Scalable Synthesis of High-Performance Quinoline Nitrile Derivatives for Optoelectronic Applications
Scalable Synthesis of High-Performance Quinoline Nitrile Derivatives for Optoelectronic Applications
The landscape of organic optoelectronic materials is undergoing a significant transformation, driven by the urgent demand for efficient solid-state emitters that overcome the limitations of traditional fluorescence quenching. Patent CN102702096B introduces a groundbreaking class of quinoline nitrile derivatives that exhibit exceptional aggregation-induced emission (AIE) performance, marking a pivotal shift from solution-phase to solid-state efficiency. These compounds, characterized by a robust quinoline core functionalized with dicyanomethylene groups, demonstrate strong fluorescence in aggregated or solid states, making them ideal candidates for electroluminescent devices and advanced fluorescent probes. The strategic design of these molecules addresses the critical bottleneck of concentration quenching found in conventional pyranonitrile derivatives like DCM, offering a pathway to higher efficiency in OLED manufacturing. By leveraging a streamlined three-step synthesis starting from 2-methylquinoline, this technology promises not only superior photophysical properties but also enhanced manufacturability for industrial applications. 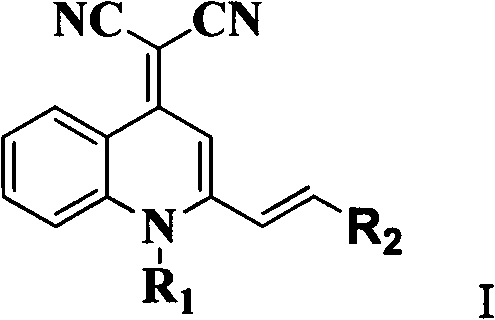
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of red-emitting organic materials has been dominated by pyranonitrile derivatives, such as the well-known DCM dye, which was first reported for electroluminescent devices in the late 1980s. However, a fundamental flaw plagues these traditional materials: they exhibit severe fluorescence quenching when transitioned from dilute solutions to solid films or aggregated states. This phenomenon, known as aggregation-caused quenching (ACQ), drastically reduces the external quantum efficiency of devices, necessitating complex doping strategies to isolate the emitter molecules within a host matrix. Furthermore, the synthetic routes to modify DCM and mitigate this quenching often involve cumbersome multi-step procedures that require stringent reaction conditions and expensive precursors. Even with these modifications, the fluorescence quantum yield in the solid state often remains suboptimal, limiting the brightness and longevity of the resulting optoelectronic devices. The reliance on complex molecular architectures to prevent pi-pi stacking interactions adds significant cost and complexity to the supply chain, creating a barrier for widespread commercial adoption in high-volume display manufacturing.
The Novel Approach
In stark contrast to the ACQ-prone conventional dyes, the quinoline nitrile derivatives disclosed in this patent leverage the phenomenon of aggregation-induced emission to turn a traditional liability into a performance asset. Instead of struggling to prevent molecular aggregation, this novel approach utilizes the restriction of intramolecular motion in the aggregated state to enhance radiative decay, resulting in intense fluorescence precisely where it is needed most: in the solid film. The synthetic strategy is elegantly simple, utilizing a convergent route that builds the functional core through well-established organic transformations such as quaternization and Knoevenagel condensation. This simplicity translates directly into process robustness, as the reactions proceed under relatively mild reflux conditions using common solvents like acetonitrile and ethanol. The ability to tune the emission properties by varying the N-alkyl chain or the aromatic aldehyde substituent provides a versatile platform for material scientists to optimize color purity and energy levels without overhauling the entire production process. This paradigm shift not only solves the quenching problem but also aligns perfectly with the industry's need for cost-effective, scalable solutions for next-generation display and lighting technologies.
Mechanistic Insights into Quinoline Nitrile Synthesis and AIE Mechanism
The chemical architecture of these AIE-active materials is constructed through a precise sequence of reactions that establish the conjugated system necessary for efficient charge transport and light emission. The process begins with the quaternization of 2-methylquinoline, where the nitrogen atom is alkylated to form a quinolinium salt, effectively activating the methyl group for subsequent nucleophilic attack. This intermediate then undergoes a Michael addition-elimination reaction with malononitrile, a step that introduces the electron-withdrawing dicyanomethylene moiety critical for the push-pull electronic structure. The final step involves a Knoevenagel condensation with an aromatic aldehyde, which extends the conjugation length and introduces the specific substituents that dictate the solid-state packing and emission wavelength. 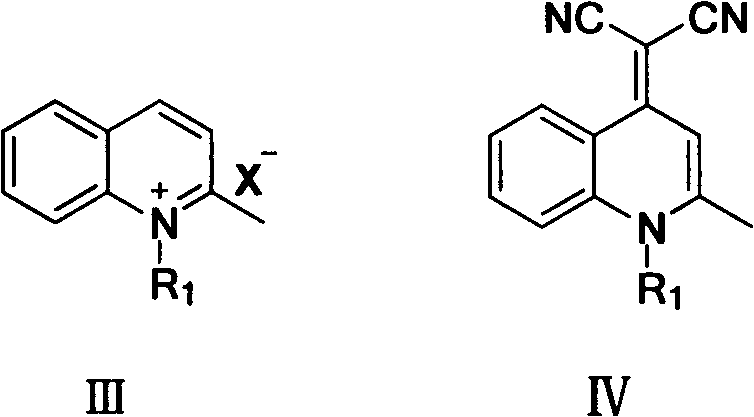
From a purity and impurity profile perspective, this synthetic route offers distinct advantages for R&D teams focused on material consistency. The use of piperidine as a mild base catalyst in the final condensation step minimizes side reactions such as polymerization or over-alkylation, which are common pitfalls in more aggressive catalytic systems. The intermediates, particularly the quinolinium salt and the dicyanomethylene intermediate, are often solids that can be purified via simple filtration or recrystallization before proceeding to the next step, ensuring that impurities do not carry through to the final product. This stepwise purification capability is crucial for achieving the high purity specifications required in OLED manufacturing, where trace metal contaminants or organic byproducts can act as quenching sites or degradation centers. The structural rigidity imparted by the quinoline ring and the dicyano group restricts non-radiative decay pathways in the solid state, which is the fundamental mechanism behind the observed AIE effect, ensuring that the energy absorbed is efficiently converted into light rather than heat.
