Advanced Heterocyclic Bridged Phenyl Ethylene Derivatives for High-Efficiency Deep Blue OLED Manufacturing
The rapid evolution of the organic optoelectronic materials sector demands continuous innovation in emissive layers to achieve superior color purity and device stability. Patent CN108516970B, published in early 2021, introduces a groundbreaking class of heterocyclic bridged phenyl ethylene derivatives that address the critical bottlenecks in deep blue organic light-emitting diode (OLED) technology. These novel compounds are engineered with a heterocyclic bridged tetraphenylethylene core, strategically modified with various electron-donating groups to optimize electronic properties. The significance of this intellectual property lies in its ability to simultaneously harness Aggregation Induced Emission (AIE) characteristics and solid-state deep blue luminescence. For R&D directors and technical procurement specialists, this represents a pivotal shift away from traditional materials that suffer from aggregation-caused quenching (ACQ). By integrating these derivatives into emission layers, manufacturers can achieve devices with narrow half-peak widths and high color purity, specifically targeting the elusive deep blue spectrum required for next-generation full-color displays and solid-state lighting applications without compromising on efficiency or operational lifetime.
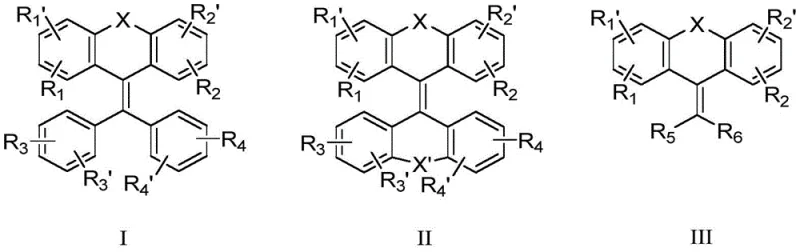
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of efficient deep blue OLEDs has been plagued by significant technical trade-offs that hinder mass adoption in high-end display panels. Traditional fluorescent materials, while offering good device stability, typically exhibit low external quantum efficiency because they can only utilize singlet excitons for light emission, wasting a substantial portion of the electrical energy input. On the other hand, phosphorescent materials and thermally activated delayed fluorescence (TADF) emitters can harvest both singlet and triplet excitons to boost efficiency, but they introduce severe drawbacks such as significant device efficiency roll-off at high brightness levels and poor operational stability. Furthermore, many high-performance blue emitters rely on expensive rare earth metals or complex organometallic complexes, which drastically inflate the bill of materials and complicate the supply chain. The aggregation-caused quenching effect is another pervasive issue where materials lose luminescence efficiency in the solid state, necessitating complex doping strategies that add processing steps and variability to the manufacturing line. These cumulative limitations create a barrier for procurement managers seeking cost-effective, reliable, and high-performance solutions for commercial scale-up of complex electronic chemicals.
The Novel Approach
The technology disclosed in the patent data offers a transformative solution by leveraging the unique photophysical properties of heterocyclic bridged phenyl ethylene derivatives. Unlike conventional fluorophores, these molecules are designed with a twisted structural conformation that prevents tight pi-pi stacking in the aggregated state, thereby effectively suppressing the ACQ effect and enabling strong solid-state emission. The core innovation involves bridging the phenyl ethylene structure with heteroatoms such as sulfur or sulfone groups, which not only stabilizes the molecular framework but also fine-tunes the energy levels for deep blue emission. This approach allows for the fabrication of OLED devices with simple structures that do not necessarily require complex doping host systems to achieve high performance. The result is a material class that delivers high efficiency with minimal efficiency roll-off, ensuring consistent brightness over the device's operational life. For supply chain heads, this simplification translates to reduced lead time for high-purity electronic chemical procurement and a more robust manufacturing process that is less sensitive to minor variations in deposition parameters, ultimately enhancing yield rates and production throughput.
