Advanced Alumina Chromatography for Commercial Scale-Up of High-Purity Acarbose API
The pharmaceutical industry continuously seeks robust purification strategies to meet stringent regulatory standards for Active Pharmaceutical Ingredients (APIs), particularly for complex molecules like Acarbose. Patent CN102603822B introduces a transformative method for improving the purity of Acarbose by specifically targeting the removal of Impurity A, a challenging isomer that closely mimics the retention time of the target molecule in High Performance Liquid Chromatography (HPLC). This innovation leverages aluminum oxide chromatography to achieve a final product purity increase of approximately 2 percentage points, elevating average content from 96.5% to 98.5%, while simultaneously ensuring that Impurity A levels are reduced to below the critical 0.6% threshold mandated by major pharmacopoeias such as the European Pharmacopoeia. For R&D directors and process engineers, this patent represents a significant leap forward in downstream processing, offering a pathway to produce high-purity bulk drugs with enhanced safety profiles for patients suffering from type II diabetes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Acarbose has been plagued by the difficulty of separating Impurity A during the extraction and refining stages. Traditional methodologies often rely on macroporous adsorption resins or complex temperature control strategies during the pretreatment phase. For instance, prior art described in Chinese patent CN101008025A necessitates controlling the fermentation broth temperature between 0°C and 25°C, a process that consumes vast amounts of cooling water and energy, thereby inflating operational expenditures. Furthermore, alternative approaches utilizing membrane filtration and macroporous resins, as seen in application CN031021331.6, often suffer from suboptimal recovery rates. When the initial concentration of Impurity A in the feed liquid is elevated, these conventional resin-based systems frequently fail to effectively remove the contaminant, resulting in yields that hover around a mere 60%. Additionally, the regeneration of these traditional columns often involves hazardous organic solvents, introducing significant safety hazards and environmental compliance burdens that modern green chemistry initiatives strive to eliminate.
The Novel Approach
In stark contrast to these legacy systems, the novel approach detailed in CN102603822B utilizes a specialized aluminum oxide chromatographic column that fundamentally alters the separation dynamics. This method capitalizes on the unique adsorption properties of aluminum oxide, which selectively binds Impurity A while allowing the Acarbose molecule to pass through with minimal retention. The process eliminates the need for energy-intensive cooling cycles and avoids the use of organic solvents during the column regeneration phase, relying instead on a safe and cost-effective acid-base wash cycle. By optimizing the column aspect ratio to between 3:1 and 5:1 and precisely controlling flow velocities based on the initial impurity load, manufacturers can consistently achieve yields exceeding 88.95%. This shift not only simplifies the technological operation by removing the requirement for high-temperature activation of the stationary phase but also drastically improves the economic viability of producing high-purity Acarbose on a commercial scale.
Mechanistic Insights into Alumina Chromatographic Separation
The core challenge in Acarbose purification lies in the structural similarity between the target API and its primary contaminant, Impurity A. As illustrated in the structural comparison below, Impurity A is an isomer of Acarbose, possessing a nearly identical molecular framework that results in very similar physicochemical properties. This structural homology makes separation via standard HPLC or simple resin adsorption exceptionally difficult, as the retention times overlap significantly. The aluminum oxide stationary phase functions through a mechanism of differential adsorption affinity. Unlike macroporous resins that may non-specifically bind both species or require extreme conditions to differentiate them, the surface chemistry of the activated aluminum oxide interacts more strongly with the specific functional group orientation of Impurity A. This allows the column to act as a highly selective filter, retaining the isomer while permitting the desired Acarbose to elute efficiently.
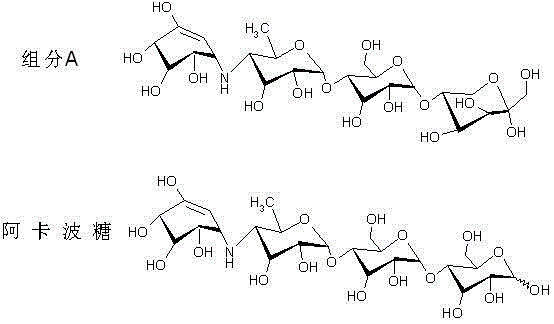
Beyond mere separation efficiency, the mechanism of this process also contributes to superior impurity profiling and color removal. The aluminum oxide treatment effectively adsorbs various pigment components present in the fermentation feed liquid, resulting in a visibly lighter and cleaner eluate. This dual function of impurity removal and decolorization is critical for meeting the aesthetic and safety standards of bulk pharmaceutical ingredients. Furthermore, the regeneration mechanism is designed to restore the column's activity without degrading the stationary phase. By sequentially flushing with deionized water, 1.5M hydrochloric acid, and 1.5M caustic soda, the column removes accumulated organic residues and resets the surface charge properties. The absence of organic solvents in this regeneration cycle ensures that there is no risk of solvent residue contamination in the final API, a critical quality attribute for regulatory approval.
