Scalable Synthesis of Pyrotinib Intermediate via Novel NCS/TEMPO Oxidation Technology
Introduction to Advanced Pyrotinib Intermediate Manufacturing
The escalating global demand for next-generation anticancer therapeutics has placed intense scrutiny on the supply chains of critical kinase inhibitor intermediates. Patent CN114920683A introduces a transformative preparation method for (R, E) - (1-methylpyrrolidin-2-yl) acrylic acid, a pivotal fragment in the synthesis of the innovative antitumor drug Pyrotinib. This technology addresses long-standing bottlenecks in proline derivative synthesis by replacing hazardous oxidants with a benign NCS/TEMPO system. The structural integrity and stereochemical purity of this molecule are paramount for biological activity, necessitating a manufacturing process that guarantees consistency from gram to ton scale. 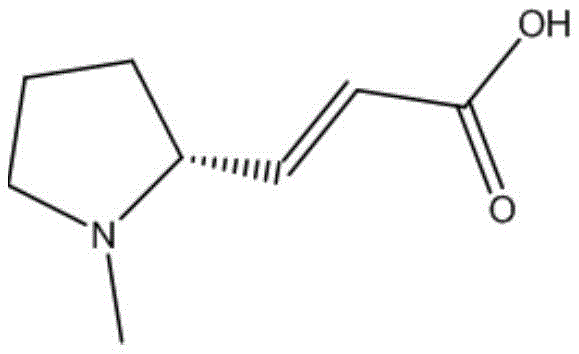 This patent represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, offering a pathway that mitigates the risks associated with traditional heavy metal oxidations and unstable hypochlorite reactions. By leveraging this intellectual property, manufacturers can secure a more resilient supply of high-purity building blocks essential for oncology drug development.
This patent represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, offering a pathway that mitigates the risks associated with traditional heavy metal oxidations and unstable hypochlorite reactions. By leveraging this intellectual property, manufacturers can secure a more resilient supply of high-purity building blocks essential for oncology drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Boc-prolinaldehyde and its derivatives has been plagued by severe operational and environmental constraints that hinder commercial viability. Early methodologies, such as those disclosed in WO2017186140A1, relied heavily on Pyridinium Chlorochromate (PCC) for the oxidation of alcohol precursors. While chemically effective on a small scale, PCC introduces toxic chromium waste streams that are increasingly prohibited under modern green chemistry regulations, creating massive disposal costs and regulatory hurdles for large-scale facilities. Furthermore, alternative routes utilizing sodium bromide and sodium hypochlorite systems, as seen in CN108314639B, exhibit extreme sensitivity to pH fluctuations. 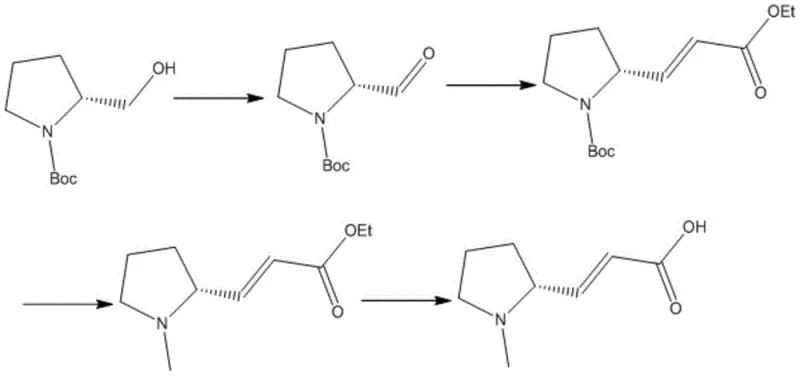 In these hypochlorite-mediated processes, even minor deviations in reaction time or acidity during amplification lead to rampant over-oxidation, causing yield collapses that render the process economically unfeasible for industrial production. Additionally, routes requiring catalytic hydrogenation impose rigorous equipment demands, necessitating high-pressure reactors that increase capital expenditure and safety risks, thereby limiting the agility of the supply chain.
