Advanced Polyamide Resin Purification for High-Purity PQQ Commercial Manufacturing
The biotechnology sector is constantly seeking more efficient pathways to produce high-value coenzymes, and Pyrroloquinoline Quinone (PQQ) stands out as a molecule of immense potential for both pharmaceutical and functional food applications. As detailed in patent CN110698472B, a novel purification method has been developed that leverages polyamide resin adsorption coupled with crystallization to achieve exceptional purity standards. This technical breakthrough addresses the longstanding challenges of cost and scalability associated with traditional extraction methods. By optimizing the interaction between the fermentation broth and the resin matrix, manufacturers can now secure a supply of PQQ that meets the rigorous demands of a reliable nutritional ingredients supplier. The structural integrity of the molecule is preserved throughout this aqueous-based process, ensuring that the final product retains its potent redox cycling capabilities.
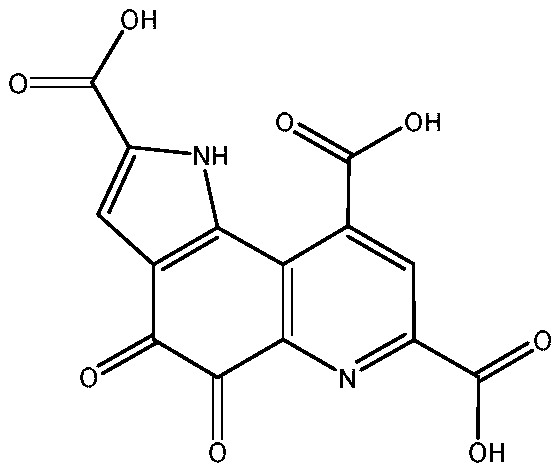
PQQ, chemically known as 4,5-dihydro-4,5-dioxy-1-hydropyrrole (2,3f)quinone-2,7,9-tricarboxylic acid, is a novel coenzyme that functions as a redox cofactor for membrane-bound dehydrogenases. Its ability to facilitate electron transport in the respiratory chain makes it a critical component in cellular energy metabolism. Recent scientific inquiries have highlighted its abnormally high redox cycling capacity, suggesting significant applications in anti-aging therapies and neuroprotection. With the U.S. Food and Drug Administration (FDA) having approved foods promoting cognitive function with PQQ as a main ingredient, the market demand for high-purity PQQ is surging. However, bridging the gap between microbial fermentation and commercial availability requires a purification strategy that is both economically viable and environmentally sustainable. The method described in the referenced patent offers a robust solution to these industrial bottlenecks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of PQQ from fermentation broth has relied on techniques that are fraught with inefficiencies and high operational expenditures. Traditional approaches often utilize DEAE-dextran gel or Seppak C18 columns for adsorption and desorption. While these materials can achieve separation, they are prohibitively expensive for large-scale manufacturing, driving up the cost reduction in nutritional ingredients manufacturing significantly. Furthermore, alternative methods involving complex extraction or ion-pair aqueous two-phase extraction require substantial volumes of organic solvents such as n-hexane, methanol, or ethanol. This reliance on volatile organic compounds not only inflates the raw material costs but also imposes a heavy burden on waste management systems due to high Chemical Oxygen Demand (COD) in the effluent. Additionally, resins like non-polar macroporous types often suffer from dead adsorption, necessitating regeneration with high-concentration organic solvents like 95% ethanol, which requires energy-intensive distillation for recovery.
The Novel Approach
In stark contrast, the novel approach utilizing polyamide resin presents a paradigm shift in downstream processing for complex nutritional ingredients. This method replaces costly chromatographic fillers with polyamide resin, which is cheap, easy to obtain, and highly effective for PQQ separation. The process is designed to operate primarily in an aqueous environment, drastically simplifying the workflow and reducing the need for hazardous organic solvents. A key innovation lies in the regeneration protocol; the used polyamide resin can be effectively restored using a simple alkaline solution, such as sodium hydroxide, rather than requiring large volumes of high-purity ethanol. This shift not only lowers the direct material costs but also mitigates the explosion-proof requirements for factory facilities, thereby enhancing overall plant safety. The result is a streamlined process that yields crystals with HPLC purity greater than or equal to 98% and content greater than or equal to 96%.
Mechanistic Insights into Polyamide Resin Adsorption and Desorption
The core of this purification technology lies in the precise control of pH and ionic strength to manipulate the adsorption and desorption equilibrium on the polyamide resin surface. During the adsorption phase, the fermentation broth filtrate is adjusted to an acidic pH range of 2.0 to 2.5. At this acidity, combined with the addition of salts like calcium chloride or calcium acetate at concentrations of 5 to 15 g/L, the PQQ molecules are optimally conditioned for binding to the resin matrix. Experimental data indicates that under these specific conditions, the resin achieves a static adsorption capacity of approximately 8 to 8.8 g/L. This high loading capacity is crucial for commercial scale-up of complex nutritional ingredients, as it maximizes the throughput of each batch while minimizing the volume of resin required. The acidic environment ensures that impurities with different isoelectric points remain in the solution or are washed away, setting the stage for high-purity PQQ isolation.