How to Synthesize High-Purity Acarbose Efficiently
The implementation of this purification protocol requires precise control over fluid dynamics and column parameters to maximize yield and purity. The process begins with the preparation of a pre-purified Acarbose liquid, obtained through standard flocculation and filtration of the fermentation broth, which is then concentrated to an appropriate volume relative to the column capacity. The critical operational window involves loading this solution onto the aluminum oxide column and adjusting the outlet valve to maintain a flow velocity that correlates with the initial impurity concentration; specifically, flows of 0.4 to 0.6 BV/h are recommended when Impurity A exceeds 1.0%, while higher flows of 0.6 to 0.8 BV/h are permissible for lower impurity loads. Detailed standardized synthesis steps and regeneration protocols follow below to ensure reproducibility.
- Prepare the pre-purified Acarbose liquid through flocculation, filtration, and initial concentration to obtain a feed solution.
- Load the solution onto an aluminum oxide chromatographic column (aspect ratio 3-5: 1) and control flow velocity between 0.4 to 0.8 BV/h based on initial impurity levels.
- Regenerate the column using sequential washes of deionized water, 1.5M hydrochloric acid, and 1.5M caustic soda, avoiding high-temperature activation or organic solvents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this alumina-based purification technology offers compelling economic and logistical benefits that directly impact the bottom line. The most significant advantage is the dramatic improvement in process yield, which jumps from the historical baseline of approximately 60% to over 88.95%. This increase in mass efficiency means that for every ton of fermentation broth processed, a substantially larger quantity of saleable API is produced, effectively lowering the cost of goods sold (COGS) without requiring additional raw material inputs. Furthermore, the elimination of organic solvents from the regeneration process removes a major cost center associated with solvent purchase, recovery, and disposal, while simultaneously reducing the facility's exposure to volatile organic compound (VOC) regulations and fire safety risks.
- Cost Reduction in Manufacturing: The transition to an acid-base regeneration system eliminates the need for expensive organic solvents and the energy-intensive high-temperature activation steps required by traditional aluminum oxide processes. This simplification of the utility profile leads to substantial cost savings in both energy consumption and waste treatment. By avoiding the use of hazardous solvents, the facility also reduces the capital expenditure required for explosion-proof equipment and solvent recovery units, further enhancing the overall economic efficiency of the manufacturing line.
- Enhanced Supply Chain Reliability: The robustness of the aluminum oxide column against high impurity loads ensures consistent production output even when fermentation quality fluctuates. Unlike resin methods that fail when Impurity A levels are high, this method maintains high recovery rates, preventing production bottlenecks and batch failures. This reliability translates to more predictable lead times for customers and a more stable supply of high-purity Acarbose, mitigating the risk of stockouts in the global pharmaceutical market.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard chromatographic equipment that can be easily expanded from pilot to commercial scales. The aqueous nature of the regeneration waste stream simplifies wastewater treatment compared to solvent-laden effluents, aligning with increasingly strict environmental protection standards. This green chemistry approach not only future-proofs the manufacturing site against tightening regulations but also enhances the brand reputation of the supplier as a sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These answers are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on yield expectations, impurity control, and operational safety. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing production lines.
Q: How effectively does this method reduce Impurity A compared to traditional resin methods?
A: This alumina chromatography method reduces Impurity A content to below 0.6%, meeting strict pharmacopoeia standards, whereas traditional macroporous resin methods often struggle to achieve this when feed liquid impurity levels are high, typically yielding only around 60% recovery.
Q: What are the regeneration requirements for the aluminum oxide column?
A: The regeneration process utilizes only acid (1.5M HCl) and base (1.5M NaOH) solutions followed by water washing. Unlike traditional methods, it does not require high-temperature activation or the use of hazardous organic solvents, significantly simplifying operation and enhancing safety.
Q: What is the expected yield improvement using this purification technique?
A: The process demonstrates a total yield of more than 88.95%, with specific embodiments showing single-step yields ranging from 86.84% to 91.78%. This represents a substantial improvement over the approximate 60% yield associated with older macroporous adsorption technologies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acarbose Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and yield in the production of complex carbohydrates like Acarbose. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory techniques like the alumina chromatography method described in CN102603822B are successfully translated into robust industrial processes. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Acarbose meets the <0.6% Impurity A requirement essential for global regulatory compliance.
We invite pharmaceutical partners to collaborate with us to leverage these advanced purification capabilities for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential yield improvements and operational savings specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the highest standards of quality and efficiency in Acarbose manufacturing.