In these hypochlorite-mediated processes, even minor deviations in reaction time or acidity during amplification lead to rampant over-oxidation, causing yield collapses that render the process economically unfeasible for industrial production. Additionally, routes requiring catalytic hydrogenation impose rigorous equipment demands, necessitating high-pressure reactors that increase capital expenditure and safety risks, thereby limiting the agility of the supply chain.
The Novel Approach
The methodology outlined in CN114920683A fundamentally reengineers the oxidation step to overcome these scalability barriers through the implementation of an N-Chlorosuccinimide (NCS) and TEMPO catalytic system. This novel approach operates effectively in a mild alkaline environment at room temperature, eliminating the need for cryogenic cooling or high-pressure infrastructure. By utilizing NCS as the terminal oxidant, the process avoids the generation of heavy metal sludge associated with chromium reagents, significantly simplifying the downstream purification workflow. The reaction demonstrates remarkable robustness, maintaining high conversion rates even when scaled from laboratory flasks to multi-kilogram batches, directly addressing the yield instability observed in prior art. This technological shift not only enhances the safety profile of the manufacturing site but also ensures a consistent supply of the critical aldehyde intermediate, which is subsequently converted to the target acrylic acid via a streamlined Horner-Wadsworth-Emmons olefination and deprotection sequence.
Mechanistic Insights into NCS/TEMPO Oxidation Chemistry
The core innovation of this process lies in the synergistic interaction between the nitroxyl radical TEMPO and the chlorinating agent NCS within a biphasic or alkaline aqueous-organic system. Mechanistically, TEMPO acts as a catalytic mediator that selectively abstracts hydrogen from the hydroxymethyl group of Boc-prolinol, forming an oxoammonium species which is subsequently regenerated by NCS. This cycle proceeds with high chemoselectivity, stopping precisely at the aldehyde stage without progressing to the carboxylic acid, a common side reaction in less controlled oxidations. The presence of a phase transfer catalyst, such as tetrabutylammonium chloride, further accelerates the reaction kinetics by facilitating the transport of ionic species across the interface, ensuring uniform reaction progress throughout the bulk mixture. This precise control over the oxidation state is critical for minimizing the formation of over-oxidized impurities that are notoriously difficult to separate from the desired product.
Impurity control is further enhanced by the specific choice of alkaline buffers, such as sodium acetate or sodium bicarbonate, which maintain the reaction pH within a narrow optimal window. Unlike hypochlorite systems where pH drift can trigger explosive decomposition or non-selective chlorination, the NCS/TEMPO system remains stable over extended reaction times, typically ranging from 0.5 to 5 hours. The workup procedure involves a strategic quenching step using sodium sulfite to reduce residual oxidants, followed by washes with sodium carbonate and brine to remove succinimide by-products and inorganic salts. This rigorous purification protocol ensures that the resulting Boc-prolinaldehyde possesses the high chemical purity required for the subsequent carbon-carbon bond-forming steps, thereby preventing the carryover of impurities that could compromise the stereochemical integrity of the final Pyrotinib intermediate.
How to Synthesize (R,E)-(1-Methylpyrrolidin-2-yl)acrylic Acid Efficiently
The synthesis of this high-value intermediate is achieved through a concise three-stage sequence that prioritizes operational simplicity and yield maximization. The process initiates with the oxidation of Boc-prolinol using the aforementioned NCS/TEMPO system to generate the key aldehyde intermediate in high yield. This aldehyde is then subjected to a Horner-Wadsworth-Emmons reaction with triethyl phosphonoacetate under basic conditions to install the acrylic ester moiety with excellent E-selectivity. Following N-methylation and ester hydrolysis, the final acid is isolated with exceptional purity. For detailed standard operating procedures and exact stoichiometric ratios, please refer to the step-by-step guide below.
- Oxidize Boc-prolinol using an NCS/TEMPO system in an alkaline environment to generate Boc-prolinaldehyde with high stability.