How to Synthesize Quinoline Nitrile Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the use of standard equipment and readily available reagents to ensure reproducibility. The process is designed to be modular, allowing for the easy substitution of different haloalkanes or aromatic aldehydes to generate a library of derivatives with tailored properties. Detailed standard operating procedures for each reaction stage, including specific reflux times and solvent ratios, are critical for maintaining batch-to-batch consistency. For a comprehensive guide on the exact stoichiometry, temperature profiles, and workup procedures required to replicate these results, please refer to the standardized synthesis steps provided below.
- Perform quaternization of 2-methylquinoline with a haloalkane in acetonitrile under reflux to form the N-substituted quinolinium salt.
- Execute Michael addition-elimination using malononitrile in an ethanol/acetic acid system to generate the intermediate quinoline nitrile.
- Complete the synthesis via Knoevenagel condensation with an aromatic aldehyde using piperidine as a catalyst in acetonitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this new class of quinoline nitrile derivatives represents a strategic opportunity to optimize costs and mitigate supply risks associated with complex optoelectronic materials. The synthesis route is characterized by its reliance on commodity chemicals rather than specialized, high-cost catalysts or rare earth metals, which significantly stabilizes the raw material cost base. The elimination of transition metal catalysts in the key bond-forming steps removes the need for expensive and time-consuming metal scavenging processes, which are often a bottleneck in the purification of pharmaceutical and electronic intermediates. This simplification of the downstream processing directly translates to reduced operational expenditures and a smaller environmental footprint, aligning with increasingly stringent regulatory requirements for chemical manufacturing. Furthermore, the high yields reported in the patent examples suggest a material-efficient process that minimizes waste generation, further enhancing the overall economic viability of large-scale production.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of precious metal catalysts such as palladium or platinum, which are subject to volatile market pricing and supply constraints. By utilizing organic bases like piperidine and common solvents, the process significantly lowers the direct material costs associated with production. Additionally, the ability to isolate intermediates as solids allows for efficient purification without the need for costly chromatographic separation on a large scale, reducing solvent consumption and waste disposal costs. This streamlined approach ensures that the cost of goods sold remains competitive even as production volumes increase, providing a sustainable margin structure for long-term supply agreements.
- Enhanced Supply Chain Reliability: The starting materials, including 2-methylquinoline and various benzaldehydes, are produced by multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate standard industrial reflux temperatures and atmospheric pressure, means that the process can be easily transferred to multiple manufacturing sites without requiring specialized high-pressure or cryogenic equipment. This flexibility enhances supply continuity, allowing for rapid scaling in response to market demand fluctuations. The stability of the intermediates also facilitates inventory management, as key precursors can be stockpiled without significant degradation, ensuring a buffer against raw material supply disruptions.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional heavy-metal catalyzed cross-coupling reactions, simplifying compliance with environmental regulations. The solvents used, such as acetonitrile and ethanol, are well-established in the industry with mature recovery and recycling infrastructure, further reducing the environmental impact. The high atom economy of the Knoevenagel condensation step ensures that most of the reactant mass is incorporated into the final product, minimizing waste. This green chemistry profile not only reduces disposal costs but also aligns with the sustainability goals of major electronics manufacturers, making these derivatives a preferred choice for eco-conscious supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these quinoline nitrile derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating these materials into your existing product lines or R&D pipelines.
Q: What is the primary advantage of these quinoline nitrile derivatives over traditional DCM dyes?
A: Unlike traditional DCM derivatives which suffer from fluorescence quenching in the solid state, these quinoline nitrile derivatives exhibit strong aggregation-induced emission (AIE), maintaining high quantum yield in solid or aggregated forms.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the synthesis utilizes readily available starting materials such as 2-methylquinoline, common haloalkanes, malononitrile, and substituted benzaldehydes, ensuring supply chain stability.
Q: Is this synthesis route suitable for large-scale industrial production?
A: The patent explicitly highlights the simplicity of the synthesis steps and the use of common solvents like acetonitrile and ethanol, indicating high feasibility for commercial scale-up without complex purification requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Nitrile Derivative Supplier
The technical potential of these aggregation-induced emission materials is immense, yet realizing their commercial value requires a partner with deep expertise in process chemistry and scale-up engineering. NINGBO INNO PHARMCHEM stands as a premier CDMO partner, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific solvent systems and purification requirements of these quinoline derivatives, ensuring that every batch meets stringent purity specifications. With rigorous QC labs and a commitment to process safety, we guarantee the consistency and quality required for high-performance optoelectronic applications, bridging the gap between patent innovation and industrial reality.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and cost requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into how our optimized manufacturing processes can reduce your overall material costs. We encourage you to contact us to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
