing and reactivating column media, contributing to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: The ability to process larger batch volumes, with capacity improvements exceeding three times that of conventional methods, provides a robust buffer against market fluctuations and demand spikes. This scalability ensures that suppliers can meet large-volume orders for high-purity pharmaceutical intermediates without the need for proportional increases in facility footprint or equipment investment. The shortened production cycle also means that lead times for order fulfillment can be drastically compressed, offering a competitive advantage in just-in-time manufacturing environments.
- Scalability and Environmental Compliance: The cleaner production profile of this method, characterized by reduced three-waste discharge, simplifies the environmental compliance burden for manufacturing sites. The ease of regenerating chromatographic resins compared to alumina means less hazardous waste is sent to landfills, supporting corporate sustainability goals. This environmental efficiency not only future-proofs the supply chain against tightening ecological regulations but also enhances the brand reputation of the supplier as a responsible manufacturer of specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cobamamide extraction technology, based on the specific advantages outlined in the patent documentation. These insights are designed to clarify the operational feasibility and quality implications for potential partners and technical evaluators.
Q: How does the new flocculation step improve Cobamamide purity compared to traditional methods?
A: The introduction of divalent zinc or trivalent aluminum salts as flocculating agents effectively removes macromolecular impurities such as proteins and pigments that traditional alumina columns struggle to separate. This results in a product with impurity content below 1.0% and active content exceeding 98.0%.
Q: What are the scalability advantages of using chromatographic resin over exclusive alumina columns?
A: Chromatographic resin offers significantly higher treatment capacity and easier regeneration compared to traditional alumina. This technological shift allows for a batch processing capacity increase of over three times, drastically improving manufacturing throughput and supply reliability.
Q: How does the dynamic crystallization process reduce production lead time?
A: By utilizing a high-purity crystallization stock solution achieved through resin and flocculation purification, the process enables rapid stirred crystallization. This reduces the crystallization cycle from approximately 72 hours in traditional methods to just 4-10 hours, significantly accelerating the overall production timeline.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cobamamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced extraction technologies to meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101948494B are fully realized in industrial practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Cobamamide meets the highest international standards for potency and impurity profiles, providing our clients with absolute confidence in their raw material supply.
We invite procurement leaders and technical directors to engage with our technical procurement team to discuss how we can tailor this extraction process to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into how our optimized manufacturing protocols can reduce your overall landed costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our commitment to delivering high-quality Vitamin B12 coenzyme intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