- Perform a Horner-Wadsworth-Emmons reaction with triethyl phosphonoacetate followed by N-methylation to form the unsaturated ester.
- Execute acidic hydrolysis to remove the protecting group and yield the final (R,E)-(1-methylpyrrolidin-2-yl)acrylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages that extend beyond mere technical feasibility. By transitioning away from regulated heavy metal oxidants and unstable hypochlorite chemistries, organizations can drastically simplify their vendor qualification processes and reduce the regulatory burden associated with environmental compliance. The elimination of chromium waste streams removes the need for specialized hazardous waste disposal contractors, leading to substantial cost savings in operational overhead. Furthermore, the robustness of the NCS/TEMPO system ensures that production schedules are not disrupted by batch failures due to over-oxidation, a frequent occurrence in legacy methods that often leads to costly delays and inventory shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction infrastructure and waste management protocols. Since the reaction proceeds efficiently at room temperature, there is no requirement for energy-intensive heating or cooling systems, resulting in significantly reduced utility consumption per kilogram of product. Additionally, the avoidance of expensive transition metal catalysts and the use of readily available commodity chemicals like NCS and TEMPO lower the raw material cost basis. The high yield and purity achieved in the oxidation step also minimize the loss of valuable chiral starting materials, ensuring that the overall cost of goods sold is optimized for competitive market positioning.
- Enhanced Supply Chain Reliability: Supply continuity is critically dependent on the scalability of the manufacturing process, and this technology excels in translating laboratory success to commercial production. The method's tolerance to minor variations in reaction parameters means that manufacturing partners can reliably produce large batches without the risk of catastrophic yield drops that plague sensitive hypochlorite routes. This stability allows for more accurate forecasting and inventory planning, reducing the need for excessive safety stock. Moreover, the reagents employed are globally sourced and not subject to the same supply constraints as specialized catalytic systems, ensuring that production lines remain operational even during periods of raw material volatility.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route aligns perfectly with the increasing stringency of global chemical regulations. The absence of heavy metals and the use of milder oxidants significantly reduce the toxicity profile of the effluent, simplifying wastewater treatment and lowering the risk of regulatory fines. The process is inherently safer, operating at ambient pressure and temperature, which reduces the likelihood of thermal runaways or pressure vessel incidents. These factors collectively enhance the long-term sustainability of the supply chain, making it easier to obtain necessary environmental permits and maintain social license to operate in key manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational requirements. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Why is the NCS/TEMPO system superior to traditional PCC oxidation for this intermediate?
A: Traditional PCC oxidation generates significant chromium waste and poses environmental hazards. The NCS/TEMPO system operates under mild, alkaline conditions at room temperature, eliminating heavy metal contamination and simplifying waste treatment protocols.
Q: How does this method address the scale-up yield issues seen in hypochlorite-based routes?
A: Prior art using sodium hypochlorite often suffers from strict pH requirements and over-oxidation during amplification. The patented NCS/TEMPO method demonstrates consistent yields (e.g., >90% in lab scale) and stable performance during scale-up, ensuring reliable commercial production.
Q: What are the critical purity specifications for this Pyrotinib intermediate?
A: High purity is essential for downstream API synthesis. The described process utilizes efficient extraction and washing steps (e.g., sodium sulfite quenching) to remove oxidants and by-products, achieving purity levels suitable for stringent pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R,E)-(1-Methylpyrrolidin-2-yl)acrylic Acid Supplier
As the pharmaceutical industry continues to evolve towards more complex small molecule therapies, the need for partners who can navigate intricate synthetic challenges has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in process chemistry to deliver high-value intermediates with unmatched consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your clinical and commercial needs are met with precision. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of (R,E)-(1-Methylpyrrolidin-2-yl)acrylic acid meets the exacting standards required for oncology drug substance manufacturing.
We invite you to collaborate with us to optimize your supply chain for Pyrotinib and related kinase inhibitors. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and value in your drug development programs.