Mechanistic Insights into Suzuki Coupling and AIE Mechanism
The synthesis of these advanced emitters relies on a robust palladium-catalyzed cross-coupling strategy, specifically the Suzuki-Miyaura reaction, which is renowned for its tolerance to functional groups and mild reaction conditions. The mechanistic pathway involves the coupling of a halogen-substituted heterocyclic anthracene derivative, acting as the electrophilic partner, with an electron-donating group-substituted boric acid derivative as the nucleophilic partner. In the presence of a palladium catalyst such as tetrakis(triphenylphosphine)palladium and a base like potassium carbonate, the carbon-halogen bond is activated, facilitating the formation of a new carbon-carbon bond that links the electron-rich donor moieties to the electron-deficient heterocyclic core. This modular synthetic approach allows for precise tuning of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) levels by varying the substituents on the benzene rings. The introduction of strong electron-donating groups such as carbazole or diphenylamine enhances the hole transport capability of the molecule, promoting balanced charge injection in the OLED device. This chemical precision ensures that the resulting materials possess the necessary electronic characteristics to function effectively as bipolar transport materials, further simplifying the device architecture by potentially reducing the need for separate charge transport layers.
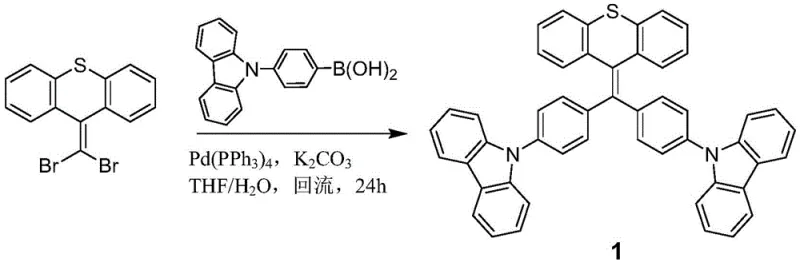
Beyond the synthetic chemistry, the operational mechanism of these materials in a device context is governed by the Aggregation Induced Emission (AIE) phenomenon. In solution, the free rotation of the phenyl rings around the central ethylene bond dissipates excited state energy through non-radiative decay pathways, resulting in weak emission. However, in the solid state or aggregated form within the OLED emission layer, these intramolecular rotations are physically restricted due to steric hindrance and intermolecular interactions. This restriction of intramolecular motion (RIM) blocks the non-radiative decay channels, forcing the excited energy to be released radiatively as photons. This mechanism is critical for achieving high solid-state luminescence quantum yields without the need for dilute doping in a host matrix, although doping can still be employed to further optimize performance. The patent data highlights that derivatives with sulfone bridges exhibit slightly different electronic properties compared to sulfur bridges, offering a tunable platform for optimizing the emission peak to fall strictly within the 450-460nm range. This level of control over the emission spectrum is vital for meeting the stringent color gamut standards of modern display technologies, ensuring that the blue sub-pixels deliver the required saturation and brightness for high-dynamic-range imaging.
How to Synthesize Heterocyclic Bridged Phenyl Ethylene Efficiently
The preparation method outlined in the technical documentation provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and high yield. The process begins with the precise stoichiometric mixing of the halogenated heterocyclic precursor and the boronic acid derivative in a mixed solvent system of tetrahydrofuran and water. This biphasic system is crucial for solubilizing both the organic reactants and the inorganic base, facilitating the catalytic cycle at the interface. The reaction mixture is subjected to rigorous degassing with nitrogen to remove dissolved oxygen, which can oxidize the palladium catalyst and inhibit the coupling reaction. Following the reflux period, the workup procedure involves standard extraction and purification techniques, utilizing silica gel column chromatography to isolate the target product with high purity. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and quality control.
- Prepare the reaction system by dissolving halogen-substituted heterocyclic anthracene derivatives and electron-donating group-substituted boric acid derivatives in a THF and water mixed solvent.
- Add tetrakis(triphenylphosphine)palladium catalyst and potassium carbonate base, then degas the mixture with nitrogen to ensure an oxygen-free environment.
- Heat the reaction mixture under reflux for 24 hours, followed by solvent removal, extraction with dichloromethane, and purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of heterocyclic bridged phenyl ethylene derivatives offers substantial strategic advantages for procurement managers and supply chain directors aiming to optimize the cost structure of OLED manufacturing. The primary value driver is the elimination of expensive noble metal catalysts typically required for phosphorescent emitters, such as iridium or platinum complexes. By utilizing purely organic fluorescent materials that achieve high efficiency through AIE mechanisms, manufacturers can significantly reduce the raw material costs associated with the emissive layer. This cost reduction in electronic chemical manufacturing is not merely theoretical; it is grounded in the fundamental chemistry of using abundant organic feedstocks rather than scarce precious metals. Furthermore, the synthetic route avoids harsh reaction conditions and complex purification steps often associated with organometallic synthesis, leading to lower energy consumption and reduced waste generation. These factors collectively contribute to a more sustainable and economically viable production model that aligns with global trends towards green chemistry and cost efficiency in the display industry.