Desorption is equally critical and is achieved by shifting the chemical environment to a near-neutral pH of 6.5 ± 0.2 using a mixed aqueous solution of acid and ammonium acetate. The addition of ammonium acetate at 2.5 g/L acts as a buffer that disrupts the interaction between the PQQ and the polyamide resin without degrading the product. This specific pH window allows for the collection of desorption liquid with a chromatographic purity exceeding 90% in a concentrated volume. Following desorption, the process employs ultrafiltration with a 10,000 Dalton membrane to remove macromolecular impurities, followed by nanofiltration with a 100 to 200 Dalton membrane for concentration. This membrane cascade effectively removes residual salts and small molecule contaminants, ensuring that the final crystallization step yields a product of exceptional quality. The final crystallization, induced by adjusting pH to 3.5 and adding NaCl, precipitates the PQQ in a highly pure crystalline form.
How to Synthesize Pyrroloquinoline Quinone Efficiently
Implementing this synthesis route requires strict adherence to the parameters outlined in the patent to ensure reproducibility and quality. The process begins with the pretreatment of the fermentation broth, where solid-liquid separation is performed using ceramic membrane filtration with a pore size of 50 nm. This initial clarification is vital for protecting the downstream resin column from fouling. The subsequent steps involve a carefully choreographed sequence of pH adjustments, salt additions, and membrane filtrations that work in concert to isolate the target molecule. For R&D teams looking to replicate or scale this process, understanding the interplay between the resin particle size (200 to 500 mesh) and the flow rates is essential. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational parameters.
- Pretreat fermentation broth by adjusting pH to 2.0-2.5 and adding salt (5-15g/L) before filtration.
- Pass filtrate through polyamide resin, wash with acidic solution, and desorb using acid-salt mixed solution at pH 6.5.
- Adjust desorption liquid pH, perform ultrafiltration and nanofiltration concentration, then crystallize and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this polyamide resin-based purification method offers substantial strategic benefits beyond mere technical feasibility. The primary advantage is the significant reduction in manufacturing costs driven by the replacement of expensive chromatographic media with affordable polyamide resin. Unlike traditional methods that rely on consumables with limited lifecycles, this resin can be regenerated multiple times using inexpensive alkaline solutions, extending its operational life and reducing the frequency of replacement purchases. This durability translates directly into a more stable cost structure for high-purity PQQ production. Furthermore, the reduction in organic solvent usage simplifies the supply chain logistics, as there is less need to procure, store, and handle large quantities of flammable chemicals like ethanol or methanol.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the elimination of costly solvents and resins. By utilizing a regeneration method that relies on sodium hydroxide rather than high-concentration ethanol, the operational expenditure on consumables is drastically simplified. The ability to recover the resin's performance without energy-intensive distillation processes means that utility costs are also kept in check. This qualitative shift in the cost base allows for more competitive pricing strategies in the global market for nutritional ingredients. The removal of expensive transition metals or specialized gels further ensures that the cost of goods sold remains low, providing a buffer against raw material price fluctuations.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents. Polyamide resin is a commodity chemical that is cheap and easy to obtain from multiple sources, reducing the risk of single-supplier dependency. The robustness of the process against variations in fermentation broth quality also means that yield consistency is higher, leading to more predictable production schedules. Reducing lead time for high-purity PQQ is achieved through the simplicity of the workflow; fewer steps and faster regeneration cycles mean that batches can be turned around more quickly. This agility allows manufacturers to respond rapidly to spikes in market demand without compromising on quality or delivery commitments.
- Scalability and Environmental Compliance: Scaling this process from pilot to industrial levels is straightforward because it avoids the complexities associated with handling large volumes of hazardous organic solvents. The significant reduction in wastewater COD makes compliance with environmental regulations much easier to achieve and maintain. Lower explosion-proof requirements for factory facilities reduce the capital expenditure needed for plant construction and retrofitting. This environmental efficiency not only mitigates regulatory risk but also aligns with the sustainability goals of modern enterprise clients. The process is inherently greener, which is an increasingly important factor for procurement decisions in the pharmaceutical and food industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the purification of Pyrroloquinoline Quinone using this advanced resin method. These answers are derived directly from the experimental data and beneficial effects described in the patent literature. They are intended to provide clarity on the feasibility, quality, and operational advantages of this specific technological route. Understanding these details is crucial for stakeholders evaluating the potential for integration into their existing manufacturing frameworks.
Q: Why is polyamide resin preferred over DEAE or HP20 resins for PQQ purification?
A: Polyamide resin is significantly cheaper and easier to obtain than DEAE-dextran gel or HP20 resin. Crucially, it can be regenerated using simple alkaline solutions, drastically reducing the need for high-concentration organic solvents like 95% ethanol, which lowers both operational costs and environmental COD load.
Q: What purity levels can be achieved with this purification method?
A: This method consistently yields Pyrroloquinoline Quinone crystals with an HPLC purity of greater than or equal to 98% and a content of greater than or equal to 96%, meeting stringent requirements for high-purity nutritional ingredients and pharmaceutical intermediates.
Q: How does this process impact environmental compliance and safety?
A: By minimizing the use of organic solvents during resin regeneration and replacing them with aqueous alkaline solutions, the Chemical Oxygen Demand (COD) of wastewater is greatly reduced. This also lowers the explosion-proof requirements for factory facilities, enhancing overall operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrroloquinoline Quinone Supplier
The technical potential of this polyamide resin purification route is immense, offering a clear path to high-quality PQQ production that balances performance with economic efficiency. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of PQQ meets the exacting standards of the global market. We understand the nuances of fermentation downstream processing and can optimize this resin-based method to maximize yield and minimize waste for your specific needs.
We invite you to collaborate with us to unlock the full value of this purification technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volumes. We encourage you to reach out to request specific COA data and route feasibility assessments to see how we can support your supply chain goals. By partnering with us, you gain access to a reliable nutritional ingredients supplier committed to innovation and quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