- Cost Reduction in Manufacturing: The synthetic pathway utilizes readily available starting materials and standard catalytic systems, which drastically simplifies the supply chain logistics. By avoiding the need for custom-synthesized metal complexes, procurement teams can source raw materials from a broader base of chemical suppliers, increasing competition and driving down input costs. The high yields reported in the patent examples indicate a robust process with minimal material loss, further enhancing the overall cost-effectiveness of the production run. This economic efficiency allows for more competitive pricing strategies in the final display panel market, providing a distinct advantage in price-sensitive consumer electronics segments.
- Enhanced Supply Chain Reliability: The reliance on stable organic compounds rather than sensitive metal complexes improves the shelf-life and storage stability of the materials. This reduces the risk of material degradation during transit and storage, ensuring that the quality of the emitter remains consistent from the supplier to the fabrication plant. The simplicity of the synthesis also means that production capacity can be scaled up more rapidly to meet surges in demand without the bottlenecks associated with sourcing limited quantities of precious metal precursors. For supply chain heads, this translates to reduced lead time for high-purity OLED material delivery and a more resilient supply network capable of withstanding market fluctuations.
- Scalability and Environmental Compliance: The process generates fewer hazardous byproducts compared to traditional phosphorescent material synthesis, simplifying waste treatment and disposal procedures. The use of common solvents like dichloromethane and tetrahydrofuran allows for established recycling and recovery protocols, minimizing the environmental footprint of the manufacturing process. This alignment with environmental regulations reduces compliance costs and mitigates the risk of production shutdowns due to regulatory issues. Additionally, the thermal stability of the final products ensures they can withstand the thermal evaporation processes used in OLED fabrication without decomposition, ensuring high throughput and yield in the device manufacturing stage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these novel materials in industrial settings. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines and for assessing the potential return on investment. We encourage technical teams to review these insights to facilitate informed discussions with their engineering and procurement departments.
Q: What are the key advantages of heterocyclic bridged phenyl ethylene derivatives over traditional fluorescent materials?
A: Unlike traditional fluorescent materials that suffer from low external quantum efficiency, these derivatives exhibit Aggregation Induced Emission (AIE) characteristics. This allows for high solid-state luminescence quantum yield, narrow half-peak width, and deep blue emission with minimal efficiency roll-off, addressing the stability and color purity issues common in prior art.
Q: How does the synthetic route impact commercial scalability for OLED manufacturers?
A: The synthesis utilizes a standard Suzuki coupling reaction with commercially available raw materials like dibromothiaanthracene and carbazole boronic acid. The process avoids expensive transition metal phosphorescent complexes, significantly simplifying the supply chain and reducing raw material costs while maintaining high yields suitable for industrial scale-up.
Q: Can these materials be used in both doped and non-doped OLED device structures?
A: Yes, the patent data demonstrates successful application in both undoped and doped device architectures. The materials show stable performance in non-doped structures with high brightness and low starting voltage, while doped configurations further enhance current efficiency and color purity, offering flexibility for diverse display engineering requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Bridged Phenyl Ethylene Supplier
As the demand for high-performance deep blue emitters continues to surge, partnering with an experienced chemical manufacturing partner is essential for securing a competitive edge in the market. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications required for OLED applications. We understand the critical nature of impurity profiles in electronic materials and employ sophisticated purification techniques to minimize trace contaminants that could affect device performance. Our commitment to technical excellence ensures that clients receive materials that are not only chemically pure but also optimized for processability in vacuum deposition systems.
We invite global partners to collaborate with us to unlock the full potential of this patented technology for their display manufacturing needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and device architectures. By engaging with us, you can access specific COA data and route feasibility assessments that will help you validate the material performance in your own pilot lines. We are dedicated to supporting your R&D efforts with rapid sample delivery and technical consultation, ensuring a smooth transition from laboratory concept to mass production. Contact us today to discuss how our heterocyclic bridged phenyl ethylene derivatives can enhance your product portfolio and drive down your overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
